2 Protons 2 Neutrons 2 Electrons
Muz Play
Apr 02, 2025 · 6 min read
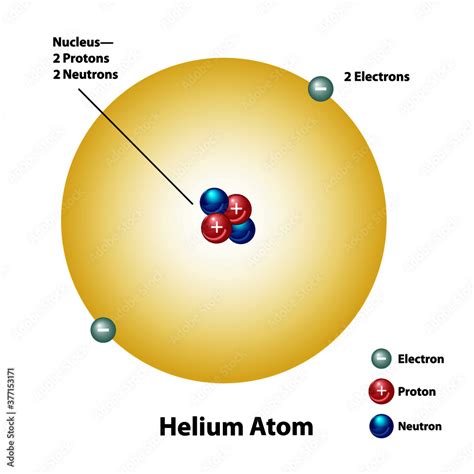
Table of Contents
Delving Deep into the 2 Protons, 2 Neutrons, 2 Electrons Configuration: Helium and Beyond
The seemingly simple arrangement of 2 protons, 2 neutrons, and 2 electrons defines a fundamental building block of our universe: the helium atom. While seemingly straightforward, this configuration holds profound implications for the structure of matter, the evolution of stars, and even the existence of life as we know it. This article will explore the intricacies of this specific atomic configuration, examining its properties, its role in larger systems, and its significance across various scientific disciplines.
Understanding the Basics: Protons, Neutrons, and Electrons
Before diving into the specifics of the 2-2-2 configuration, let's refresh our understanding of the fundamental subatomic particles:
-
Protons: Positively charged particles residing in the atom's nucleus. The number of protons defines an element's atomic number and its chemical identity. In our case, two protons signify helium.
-
Neutrons: Neutral particles (no charge) also located in the nucleus. They contribute to the atom's mass but not its charge. The number of neutrons can vary within an element, leading to isotopes.
-
Electrons: Negatively charged particles orbiting the nucleus in electron shells. They are significantly lighter than protons and neutrons. The number of electrons usually equals the number of protons in a neutral atom, determining its chemical reactivity.
Helium: The Archetypal 2-2-2 Atom
Helium, with its 2 protons, 2 neutrons, and 2 electrons, represents the quintessential example of this atomic configuration. Its properties are directly linked to this specific arrangement:
-
Inertness: Helium's two electrons completely fill its first electron shell, resulting in exceptional chemical inertness. It rarely forms chemical bonds with other elements, making it a noble gas. This stability is a direct consequence of its filled electron shell, a fundamental principle in chemistry.
-
Low Density: Helium possesses the lowest density of all gases, except for hydrogen. This is due to the small mass of its nucleus and the relatively large distance between its nucleus and electrons. This low density has numerous practical applications, from filling balloons to cooling superconducting magnets.
-
Low Boiling Point: Helium has an exceptionally low boiling point, remaining a gas even at extremely low temperatures. This is a consequence of the weak interatomic forces between helium atoms, again a direct result of its electron configuration and resulting stability.
Isotopes of Helium: Variations on a Theme
While the most common isotope of helium has 2 neutrons, isotopes with different neutron counts exist. These isotopes, still possessing 2 protons and 2 electrons, exhibit variations in their properties, primarily their mass and stability. For instance:
-
Helium-3 (³He): Contains 1 neutron. It's a stable isotope but much less abundant than helium-4.
-
Helium-4 (⁴He): Contains 2 neutrons. This is the most abundant and stable isotope of helium.
The difference in neutron number affects the isotope's mass and, in some cases, its nuclear stability. While both isotopes remain chemically inert due to their identical electron configuration, their physical properties, such as density and nuclear stability, differ.
The Role of Helium in Stellar Nucleosynthesis
The creation of helium, specifically helium-4, plays a crucial role in the life cycle of stars. Stellar nucleosynthesis, the process by which elements are formed within stars, begins with the fusion of hydrogen nuclei (protons) to form helium. This process releases immense energy, powering the stars' luminosity for billions of years. This proton-proton chain reaction is central to the energy production in stars like our Sun.
The formation of helium-4 through nuclear fusion represents a milestone in stellar evolution, marking the transition from hydrogen burning to subsequent stages of nuclear fusion involving heavier elements. Understanding the properties of helium, specifically its stability and abundance, is crucial to modelling stellar evolution and the distribution of elements in the universe.
Helium's Applications: From Balloons to MRI Machines
The unique properties of helium, stemming directly from its 2-2-2 configuration, have led to a wide range of applications:
-
Balloons and Airships: Its low density makes it ideal for lifting objects in the air.
-
Deep-Sea Diving: Helium-oxygen mixtures are used in deep-sea diving to prevent decompression sickness.
-
Cryogenics: Helium's extremely low boiling point makes it essential for cooling superconducting magnets in MRI machines, particle accelerators, and other scientific instruments.
-
Leak Detection: Its low density and inertness make it suitable for leak detection in various industrial applications.
-
Welding: Helium is used as a shielding gas in welding processes.
Beyond Helium: The Significance of the 2-2-2 Configuration in Nuclear Physics
The 2-2-2 configuration's importance transcends the realm of chemistry and extends into the field of nuclear physics. The alpha particle, a highly stable particle consisting of 2 protons and 2 neutrons, is essentially a helium-4 nucleus. Alpha decay, a type of radioactive decay, involves the emission of an alpha particle from an unstable nucleus. The stability of the alpha particle directly relates to the strong nuclear force holding its constituents together. Understanding the binding energy and stability of this configuration is crucial for comprehending radioactive decay processes and nuclear reactions.
The 2-2-2 Configuration and the Periodic Table: A Foundation for Atomic Structure
The helium atom, with its 2-2-2 configuration, occupies a unique position at the beginning of the periodic table. Its filled electron shell provides a baseline for understanding the electron configurations and chemical properties of other elements. The subsequent elements build upon this foundation, adding more protons, neutrons, and electrons, resulting in increasingly complex electron configurations and diverse chemical properties. The simple 2-2-2 arrangement serves as a fundamental building block upon which the elaborate structure of the periodic table and the vast diversity of matter are built.
Future Research and Applications: Exploring the Potential
The study of the 2-2-2 configuration and its implications continues to evolve. Research in areas such as:
-
Nuclear fusion: Harnessing the energy released during helium formation is a major goal in the pursuit of sustainable energy.
-
Quantum computing: Helium's unique properties may play a role in the development of future quantum computing technologies.
-
Material science: The study of helium's behavior in extreme conditions could lead to advancements in material science.
This simple yet profoundly significant atomic arrangement, represented by the 2 protons, 2 neutrons, and 2 electrons of helium, serves as a cornerstone for our understanding of the universe, from the smallest atomic scales to the vast expanse of stars and galaxies. Its seemingly simple structure belies a complexity that continues to fascinate and challenge scientists, revealing deeper insights into the fundamental laws governing our physical world. The future promises further discoveries and innovative applications built upon our ever-increasing knowledge of this foundational building block of matter.
Latest Posts
Latest Posts
-
What Are The Four Main Types Of Context
Apr 03, 2025
-
How To Know If A Graph Is Symmetric
Apr 03, 2025
-
Is Blood Clotting Negative Or Positive Feedback
Apr 03, 2025
-
Evaluating Functions Linear And Quadratic Or Cubic
Apr 03, 2025
-
What Are The Reactants In The Calvin Cycle
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about 2 Protons 2 Neutrons 2 Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
