Amino Acids Can Be Distinguished From One Another By
Muz Play
Apr 03, 2025 · 6 min read
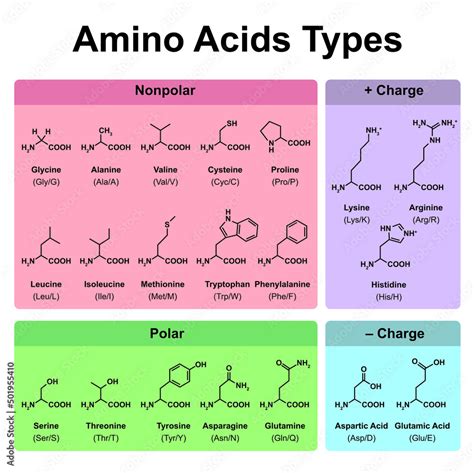
Table of Contents
Amino Acids: A Detailed Look at Their Distinguishing Features
Amino acids, the fundamental building blocks of proteins, are remarkable molecules with diverse properties and crucial roles in biological systems. While they share a common core structure, it's the subtle variations in their side chains (also known as R-groups) that distinguish them from one another. Understanding these distinctions is critical for comprehending protein structure, function, and the intricacies of biological processes. This article will delve into the various ways amino acids can be differentiated, emphasizing the importance of their side chains and their impact on protein characteristics.
The Basic Amino Acid Structure: A Common Ground
Before exploring the differences, let's establish the common ground. All amino acids, with the exception of proline (which has a unique cyclic structure), share a basic structure consisting of:
- A central carbon atom (α-carbon): This carbon atom is chiral (except for glycine), meaning it's bonded to four different groups.
- An amino group (-NH₂): This group is basic and readily accepts protons.
- A carboxyl group (-COOH): This group is acidic and readily donates protons.
- A hydrogen atom (-H): A simple hydrogen atom bonded to the α-carbon.
- A variable side chain (R-group): This is the unique group that differentiates one amino acid from another. The R-group's properties dictate the amino acid's characteristics and its role in protein structure and function.
Distinguishing Amino Acids: Focusing on the R-Group
The remarkable diversity of amino acids stems from the variety of R-groups. These side chains vary drastically in size, shape, charge, polarity, and chemical reactivity. These differences are the key to distinguishing one amino acid from another and understanding their individual functions within proteins. We can categorize amino acids based on several characteristics of their R-groups:
1. Classification based on Polarity and Charge:
This is arguably the most common and useful classification system. It divides amino acids into four main categories:
-
Nonpolar, aliphatic amino acids: These amino acids have hydrocarbon side chains that are hydrophobic (water-repelling). They tend to cluster in the interior of proteins, away from the aqueous environment. Examples include Glycine (Gly, G), Alanine (Ala, A), Valine (Val, V), Leucine (Leu, L), Isoleucine (Ile, I), and Methionine (Met, M). Glycine, being the smallest, is unique in its flexibility, often found in areas requiring conformational flexibility. Methionine, with its sulfur atom, plays a role in protein initiation during translation.
-
Aromatic amino acids: These amino acids possess aromatic rings in their side chains. They are relatively nonpolar but can participate in hydrophobic interactions. Their aromatic rings can also absorb UV light, a property exploited in protein analysis. Examples include Phenylalanine (Phe, F), Tyrosine (Tyr, Y), and Tryptophan (Trp, W). Tyrosine can be phosphorylated, adding another layer of regulation.
-
Polar, uncharged amino acids: These amino acids have side chains that are hydrophilic (water-loving) due to the presence of polar functional groups such as hydroxyl (-OH), thiol (-SH), or amide (-CONH₂) groups. They tend to be located on the surface of proteins, interacting with the aqueous environment. Examples include Serine (Ser, S), Threonine (Thr, T), Cysteine (Cys, C), Asparagine (Asn, N), and Glutamine (Gln, Q). Cysteine's thiol group can form disulfide bonds, crucial for protein structure stabilization.
-
Charged amino acids: These amino acids have side chains with a net positive or negative charge at physiological pH. These charges strongly influence protein folding and interactions.
-
Acidic amino acids: These amino acids have negatively charged carboxyl groups in their side chains at physiological pH. Examples include Aspartic acid (Asp, D) and Glutamic acid (Glu, E).
-
Basic amino acids: These amino acids have positively charged amino groups in their side chains at physiological pH. Examples include Lysine (Lys, K), Arginine (Arg, R), and Histidine (His, H). Histidine's imidazole ring can act as a buffer, important for pH regulation in enzymes.
-
2. Classification based on Chemical Properties of the R-Group:
Further distinctions can be made based on the specific chemical functionalities present within the R-group. This allows for a more nuanced understanding of the amino acid's reactivity and potential interactions:
-
Hydroxyl-containing amino acids: Serine, threonine, and tyrosine contain hydroxyl groups, making them susceptible to phosphorylation, a crucial post-translational modification that regulates protein activity.
-
Sulfur-containing amino acids: Methionine and cysteine contain sulfur atoms. Methionine is involved in initiation of protein synthesis, while cysteine's thiol group can form disulfide bonds, stabilizing protein tertiary structure.
-
Amino acids with amide groups: Asparagine and glutamine possess amide groups, contributing to their polarity and hydrogen bonding capabilities.
-
Imino acid: Proline is unique, possessing an imino group instead of an amino group, leading to a rigid cyclic structure that affects protein backbone conformation.
3. Classification based on Nutritional Requirements:
Amino acids are also categorized based on whether the human body can synthesize them (non-essential) or whether they must be obtained from the diet (essential). Essential amino acids cannot be synthesized de novo and are crucial for various physiological functions:
-
Essential amino acids: Histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.
-
Non-essential amino acids: Alanine, asparagine, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, and tyrosine. Note that while these are considered non-essential, they still play vital roles and their availability is crucial for optimal health. Some non-essential amino acids can become conditionally essential under specific circumstances (e.g., illness or malnutrition).
The Impact of R-Group Differences on Protein Properties
The diverse characteristics of amino acid R-groups significantly influence the overall properties and functions of the proteins they constitute:
-
Protein folding: The interactions between amino acid side chains (hydrophobic interactions, hydrogen bonds, ionic bonds, disulfide bonds) drive protein folding into its unique three-dimensional structure. The arrangement of amino acids within the protein sequence dictates its final conformation.
-
Protein stability: The strength and nature of the interactions between R-groups contribute to the stability of the folded protein. Disulfide bonds, in particular, provide significant stability.
-
Protein function: The specific arrangement and chemical properties of R-groups at the protein's active site determine its enzymatic activity, binding specificity, and other functional roles. For example, the charged R-groups may be crucial for interacting with specific substrates or for binding to other molecules.
-
Post-translational modifications: Some R-groups are susceptible to various post-translational modifications (PTMs) such as phosphorylation, glycosylation, and acetylation. These modifications often alter protein function and activity.
Analytical Techniques for Distinguishing Amino Acids
Several analytical techniques allow scientists to identify and quantify amino acids in samples:
-
Chromatography: Techniques like high-performance liquid chromatography (HPLC) and gas chromatography (GC) separate amino acids based on their physical and chemical properties, allowing for identification and quantification.
-
Mass spectrometry: This technique measures the mass-to-charge ratio of ionized molecules, providing information about the amino acid's mass and structure.
Conclusion
The ability to distinguish amino acids from one another is fundamental to understanding protein structure, function, and the broader intricacies of biological processes. While they share a basic structural framework, it is the diversity of their R-groups that defines their individual characteristics, influencing protein folding, stability, function, and susceptibility to post-translational modifications. The various classification systems based on polarity, charge, chemical properties, and nutritional requirements provide a comprehensive understanding of these vital biomolecules. Further exploration into the specific properties of each amino acid is essential for advancing our understanding of biological systems and developing new therapeutic interventions.
Latest Posts
Latest Posts
-
Area Between Curves With Respect To Y
Apr 04, 2025
-
Where In Eukaryotic Cells Does The Calvin Cycle Take Place
Apr 04, 2025
-
6 Characteristics Of Family Systems Theory
Apr 04, 2025
-
Difference Between A Triglyceride And A Phospholipid
Apr 04, 2025
-
How To Do Alternating Series Test
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Amino Acids Can Be Distinguished From One Another By . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
