Amino Acids With Ionizable Side Chains
Muz Play
Apr 05, 2025 · 7 min read
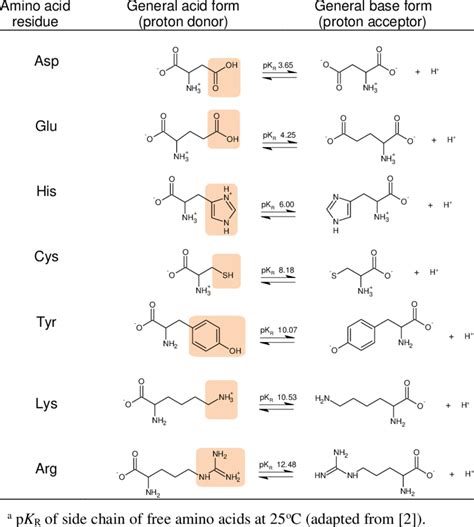
Table of Contents
Amino Acids with Ionizable Side Chains: A Deep Dive
Amino acids are the fundamental building blocks of proteins, and their unique properties significantly influence the structure and function of these macromolecules. While all amino acids share a common core structure – a central carbon atom (α-carbon) bonded to an amino group (-NH₂), a carboxyl group (-COOH), a hydrogen atom (-H), and a side chain (R group) – it's the diversity of these side chains that imparts the remarkable versatility of proteins. A particularly intriguing subset of amino acids possesses ionizable side chains, meaning these R groups can gain or lose a proton (H⁺) depending on the surrounding pH. This ionization significantly impacts their charge and, consequently, their interactions within a protein and with its environment. Understanding these ionizable side chains is crucial for comprehending protein structure, function, and their roles in various biological processes.
The Importance of Ionizable Side Chains in Protein Structure and Function
The ionizable side chains of certain amino acids play a pivotal role in determining the overall charge of a protein. This charge distribution is essential for various aspects of protein behavior, including:
1. Protein Folding and Stability
The electrostatic interactions between ionizable side chains are crucial for driving protein folding into their native three-dimensional structures. Attractive forces between oppositely charged residues (e.g., salt bridges between acidic and basic side chains) stabilize the folded conformation. Conversely, repulsive forces between similarly charged residues can destabilize the protein structure. The precise balance of these attractive and repulsive forces is critical for achieving the correct protein fold and maintaining its stability.
2. Enzyme Catalysis
Many enzymes rely on ionizable side chains within their active sites to facilitate catalytic activity. These residues can act as acids or bases, donating or accepting protons during the reaction mechanism. The precise pKa (acid dissociation constant) of these side chains, which is influenced by the local environment, determines their ionization state and, therefore, their catalytic efficiency.
3. Protein-Protein Interactions
Ionizable side chains contribute significantly to protein-protein interactions. Electrostatic interactions between oppositely charged residues on different protein surfaces mediate binding specificity and affinity. These interactions are crucial for a wide range of biological processes, including signal transduction, immune responses, and cellular regulation.
4. Protein-Ligand Interactions
Similar to protein-protein interactions, ionizable side chains are vital for protein-ligand binding. Ligands, such as drugs or substrates, can interact with ionizable residues on the protein surface through electrostatic forces. The precise arrangement of these charged residues within the binding site determines the binding specificity and affinity of the interaction.
Amino Acids with Ionizable Side Chains: A Detailed Examination
Let's delve into the individual amino acids with ionizable side chains, examining their characteristics and their roles in biological systems. These amino acids are typically classified into acidic, basic, and polar neutral groups, based on the charge and properties of their side chains at physiological pH (around 7.4).
Acidic Amino Acids: Aspartic Acid (Asp, D) and Glutamic Acid (Glu, E)
Aspartic acid and glutamic acid both possess carboxyl groups (-COOH) in their side chains. At physiological pH, these carboxyl groups are deprotonated, carrying a negative charge (-COO⁻). This negative charge makes them highly hydrophilic and often found on the protein surface, interacting with water molecules and other charged residues.
-
Aspartic Acid (Asp, D): Has a shorter side chain compared to glutamic acid, which can influence its interactions within a protein. It plays various roles in enzymatic catalysis, protein-protein interactions, and receptor binding.
-
Glutamic Acid (Glu, E): With a longer side chain, glutamic acid can reach further within a protein structure and participate in more complex interactions. It is also involved in diverse biological functions, including enzymatic activity, signaling pathways, and structural stabilization.
Basic Amino Acids: Lysine (Lys, K), Arginine (Arg, R), and Histidine (His, H)
The basic amino acids possess positively charged side chains at physiological pH. These positive charges contribute significantly to the electrostatic interactions within and between proteins.
-
Lysine (Lys, K): Its side chain terminates in a primary amino group (-NH₃⁺), which is strongly basic and carries a positive charge at physiological pH. Lysine is often involved in interactions with negatively charged molecules or residues.
-
Arginine (Arg, R): Possesses a guanidinium group in its side chain, which is even more basic than the amino group of lysine. This group carries a positive charge and is involved in numerous protein-protein interactions, enzyme catalysis, and DNA binding.
-
Histidine (His, H): This is a unique basic amino acid because its imidazole ring has a pKa close to physiological pH (around 6). This means that histidine can exist in both protonated (positively charged) and deprotonated (neutral) forms near pH 7.4. This unique property makes histidine a crucial residue in many enzymes, where it acts as an acid or base catalyst, depending on its ionization state. Its ability to switch between these states is crucial for mediating proton transfer reactions.
Polar Neutral Amino Acids with Ionizable Side Chains: Tyrosine (Tyr, Y) and Cysteine (Cys, C)
While generally classified as polar neutral, tyrosine and cysteine possess ionizable side chains that can affect their charge and interactions.
-
Tyrosine (Tyr, Y): Contains a phenolic hydroxyl group (-OH) in its side chain. While not strongly acidic or basic, the hydroxyl group can be deprotonated at high pH, yielding a negative charge. This deprotonation can influence protein-protein interactions and other intermolecular interactions.
-
Cysteine (Cys, C): Possesses a thiol group (-SH) in its side chain. The thiol group has a relatively low pKa, meaning it can easily lose a proton and become deprotonated. This deprotonated form (-S⁻) can form disulfide bonds with other cysteine residues, a crucial mechanism for stabilizing protein structures, particularly in extracellular proteins. The redox state of cysteine is also crucial in regulating enzymatic activity and other cellular processes.
The pKa and its Influence on Ionization
The pKa is a crucial parameter determining the ionization state of an ionizable side chain. The pKa represents the pH at which half of the molecules are protonated and half are deprotonated. The relationship between pH and ionization is described by the Henderson-Hasselbalch equation:
pH = pKa + log ([A⁻]/[HA])
where [A⁻] is the concentration of the deprotonated form and [HA] is the concentration of the protonated form.
The pKa of an ionizable side chain is influenced by its local environment within the protein. Factors such as proximity to other charged groups, hydrogen bonding, and the dielectric constant of the surrounding medium can significantly shift the pKa from its intrinsic value in solution. This microenvironment-dependent pKa shift is critical for fine-tuning the ionization state of side chains and their functional roles.
Methods for Studying Ionizable Side Chains
Several techniques are used to study the ionization state and properties of ionizable side chains in proteins:
-
X-ray crystallography: This technique can provide high-resolution structural information, including the positions of ionizable side chains and their interactions with other residues.
-
Nuclear magnetic resonance (NMR) spectroscopy: NMR can reveal information about the dynamics and ionization states of individual side chains in solution.
-
pH titration: Monitoring the change in protein charge as a function of pH can provide information about the pKa values of ionizable residues.
-
Computational methods: Molecular dynamics simulations and other computational techniques can predict the ionization states and pKa values of ionizable side chains, based on their three-dimensional structures and interactions with the surrounding environment.
Conclusion
Amino acids with ionizable side chains are essential for the structure, function, and diverse roles of proteins in biological systems. Their ionization states, governed by the pKa and influenced by the local environment, critically affect protein folding, stability, catalytic activity, protein-protein interactions, and many other biological processes. Understanding the properties and behavior of these ionizable side chains is crucial for deciphering the complexities of protein biology and developing new therapeutics and technologies. The diverse techniques used to study these residues provide valuable insights into their crucial roles within the intricate world of biomolecules. Future research in this area continues to unravel the intricacies of these amino acid side chains and their significant impact on protein function and biology.
Latest Posts
Latest Posts
-
Find The Equation Of The Tangent Plane To The Surface
Apr 05, 2025
-
Third Trophic Level In The Food Chain
Apr 05, 2025
-
How To Solve 3 Equations 3 Unknowns
Apr 05, 2025
-
How Many Times More Acidic Is Ph3 Than Ph5
Apr 05, 2025
-
What Instruments Are Used To Measure Humidity
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Amino Acids With Ionizable Side Chains . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
