Are Molecules The Same As Moles
Muz Play
Apr 05, 2025 · 5 min read
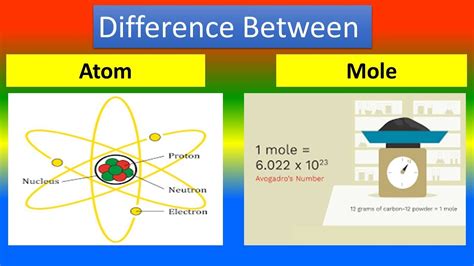
Table of Contents
Are Molecules the Same as Moles? Understanding the Fundamental Difference
The terms "molecule" and "mole" are frequently encountered in chemistry, and while they are closely related, they are not interchangeable. Understanding the distinction between these two concepts is crucial for grasping fundamental chemical principles. This article delves deep into the definitions, explains the relationship, and clarifies the significant differences between molecules and moles. We'll explore how they are used in calculations and highlight common misconceptions.
Understanding Molecules: The Building Blocks of Matter
A molecule is a group of two or more atoms held together by chemical bonds. These atoms can be of the same element (e.g., O₂ - oxygen molecule) or different elements (e.g., H₂O - water molecule, CO₂ - carbon dioxide molecule). The key characteristic of a molecule is its discrete structure; it's a distinct entity with a specific arrangement of atoms. The properties of a molecule depend on both the types of atoms it contains and the way those atoms are bonded together. This arrangement determines its shape, reactivity, and other physical and chemical properties.
Types of Molecules:
- Diatomic Molecules: Composed of two atoms of the same element (e.g., H₂, N₂, O₂).
- Triatomic Molecules: Composed of three atoms (e.g., H₂O, CO₂).
- Polyatomic Molecules: Composed of more than three atoms (e.g., C₆H₁₂O₆ – glucose).
- Macromolecules: Very large molecules, often polymers, composed of many repeating subunits (e.g., proteins, DNA).
The size and complexity of molecules vary enormously. Some are simple, like diatomic oxygen, while others are incredibly complex, like proteins with thousands of atoms. Regardless of their size, each molecule maintains its unique identity and characteristics.
Understanding Moles: Counting Immense Numbers of Molecules
A mole (mol) is not a type of particle; instead, it's a unit of measurement, much like a dozen (12) or a gross (144). However, unlike these everyday units, a mole represents an incredibly large number of particles: Avogadro's number (approximately 6.022 x 10²³). This number is chosen because it connects the atomic mass unit (amu) to grams. One mole of a substance contains Avogadro's number of elementary entities, which could be atoms, molecules, ions, or formula units.
The Significance of Avogadro's Number:
Avogadro's number provides a practical way to handle the vast quantities of atoms and molecules involved in chemical reactions. It allows chemists to work with manageable numbers in grams instead of dealing with astronomically large numbers of individual particles. For example, it's much simpler to say "one mole of water" (containing 6.022 x 10²³ water molecules) than to state the actual number of water molecules.
Moles and Molar Mass:
The molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It's numerically equal to the atomic or molecular weight of the substance. For example, the molar mass of water (H₂O) is approximately 18 g/mol (16 g/mol for oxygen + 2 x 1 g/mol for hydrogen). This means that one mole of water weighs 18 grams.
The Relationship Between Molecules and Moles: A Crucial Connection
The relationship between molecules and moles is fundamental to stoichiometry, the study of quantitative relationships in chemical reactions. A mole allows us to convert between the macroscopic world (grams) and the microscopic world (number of molecules).
In essence: A mole is a counting unit for molecules (or other particles). One mole of any substance always contains the same number of elementary entities: Avogadro's number.
Example:
Let's say we have 18 grams of water (H₂O). Since the molar mass of water is 18 g/mol, this means we have one mole of water. This one mole of water contains Avogadro's number (6.022 x 10²³) of water molecules.
Common Misconceptions and Clarifications
Several misconceptions often arise when comparing molecules and moles:
Misconception 1: A mole is a type of molecule.
Clarification: A mole is a unit of measurement, not a type of particle. It's a way to count a very large number of particles, including molecules.
Misconception 2: One mole of a substance always weighs the same.
Clarification: The mass of one mole of a substance varies depending on the substance's molar mass. One mole of water weighs 18 grams, while one mole of carbon dioxide weighs 44 grams.
Misconception 3: Moles are only used for molecules.
Clarification: Moles can be used to represent the amount of any chemical entity, including atoms, ions, and formula units, not just molecules.
Moles in Chemical Calculations: Practical Applications
Moles are essential for performing many calculations in chemistry. Here are some key applications:
-
Determining the number of molecules: Knowing the number of moles of a substance allows you to calculate the number of molecules present using Avogadro's number.
-
Stoichiometric calculations: Moles are crucial in balancing and interpreting chemical equations. They allow us to determine the amount of reactants needed and the amount of products formed in a chemical reaction.
-
Concentration calculations: Molarity, a common measure of concentration, is defined as moles of solute per liter of solution.
-
Gas law calculations: The ideal gas law uses moles to relate the pressure, volume, temperature, and amount of a gas.
Conclusion: Distinct Yet Interdependent Concepts
While molecules and moles are different concepts, they are intrinsically linked. Molecules are the fundamental building blocks of matter, while moles provide a practical way to count and quantify these microscopic particles. Understanding this crucial distinction and the relationship between them is vital for mastering fundamental chemistry concepts and performing essential chemical calculations. The ability to seamlessly transition between the macroscopic world of grams and the microscopic world of molecules is a cornerstone of chemical understanding. By grasping these fundamental concepts, you’ll unlock a deeper appreciation of the quantitative nature of chemistry and its power to explain the world around us.
Latest Posts
Latest Posts
-
Which Type Of Plant Has No Vascular Tissue
Apr 06, 2025
-
An Organism That Cannot Tolerate An Oxygen Environment Is A
Apr 06, 2025
-
What Are The Unifying Themes Of Biology
Apr 06, 2025
-
State The Law Of Constant Composition
Apr 06, 2025
-
Which Electrons Participate In Chemical Bonding
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Are Molecules The Same As Moles . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
