Calculate The Ph At The Equivalence Point For The Titration
Muz Play
Apr 02, 2025 · 5 min read
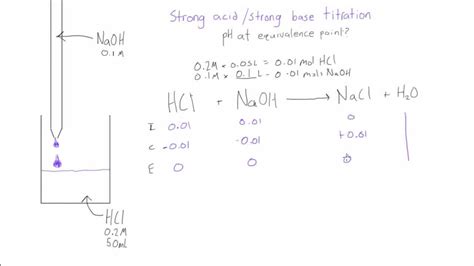
Table of Contents
- Calculate The Ph At The Equivalence Point For The Titration
- Table of Contents
- Calculating the pH at the Equivalence Point for a Titration
- Understanding the Equivalence Point
- Strong Acid-Strong Base Titration
- Weak Acid-Strong Base Titration
- Weak Base-Strong Acid Titration
- Polyprotic Acid Titration
- Importance of Equivalence Point pH Calculation
- Conclusion
- Latest Posts
- Latest Posts
- Related Post
Calculating the pH at the Equivalence Point for a Titration
Determining the pH at the equivalence point of a titration is crucial for understanding the titration curve and selecting appropriate indicators. This point represents the complete neutralization of the analyte, and the pH depends entirely on the nature of the resulting solution. This article will delve into the methods and calculations needed to determine the pH at the equivalence point for various titrations, focusing on strong acid-strong base, weak acid-strong base, weak base-strong acid, and polyprotic acid titrations. We will also discuss the importance of this calculation in analytical chemistry.
Understanding the Equivalence Point
The equivalence point in a titration is the point at which the moles of titrant added are stoichiometrically equal to the moles of analyte present. This doesn't necessarily mean the pH is 7; it depends on the strength of the acid and base involved. Before the equivalence point, the pH changes gradually. However, at the equivalence point, a sharp change in pH occurs, often signifying the end of the titration. This sharp change makes it possible to visually detect the equivalence point using appropriate indicators, which change color within a specific pH range.
Strong Acid-Strong Base Titration
This is the simplest type of titration. The reaction involves a strong acid (e.g., HCl) and a strong base (e.g., NaOH), resulting in the formation of water and a neutral salt. At the equivalence point, the solution contains only the salt and water. Since strong acids and strong bases completely dissociate, the pH at the equivalence point is 7.
Example: Consider the titration of 25.00 mL of 0.100 M HCl with 0.100 M NaOH. At the equivalence point, the moles of HCl will equal the moles of NaOH:
Moles of HCl = (0.100 mol/L) * (0.02500 L) = 0.00250 mol
This requires 0.00250 mol of NaOH, which corresponds to:
Volume of NaOH = (0.00250 mol) / (0.100 mol/L) = 0.02500 L = 25.00 mL
At the equivalence point, the resulting solution contains only NaCl and water. NaCl is a neutral salt, so the pH is 7.
Weak Acid-Strong Base Titration
Titrating a weak acid with a strong base leads to a more complex scenario. At the equivalence point, the solution contains the conjugate base of the weak acid. This conjugate base can undergo hydrolysis, reacting with water to produce hydroxide ions (OH⁻), increasing the pH above 7.
To calculate the pH, we need to consider the hydrolysis reaction of the conjugate base. Let's denote the weak acid as HA and its conjugate base as A⁻. The hydrolysis reaction is:
A⁻ + H₂O ⇌ HA + OH⁻
The pH can be calculated using the following steps:
-
Determine the concentration of the conjugate base: This depends on the initial concentration of the weak acid and the volume of the strong base added at the equivalence point.
-
Calculate the Kb: The base dissociation constant (Kb) for the conjugate base can be determined using the relationship Kb = Kw/Ka, where Kw is the ion product of water (1.0 x 10⁻¹⁴ at 25°C) and Ka is the acid dissociation constant of the weak acid.
-
Use the Kb expression: Set up an ICE (Initial, Change, Equilibrium) table to determine the equilibrium concentrations of A⁻, HA, and OH⁻.
-
Calculate the pOH: Solve the Kb expression for [OH⁻] and then calculate the pOH using the formula pOH = -log[OH⁻].
-
Calculate the pH: Finally, calculate the pH using the relationship pH + pOH = 14.
Weak Base-Strong Acid Titration
This titration is analogous to the weak acid-strong base case. At the equivalence point, the solution contains the conjugate acid of the weak base, which can undergo hydrolysis to produce hydronium ions (H₃O⁺), resulting in a pH below 7.
The calculation steps are similar to those for a weak acid-strong base titration, but we'll use the Ka of the conjugate acid instead of the Kb of the conjugate base. The Ka is related to Kb by the equation Ka = Kw/Kb.
Polyprotic Acid Titration
Polyprotic acids have multiple ionizable protons. Their titration curves exhibit multiple equivalence points, one for each proton. Calculating the pH at each equivalence point requires a stepwise approach, considering the dissociation of each proton.
For example, consider the titration of a diprotic acid, H₂A. The first equivalence point represents the complete neutralization of the first proton, resulting in the formation of HA⁻. The pH at this point depends on the Ka1 of the acid and the hydrolysis of HA⁻. The second equivalence point represents the complete neutralization of both protons, resulting in A²⁻. The pH at this point depends on the Ka2 of the acid and the hydrolysis of A²⁻. This requires solving multiple equilibrium expressions simultaneously.
Importance of Equivalence Point pH Calculation
The precise calculation of the pH at the equivalence point is essential for several reasons:
-
Indicator Selection: The equivalence point pH determines the appropriate indicator to use in a titration. The indicator should change color within the pH range encompassing the equivalence point.
-
Accuracy of Titration: A properly selected indicator ensures accurate determination of the analyte concentration. An indicator that changes color far from the equivalence point will result in a significant error.
-
Understanding Titration Curves: The equivalence point pH is a critical point on the titration curve, helping to understand the behavior of the acid-base system being titrated.
-
Applications in Analytical Chemistry: Acid-base titrations are widely used in analytical chemistry for determining the concentration of various substances. Accurate equivalence point pH calculations are vital for reliable results.
Conclusion
Calculating the pH at the equivalence point requires understanding the nature of the acid and base involved. While strong acid-strong base titrations yield a neutral pH, weak acid-strong base and weak base-strong acid titrations require consideration of hydrolysis. Polyprotic acid titrations demand a stepwise approach. Mastering these calculations is crucial for conducting successful titrations and interpreting the results accurately in various analytical chemistry applications. This understanding allows for proper indicator selection and the precise determination of analyte concentrations. The accurate determination of the equivalence point pH is a cornerstone of quantitative chemical analysis.
Latest Posts
Latest Posts
-
Periodic Table With Metals Nonmetals And Metalloids Color Coded
Apr 04, 2025
-
Use The Graph To Determine The Following
Apr 04, 2025
-
Is Meiosis Asexual Or Sexual Reproduction
Apr 04, 2025
-
Why Is Organization Important To A Speech
Apr 04, 2025
-
How To Find The Mass Of An Isotope
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Calculate The Ph At The Equivalence Point For The Titration . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
