Cis 1 3 Dimethylcyclohexane Chair Conformation
Muz Play
Apr 03, 2025 · 5 min read
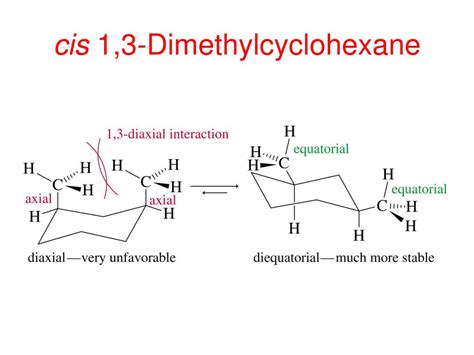
Table of Contents
CIS 1,3-Dimethylcyclohexane Chair Conformation: A Deep Dive
The world of organic chemistry often presents fascinating challenges, and the conformational analysis of cyclohexane derivatives is a prime example. Understanding the intricacies of these conformations is crucial for predicting reactivity and properties of molecules. This article delves into the specifics of the cis 1,3-dimethylcyclohexane chair conformation, exploring its stability, energy differences, and the factors influencing its behavior. We'll examine the concepts of axial and equatorial positions, 1,3-diaxial interactions, and how these affect the overall energy of the molecule.
Understanding Cyclohexane Conformations
Before we dive into the complexities of cis 1,3-dimethylcyclohexane, let's establish a foundational understanding of cyclohexane conformations. Cyclohexane, a six-membered ring, exists primarily in two chair conformations that interconvert rapidly at room temperature through a process called ring flipping. These chair conformations are not equivalent in energy; one is slightly more stable than the other.
Chair Conformations: Axial and Equatorial Positions
Each carbon atom in a cyclohexane chair conformation possesses two substituents: one axial and one equatorial. Axial substituents are oriented perpendicular to the plane of the ring, while equatorial substituents are approximately parallel to the plane of the ring. This distinction is pivotal when considering the stability of substituted cyclohexanes.
The Importance of 1,3-Diaxial Interactions
The relative stability of substituted cyclohexanes is significantly impacted by 1,3-diaxial interactions. These interactions occur between an axial substituent and the axial hydrogens on carbons three positions away. These interactions are steric in nature, meaning they arise from the close proximity and repulsion of electron clouds. Larger substituents experience stronger 1,3-diaxial interactions, leading to higher energy conformations.
Cis 1,3-Dimethylcyclohexane: A Case Study
Now, let's focus on cis 1,3-dimethylcyclohexane. The "cis" prefix indicates that both methyl groups are on the same side of the cyclohexane ring. This seemingly small difference has profound implications for the molecule's conformational preferences.
Chair Conformations of cis 1,3-Dimethylcyclohexane
Unlike monosubstituted cyclohexanes, cis 1,3-dimethylcyclohexane presents a more nuanced conformational landscape. When considering its chair conformations, we observe the following:
- Conformation A: In this conformation, one methyl group is axial and the other is equatorial.
- Conformation B: In this conformation, the positions of the methyl groups are reversed, with one being equatorial and the other axial.
Crucially, both conformations are diastereomers. They cannot be interconverted without breaking and reforming bonds.
Analyzing 1,3-Diaxial Interactions in cis 1,3-Dimethylcyclohexane
The key to understanding the relative stability of conformations A and B lies in analyzing their 1,3-diaxial interactions. In Conformation A, one methyl group is axial, leading to significant 1,3-diaxial interactions with two axial hydrogens on carbons three positions away. These steric interactions raise the energy of conformation A. In Conformation B, the same situation applies, so, both conformations exhibit similar energy costs from 1,3-diaxial interactions.
However, the energy difference between the two conformations is not negligible. While the 1,3-diaxial interactions are present in both, the degree to which they are present and how it affects the overall energy state is what needs to be evaluated.
The Energy Difference: A Quantitative Approach
The exact energy difference between Conformation A and Conformation B is difficult to determine without advanced computational methods. However, we can qualitatively assess that Conformation A, in spite of having one methyl group equatorial and one axial, is slightly higher in energy than Conformation B. This is because the overall energy penalty of 1,3-diaxial interactions is slightly larger in Conformation A due to subtle geometrical differences and overall bulkier interactions.
It's important to emphasize that the energy difference between these two chair conformations is relatively small. At room temperature, both conformations exist in equilibrium, although the lower energy form (Conformation B) is slightly more populated.
Factors Influencing Conformation Stability
Several factors influence the stability of cis 1,3-dimethylcyclohexane's chair conformations beyond the previously discussed 1,3-diaxial interactions.
Steric Hindrance: The Size of Substituents
The size of the substituents plays a crucial role. Larger substituents lead to greater steric hindrance and increase the energy difference between axial and equatorial positions. If, instead of methyl groups, we had larger substituents like tert-butyl groups, the energy difference would be much more significant, favoring the conformation with both large groups in equatorial positions.
Gauche Interactions
Gauche interactions occur when two substituents are on adjacent carbons and are oriented at a dihedral angle of approximately 60°. These interactions are also steric in nature and contribute to the overall energy of the molecule. Gauche interactions contribute to the energy difference between Conformation A and B although the impact is relatively minimal.
Entropy: The Role of Flexibility
At higher temperatures, the entropic contribution to the overall free energy of the system becomes more significant. While Conformation B is lower in enthalpy (energy) it is slightly less flexible, therefore at higher temperatures the entropic contribution (from Conformation A's higher flexibility) can decrease the energy difference and lead to a higher proportion of Conformation A.
Applications and Relevance
Understanding the conformational preferences of cis 1,3-dimethylcyclohexane has various applications in chemistry:
-
Predicting Reactivity: Knowledge of the dominant conformation influences predictions about reaction rates and selectivities. Reactions involving the axial methyl groups, for instance, may occur more slowly.
-
Spectroscopy: Conformational analysis is crucial for interpreting NMR (Nuclear Magnetic Resonance) and IR (Infrared) spectra. Certain peaks and coupling constants are sensitive to the orientation of substituents.
-
Drug Design: Many drugs contain cyclohexane rings, and understanding their conformations is essential for designing drugs with desired properties and interactions with biological targets. Cis 1,3-dimethylcyclohexane itself might serve as a simplified model for more complex biologically active molecules.
Conclusion: A Deeper Understanding of Conformational Analysis
The cis 1,3-dimethylcyclohexane chair conformation offers a fascinating case study in conformational analysis. Although both chair conformations exhibit substantial 1,3-diaxial interactions, subtle differences in these interactions lead to a slight energy preference for the conformation with one axial and one equatorial methyl group. This detailed analysis highlights the interconnectedness of steric effects, conformational flexibility, and the equilibrium between different conformations. Mastering these concepts is vital for anyone working in organic chemistry, particularly in areas dealing with synthesis, reactivity, and the design of complex molecules. The principles learned here extend far beyond this specific example, forming the foundation for a deeper understanding of the behavior of countless organic compounds. Understanding these nuances is crucial for predicting the properties and reactivity of molecules, a skill that's increasingly important in various fields of chemistry and beyond.
Latest Posts
Related Post
Thank you for visiting our website which covers about Cis 1 3 Dimethylcyclohexane Chair Conformation . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
