At Room Temperature The Various Conformations Of Butane
Muz Play
Apr 03, 2025 · 6 min read
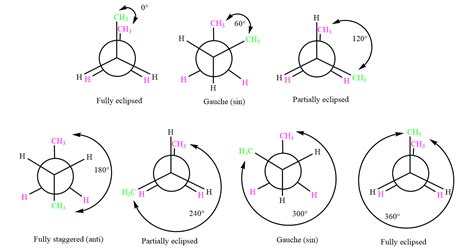
Table of Contents
At Room Temperature: Exploring the Conformational Landscape of Butane
Butane, a simple alkane with the formula C₄H₁₀, serves as an excellent model for understanding conformational isomerism – the phenomenon where molecules with the same connectivity exhibit different three-dimensional arrangements due to rotations around single bonds. While seemingly straightforward, the conformational analysis of butane at room temperature reveals a rich interplay of steric interactions, energy differences, and dynamic equilibrium, providing a fascinating case study in organic chemistry. This article delves deep into the various conformations of butane at room temperature, exploring their relative energies, stability, and the factors influencing their populations.
Understanding Conformations and Energy Barriers
Before diving into the specifics of butane, let's establish a foundational understanding of conformational isomerism. Conformational isomers, or conformers, are different spatial arrangements of a molecule that can be interconverted by rotation around single bonds. Unlike stereoisomers, conformers cannot be isolated because the energy barriers separating them are relatively low, allowing for rapid interconversion at room temperature. This interconversion occurs through bond rotation.
The energy associated with different conformations arises primarily from steric interactions – the repulsive forces between atoms or groups that are brought too close together during rotation. These interactions influence the stability of each conformer. Higher energy conformations are less stable and less populated in an equilibrium mixture, while lower energy conformations are more stable and more populated. The energy difference between conformers is often represented as an energy barrier, which must be overcome for interconversion to occur. This barrier is typically expressed in kilocalories per mole (kcal/mol) or kilojoules per mole (kJ/mol).
The Conformers of Butane: A Detailed Analysis
Butane's carbon chain allows for rotation around the central C-C single bond. This rotation leads to various conformations, but the most significant are the staggered and eclipsed conformations.
1. Staggered Conformations
Staggered conformations are characterized by the methyl groups (CH₃) being as far apart as possible. This minimizes steric interactions and leads to lower energy. Among the staggered conformations, two are particularly important:
-
Anti Conformation: In the anti conformation, the two methyl groups are positioned 180° apart. This arrangement represents the lowest energy conformation of butane, due to the maximum distance between the bulky methyl groups, minimizing steric repulsion. It is the most stable conformer.
-
Gauche Conformations: Two gauche conformations exist where the methyl groups are 60° apart. While still staggered, the proximity of the methyl groups results in some steric hindrance, making them slightly higher in energy than the anti conformation. However, they are significantly more stable than the eclipsed conformations. There are two gauche conformations due to the symmetry, one with the methyl group on the left and one with the methyl group on the right.
2. Eclipsed Conformations
Eclipsed conformations occur when the methyl groups are positioned directly opposite each other, resulting in significant steric repulsion. There are three eclipsed conformations, with varying degrees of steric interaction:
-
Totally Eclipsed Conformation: This is the highest energy conformation. It features the methyl groups directly overlapping, leading to maximum steric clash. This is the least stable conformation.
-
Partially Eclipsed Conformations: Two partially eclipsed conformations are present, where one methyl group eclipses a hydrogen. These conformations are higher in energy than the staggered conformations but lower than the totally eclipsed conformation.
Energy Differences and Population Distribution at Room Temperature
The energy differences between the various butane conformations are crucial in determining their relative populations at room temperature. The anti conformation is the most stable, with the gauche conformations being somewhat less stable, and the eclipsed conformations significantly less stable. This energy difference means that the anti conformer is the most populated at room temperature; however, the energy barrier is low enough to allow interconversion between all conformers. The gauche conformations, despite being higher in energy than the anti conformer, are still significantly populated due to the relatively low energy barrier between the conformations.
The population distribution at room temperature is not a static picture. Instead, it represents a dynamic equilibrium where molecules are constantly interconverting between different conformations. The equilibrium strongly favors the anti conformer, but a substantial fraction of molecules exist in the gauche conformations, with a negligible fraction in the eclipsed conformations.
Factors Influencing Conformational Equilibrium
Several factors influence the conformational equilibrium of butane and other molecules:
-
Steric effects: The most dominant factor. Bulky substituents tend to adopt staggered conformations to minimize steric repulsion.
-
Temperature: Increased temperature provides more energy for interconversion between conformers, but the overall population distribution will still strongly favour the lower energy conformers.
-
Solvent effects: The solvent can influence the conformational equilibrium, particularly through interactions with the molecule. Polar solvents, for instance, might favour conformations with higher dipole moments.
Experimental Techniques for Studying Butane Conformations
Several experimental techniques can be used to study the conformations of butane and determine their relative populations:
-
Infrared (IR) spectroscopy: Different conformers exhibit distinct vibrational frequencies in IR spectroscopy, allowing for the identification and relative quantification of different conformers.
-
Nuclear Magnetic Resonance (NMR) spectroscopy: NMR spectroscopy, especially ¹H NMR and ¹³C NMR, can provide valuable insights into the dynamic equilibrium of butane conformers. The coupling constants and chemical shifts can be used to distinguish between conformers.
Butane Conformations and Their Significance
The understanding of butane's conformational landscape extends far beyond a simple academic exercise. The principles elucidated through the study of butane's conformers are applicable to more complex molecules and are crucial in several areas:
-
Drug design and discovery: Understanding conformational preferences is critical in designing drugs that can effectively bind to their target molecules. The ability of a drug to adopt a specific conformation is crucial for its activity.
-
Polymer science: Polymer properties are heavily influenced by the conformations of their constituent monomer units. Understanding conformational preferences aids in predicting polymer properties and designing polymers with specific characteristics.
-
Catalysis: In many catalytic reactions, the conformation of the reactant molecules plays a vital role in determining the reaction pathway and efficiency.
-
Computational Chemistry: Butane serves as a benchmark molecule for testing and validating computational methods used to predict and analyze molecular conformations.
Conclusion
The seemingly simple molecule of butane presents a rich and fascinating study in conformational isomerism. At room temperature, the dynamic equilibrium between different conformations – primarily the anti and gauche conformations – is dictated by a delicate balance of steric interactions and energy differences. Understanding these interactions is not merely an academic exercise but holds significant implications across various scientific disciplines, highlighting the importance of appreciating the three-dimensional structure of molecules in explaining their properties and behavior. The principles learned from studying butane's conformations form the foundation for understanding the conformational behavior of more complex and biologically relevant molecules.
Latest Posts
Latest Posts
-
Dense Regular Connective Tissue Under Microscope
Apr 04, 2025
-
How Did The Renaissance Affect The Power Of Independent Monarchs
Apr 04, 2025
-
Are Ion Channels Active Or Passive
Apr 04, 2025
-
Prokaryotes That Obtain Their Energy From Chemical Compounds Are Called
Apr 04, 2025
-
What Does High Absorbance Mean In Spectrophotometry
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about At Room Temperature The Various Conformations Of Butane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
