Difference Between Molar Mass And Atomic Mass
Muz Play
Apr 02, 2025 · 5 min read
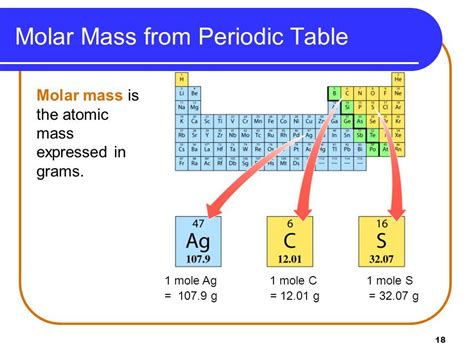
Table of Contents
Delving Deep: Understanding the Difference Between Molar Mass and Atomic Mass
The terms "atomic mass" and "molar mass" are frequently used in chemistry, often interchangeably, leading to confusion. While related, they represent distinct concepts crucial for understanding chemical reactions and stoichiometry. This comprehensive guide aims to clarify the differences, highlighting their applications and significance. We'll explore each concept in detail, providing clear examples to solidify your understanding.
Atomic Mass: The Mass of an Atom
Atomic mass, also known as atomic weight, refers to the average mass of an atom of an element, taking into account the different isotopes of that element. It's expressed in atomic mass units (amu), where 1 amu is defined as 1/12 the mass of a carbon-12 atom. This is crucial because most elements exist as a mixture of isotopes.
Isotopes and Their Role in Atomic Mass
Isotopes are atoms of the same element with the same number of protons (atomic number) but different numbers of neutrons. This difference in neutron number leads to variations in their mass. For instance, carbon has two stable isotopes: carbon-12 (¹²C) and carbon-13 (¹³C), with trace amounts of the radioactive isotope carbon-14 (¹⁴C).
The atomic mass listed on the periodic table is a weighted average of the masses of all naturally occurring isotopes of that element. The weighting factor is the relative abundance of each isotope. For example, carbon's atomic mass is approximately 12.01 amu, not exactly 12 amu, because it accounts for the presence of ¹³C and ¹⁴C, albeit in small quantities.
Calculating Atomic Mass: A Step-by-Step Example
Let's illustrate the calculation using chlorine, which has two major isotopes: ³⁵Cl (75.77% abundance) and ³⁷Cl (24.23% abundance).
-
Convert percentages to decimal fractions:
- ³⁵Cl: 75.77% = 0.7577
- ³⁷Cl: 24.23% = 0.2423
-
Multiply each isotope's mass (in amu) by its abundance:
- ³⁵Cl: 34.97 amu * 0.7577 = 26.49 amu
- ³⁷Cl: 36.97 amu * 0.2423 = 8.95 amu
-
Add the weighted masses:
- 26.49 amu + 8.95 amu = 35.44 amu
Therefore, the atomic mass of chlorine is approximately 35.44 amu. This is the value you'll find on the periodic table.
Molar Mass: Mass of One Mole of a Substance
Molar mass is the mass of one mole of a substance. A mole (mol) is a fundamental unit in chemistry, representing Avogadro's number (approximately 6.022 x 10²³) of particles (atoms, molecules, ions, etc.). The molar mass is expressed in grams per mole (g/mol).
Crucially, the numerical value of the molar mass of an element is the same as its atomic mass, but the units are different. For example, the atomic mass of carbon is approximately 12.01 amu, while its molar mass is 12.01 g/mol. This equivalence arises because Avogadro's number establishes a direct link between the atomic mass unit and the gram.
Molar Mass of Compounds
For compounds, the molar mass is the sum of the molar masses of all the atoms in the chemical formula. Let's take water (H₂O) as an example.
-
Determine the molar mass of each element:
- Hydrogen (H): 1.01 g/mol
- Oxygen (O): 16.00 g/mol
-
Multiply each element's molar mass by the number of atoms in the formula:
- Hydrogen: 1.01 g/mol * 2 = 2.02 g/mol
- Oxygen: 16.00 g/mol * 1 = 16.00 g/mol
-
Add the molar masses of all atoms:
- 2.02 g/mol + 16.00 g/mol = 18.02 g/mol
Thus, the molar mass of water is 18.02 g/mol. This means that one mole of water (6.022 x 10²³ water molecules) has a mass of 18.02 grams.
Applications of Molar Mass
Molar mass is a cornerstone of stoichiometric calculations. It allows chemists to convert between mass and the number of moles, enabling precise predictions and measurements in chemical reactions. For example:
-
Determining the number of moles from mass: If you have 10 grams of water, you can calculate the number of moles using the molar mass: 10 g / 18.02 g/mol ≈ 0.555 moles.
-
Determining the mass from the number of moles: If you need 2 moles of sodium chloride (NaCl), you can calculate the required mass using its molar mass (58.44 g/mol): 2 mol * 58.44 g/mol = 116.88 g.
-
Stoichiometric calculations: Molar mass is essential for balancing chemical equations and determining the amount of reactants or products involved in a reaction.
Key Differences Summarized
| Feature | Atomic Mass | Molar Mass |
|---|---|---|
| Definition | Average mass of an atom of an element | Mass of one mole of a substance |
| Unit | Atomic mass units (amu) | Grams per mole (g/mol) |
| Scope | Applies to individual atoms and isotopes | Applies to a macroscopic amount of substance |
| Numerical Value | Same as molar mass for elements | Same as atomic mass for elements; different for compounds |
| Calculation | Weighted average of isotopic masses | Sum of molar masses of constituent atoms |
Practical Applications and Significance
Understanding the distinction between atomic mass and molar mass is crucial for various applications in chemistry and related fields:
-
Analytical Chemistry: Determining the composition of substances through quantitative analysis relies heavily on molar mass calculations.
-
Biochemistry: Molar mass is essential for understanding the properties and interactions of biomolecules like proteins and DNA.
-
Pharmaceutical Chemistry: Accurate calculations of molar mass are critical in drug formulation and dosage determination.
-
Materials Science: Molar mass plays a significant role in characterizing and understanding the properties of different materials.
-
Environmental Science: Studying pollutant concentrations and their impact often involves molar mass calculations.
Conclusion: Bridging the Gap Between the Micro and Macro Worlds
Atomic mass and molar mass are fundamentally intertwined concepts. Atomic mass focuses on the individual atom and its isotopes, reflecting the micro-world of atoms and subatomic particles. Molar mass bridges the gap between this micro-world and the macroscopic world, enabling us to quantify and manipulate substances in chemical reactions and other applications. Mastering the difference and applications of these two essential concepts is paramount to success in the study and practice of chemistry. By understanding the weighted average nature of atomic mass and the mole-based definition of molar mass, you gain a more profound understanding of the fundamental building blocks of matter and their interactions. This knowledge forms the basis for numerous important calculations and applications across various scientific disciplines.
Latest Posts
Latest Posts
-
Is The Freezing Of Water A Chemical Change
Apr 03, 2025
-
What Are The Functions Of Stems
Apr 03, 2025
-
Heat Of Vaporization Of Water J Kg
Apr 03, 2025
-
What Happens To Plant Cells In A Hypertonic Solution
Apr 03, 2025
-
What Is A Disadvantage Of A Corporation
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Difference Between Molar Mass And Atomic Mass . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
