Choose The Component Parts Of An Amino Acid.
Muz Play
Apr 02, 2025 · 6 min read
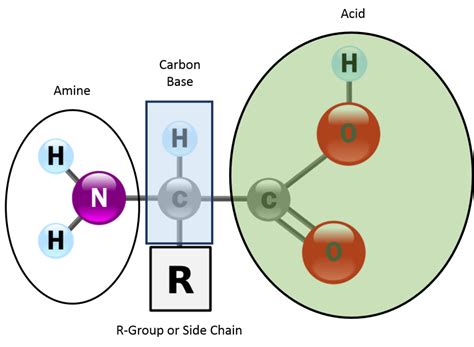
Table of Contents
Choose the Component Parts of an Amino Acid: A Deep Dive into the Building Blocks of Life
Amino acids. The very name conjures images of complex biological processes, but at their core, these molecules are surprisingly simple. Understanding their component parts is key to grasping their vital role in everything from protein synthesis to cellular signaling. This comprehensive guide delves into the fundamental structure of amino acids, exploring each component and its significance. We'll also touch upon the different types of amino acids and their diverse functions within the human body.
The Fundamental Trio: Carbon, Amino, and Carboxyl
At the heart of every amino acid lies a central carbon atom, often referred to as the alpha carbon. This carbon atom acts as the anchor, connecting three crucial functional groups:
1. The Amino Group (-NH2)
This is where the "amino" in "amino acid" comes from. The amino group is a nitrogen atom bonded to two hydrogen atoms. It's a basic group, meaning it can accept a proton (H+), thereby increasing the pH of a solution. This basicity is crucial for the amino acid's behavior within the cellular environment. The amino group's ability to participate in hydrogen bonding is also significant for the formation of complex protein structures.
2. The Carboxyl Group (-COOH)
The carboxyl group, consisting of a carbon atom double-bonded to an oxygen atom and single-bonded to a hydroxyl group (-OH), is an acidic group. It can donate a proton (H+), thus lowering the pH of a solution. This acidic nature plays a critical role in many biochemical reactions. The carboxyl group, like the amino group, readily participates in hydrogen bonding, contributing to the intricate three-dimensional folding of proteins.
3. The Side Chain (R-group)
This is the variable component that distinguishes one amino acid from another. The R-group, or side chain, can range from a simple hydrogen atom (as in glycine) to complex aromatic rings (as in tryptophan). The nature of the R-group dictates the amino acid's chemical properties, including its size, shape, charge, and polarity. These properties, in turn, determine the amino acid's role in protein structure and function. We'll explore the diverse R-groups in more detail later.
The Peptide Bond: Linking Amino Acids Together
Amino acids don't exist in isolation; they link together to form long chains called polypeptides. This linkage occurs through a peptide bond, a special type of covalent bond formed between the carboxyl group of one amino acid and the amino group of another. This reaction releases a water molecule (H₂O), a process known as dehydration synthesis or condensation.
The peptide bond creates a repeating backbone of –N-C-C– units, with the R-groups extending outward. The sequence of amino acids in a polypeptide chain determines its primary structure, which in turn influences the higher-order structures (secondary, tertiary, and quaternary) that dictate the protein's overall function.
Categorizing Amino Acids Based on their R-Groups
The R-group is the key to understanding the diversity and functionality of amino acids. Based on the properties of their R-groups, amino acids are categorized into several groups:
1. Nonpolar, Aliphatic Amino Acids
These amino acids have hydrophobic (water-repelling) R-groups. They are typically found buried within the protein's core, away from the aqueous cellular environment. Examples include:
- Glycine (Gly, G): The simplest amino acid, with a hydrogen atom as its R-group.
- Alanine (Ala, A): A methyl group (-CH₃) constitutes its R-group.
- Valine (Val, V), Leucine (Leu, L), Isoleucine (Ile, I): These have branched aliphatic side chains.
2. Aromatic Amino Acids
These amino acids have R-groups containing aromatic rings, which are relatively hydrophobic. They often participate in interactions involving π-electrons. Examples include:
- Phenylalanine (Phe, F): Contains a benzene ring.
- Tyrosine (Tyr, Y): Contains a phenol group (benzene ring with a hydroxyl group).
- Tryptophan (Trp, W): Contains an indole ring.
3. Polar, Uncharged Amino Acids
These amino acids have hydrophilic (water-attracting) R-groups that often participate in hydrogen bonding. They are often found on the protein's surface, interacting with the surrounding water molecules. Examples include:
- Serine (Ser, S), Threonine (Thr, T): Contain hydroxyl groups (-OH).
- Cysteine (Cys, C): Contains a thiol group (-SH), capable of forming disulfide bonds.
- Asparagine (Asn, N), Glutamine (Gln, Q): Contain amide groups (-CONH₂).
4. Positively Charged (Basic) Amino Acids
These amino acids have R-groups that carry a positive charge at physiological pH. They often participate in ionic interactions within proteins and with other molecules. Examples include:
- Lysine (Lys, K): Contains an amino group (-NH₃⁺) at the end of its side chain.
- Arginine (Arg, R): Contains a guanidinium group.
- Histidine (His, H): Contains an imidazole ring, which can be positively or neutrally charged depending on the pH.
5. Negatively Charged (Acidic) Amino Acids
These amino acids have R-groups that carry a negative charge at physiological pH. They also participate in ionic interactions. Examples include:
- Aspartic acid (Asp, D): Contains a carboxyl group (-COO⁻).
- Glutamic acid (Glu, E): Contains a carboxyl group (-COO⁻) further away from the alpha-carbon.
Essential vs. Nonessential Amino Acids
The human body can synthesize some amino acids, but others must be obtained from the diet. This distinction leads to the classification of amino acids as either:
-
Essential amino acids: These cannot be synthesized by the human body and must be obtained through dietary intake. They are: histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine.
-
Nonessential amino acids: These can be synthesized by the human body and therefore don't need to be consumed directly. Examples include alanine, asparagine, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, and tyrosine.
The Importance of Amino Acids in the Body
Amino acids are the fundamental building blocks of proteins, which perform a myriad of functions within the body:
-
Structural components: Collagen, keratin, and elastin are examples of proteins providing structural support to tissues and organs.
-
Enzymes: Enzymes are proteins that catalyze biochemical reactions, essential for metabolism.
-
Hormones: Many hormones, such as insulin and glucagon, are peptide hormones, playing crucial roles in regulating various bodily processes.
-
Antibodies: Antibodies are proteins that form part of the immune system, defending against pathogens.
-
Neurotransmitters: Some neurotransmitters, such as glutamate and GABA, are amino acid derivatives, playing vital roles in nerve impulse transmission.
-
Transport proteins: Hemoglobin, a protein responsible for oxygen transport in the blood, is a prime example.
Beyond the Basics: Understanding Amino Acid Modifications
Amino acids can undergo various post-translational modifications, altering their properties and functions. These modifications include:
-
Phosphorylation: The addition of a phosphate group, often regulating protein activity.
-
Glycosylation: The addition of sugar molecules, impacting protein folding and cell recognition.
-
Acetylation: The addition of an acetyl group, often affecting protein stability and interactions.
-
Methylation: The addition of a methyl group, frequently modifying gene expression and protein function.
Understanding the component parts of amino acids, their classification, and their various modifications provides a foundational understanding of the complexity and vital importance of these molecules in all aspects of life. Their intricate interplay dictates the structure and function of proteins, the workhorses of the human body, underscoring their crucial role in maintaining health and wellbeing.
Latest Posts
Latest Posts
-
What Is The Difference Between Dehydration And Hydrolysis
Apr 03, 2025
-
What Is The Most Complex Level Of Organization
Apr 03, 2025
-
What Determines The Volume Of Gas
Apr 03, 2025
-
Non Mendelian Genetics Practice Packet Answers
Apr 03, 2025
-
Similarities Between Endocrine And Nervous System
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Choose The Component Parts Of An Amino Acid. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
