Differences Between Voltaic And Electrolytic Cells
Muz Play
Apr 06, 2025 · 6 min read
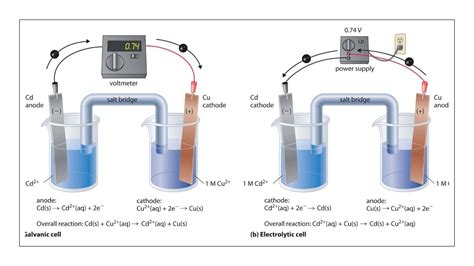
Table of Contents
Voltaic vs. Electrolytic Cells: A Deep Dive into Electrochemical Differences
Electrochemistry, the study of the interconversion of chemical and electrical energy, hinges on two fundamental cell types: voltaic (or galvanic) cells and electrolytic cells. While both involve electrochemical reactions, they differ significantly in their function, energy flow, and applications. Understanding these differences is crucial for anyone studying chemistry, engineering, or related fields. This comprehensive guide will explore the key distinctions between voltaic and electrolytic cells, delving into their mechanisms, applications, and practical implications.
The Core Difference: Spontaneous vs. Non-Spontaneous Reactions
The most fundamental difference between voltaic and electrolytic cells lies in the nature of the redox reactions they facilitate:
-
Voltaic cells harness spontaneous redox reactions to generate electrical energy. The chemical reactions within the cell occur naturally and release energy, which is then converted into a usable electrical current. This energy release drives the electron flow from the anode (oxidation) to the cathode (reduction).
-
Electrolytic cells, conversely, utilize non-spontaneous redox reactions. They require an external source of electrical energy (like a battery) to force a chemical reaction to occur. The external voltage overcomes the inherent resistance of the non-spontaneous reaction, driving electrons against their natural flow from the cathode (reduction) to the anode (oxidation).
Anatomy of a Voltaic Cell: Harnessing Spontaneous Reactions
A typical voltaic cell consists of two half-cells: an anode and a cathode. Each half-cell contains an electrode immersed in an electrolyte solution. The electrodes are typically made of different metals or other conductive materials.
Key Components and their Roles:
-
Anode: The electrode where oxidation occurs. Electrons are released at the anode, making it negatively charged. The anode is consumed during the reaction.
-
Cathode: The electrode where reduction occurs. Electrons are gained at the cathode, making it positively charged. The cathode gains mass during the reaction.
-
Electrolyte: An ionic conductor (solution or molten salt) that allows the flow of ions between the anode and cathode. This maintains electrical neutrality within the cell.
-
Salt Bridge (or Porous Membrane): Connects the two half-cells, allowing the flow of ions to balance the charge buildup caused by the electron flow. This prevents the buildup of excess positive charge at the cathode and excess negative charge at the anode, which would halt the reaction.
A Classic Example: The Daniell Cell
The Daniell cell, a classic voltaic cell, beautifully illustrates these principles. It consists of a zinc electrode (anode) immersed in zinc sulfate solution and a copper electrode (cathode) immersed in copper sulfate solution, connected by a salt bridge. The spontaneous reaction is:
Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s)
Zinc readily oxidizes (loses electrons), becoming Zn²⁺ ions, while copper ions (Cu²⁺) reduce (gain electrons), forming solid copper. The electrons flow from the zinc anode, through an external circuit (powering a device), to the copper cathode. The salt bridge maintains electrical neutrality by allowing the flow of anions (SO₄²⁻) to the anode compartment and cations (Zn²⁺) to the cathode compartment.
Anatomy of an Electrolytic Cell: Driving Non-Spontaneous Reactions
Electrolytic cells are the reverse of voltaic cells. They use an external power source (a battery or other DC power supply) to drive a non-spontaneous redox reaction. The components are similar, but the roles and energy flow are reversed.
Key Components and their Roles:
-
Anode: The electrode where oxidation occurs. However, unlike in voltaic cells, it's positively charged because electrons are forced out of the anode by the external power source.
-
Cathode: The electrode where reduction occurs. It's negatively charged because electrons are forced into the cathode by the external power source.
-
Electrolyte: The ionic conductor that facilitates ion movement.
-
External Power Source: Provides the necessary voltage and current to drive the non-spontaneous reaction.
A Practical Example: Electrolysis of Water
The electrolysis of water is a prime example of an electrolytic process. By applying a sufficient voltage, water (H₂O) can be decomposed into hydrogen gas (H₂) and oxygen gas (O₂).
2H₂O(l) → 2H₂(g) + O₂(g)
In this process, water acts as both the electrolyte and the reactant. At the anode (positive electrode), water is oxidized to produce oxygen gas and hydrogen ions:
2H₂O(l) → O₂(g) + 4H⁺(aq) + 4e⁻
At the cathode (negative electrode), water is reduced to produce hydrogen gas and hydroxide ions:
4H₂O(l) + 4e⁻ → 2H₂(g) + 4OH⁻(aq)
Comparing Voltaic and Electrolytic Cells: A Table Summary
| Feature | Voltaic Cell | Electrolytic Cell |
|---|---|---|
| Reaction | Spontaneous redox reaction | Non-spontaneous redox reaction |
| Energy Flow | Chemical energy → Electrical energy | Electrical energy → Chemical energy |
| Anode | Negative, oxidation occurs | Positive, oxidation occurs |
| Cathode | Positive, reduction occurs | Negative, reduction occurs |
| External Source | None required | External power source (battery) required |
| Electron Flow | Anode → Cathode (through external circuit) | Cathode → Anode (forced by external source) |
| Applications | Batteries, fuel cells | Electroplating, electrolysis, metal refining |
Practical Applications: A Vast Landscape
Both voltaic and electrolytic cells have numerous applications across various industries and technologies.
Applications of Voltaic Cells:
-
Batteries: From everyday batteries in electronics to large-scale batteries for energy storage, voltaic cells power a significant portion of our modern world. These include primary cells (non-rechargeable) and secondary cells (rechargeable).
-
Fuel Cells: These generate electricity through the controlled electrochemical oxidation of a fuel, typically hydrogen. They offer high efficiency and are increasingly used in transportation and stationary power generation.
-
Sensors: Voltaic cells are employed in various electrochemical sensors to measure the concentration of specific ions or molecules.
-
Corrosion Prevention: Sacrificial anodes in voltaic cells are used to protect metal structures (like ships and pipelines) from corrosion.
Applications of Electrolytic Cells:
-
Electroplating: This process deposits a thin layer of metal onto a surface, improving its appearance, corrosion resistance, or other properties.
-
Electrolysis: Used to produce various chemicals, such as chlorine gas (from brine solutions) and aluminum metal (from alumina).
-
Metal Refining: Electrorefining purifies metals by selectively dissolving and redepositing them.
-
Water Purification: Electrolysis can be used to remove impurities from water.
-
Battery Charging: Rechargeable batteries (secondary cells) are charged using electrolytic cells, converting electrical energy back into chemical energy.
Beyond the Basics: Exploring Advanced Concepts
Understanding the fundamental differences between voltaic and electrolytic cells provides a solid foundation for exploring more advanced topics in electrochemistry:
-
Electrode Potentials and Cell Potentials: These values quantitatively describe the tendency of half-reactions to occur and predict the overall cell voltage.
-
Nernst Equation: This equation relates the cell potential to the concentrations of the reactants and products, showing the influence of concentration on cell voltage.
-
Electrode Kinetics: This involves studying the rate of electrode reactions and the factors that affect them (e.g., electrode material, surface area, temperature).
-
Corrosion and Passivation: Understanding electrochemical principles is crucial for mitigating corrosion and designing corrosion-resistant materials.
Conclusion: Mastering the Fundamentals of Electrochemical Cells
The distinction between voltaic and electrolytic cells is central to understanding electrochemistry. Voltaic cells harness spontaneous reactions to produce electrical energy, while electrolytic cells utilize an external power source to drive non-spontaneous reactions. Both cell types are essential components of many technologies, showcasing the breadth and importance of electrochemistry in modern science and engineering. By grasping the fundamental principles discussed here, you are well-equipped to tackle more complex electrochemical concepts and appreciate the vital role these cells play in shaping our world.
Latest Posts
Latest Posts
-
What Is The Driving Force For An Acid Base Neutralization Reaction
Apr 06, 2025
-
How To Find Basis Of A Subspace
Apr 06, 2025
-
How To Tell If A Transformation Is Linear
Apr 06, 2025
-
How To Find The Kernel Of A Linear Transformation
Apr 06, 2025
-
Examples Of Strong And Weak Bases
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Differences Between Voltaic And Electrolytic Cells . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
