Do Double Bonds Increase Boiling Point
Muz Play
Apr 03, 2025 · 5 min read
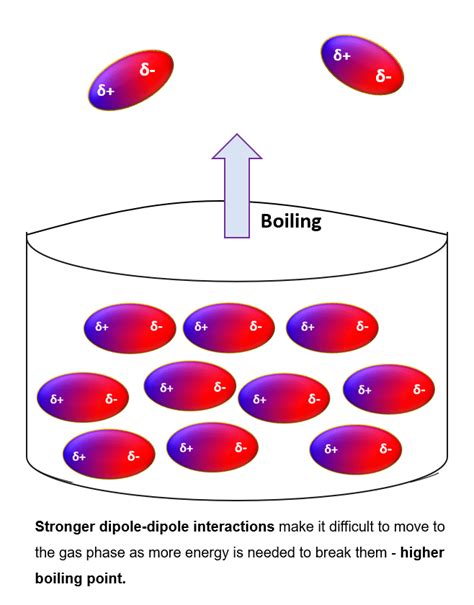
Table of Contents
Do Double Bonds Increase Boiling Point? A Deep Dive into Intermolecular Forces
The relationship between molecular structure and boiling point is a fundamental concept in chemistry. One frequently asked question revolves around the impact of double bonds on boiling points. While the presence of a double bond doesn't automatically guarantee a higher boiling point, it often plays a significant role, but the effect is nuanced and depends on several factors. This article will delve deep into the intricacies of this relationship, exploring the underlying principles and providing examples to illustrate the complexities involved.
Understanding Boiling Point and Intermolecular Forces
Before we investigate the influence of double bonds, let's establish a firm understanding of boiling point and the forces that govern it. The boiling point of a substance is the temperature at which its vapor pressure equals the atmospheric pressure. In simpler terms, it's the temperature at which a liquid transforms into a gas. This transition is dictated by the strength of intermolecular forces (IMFs) holding the molecules together in the liquid phase. The stronger the IMFs, the more energy (higher temperature) is required to overcome them and initiate boiling.
There are several types of IMFs, including:
1. London Dispersion Forces (LDFs):
These are the weakest type of IMF and are present in all molecules, regardless of polarity. LDFs arise from temporary, instantaneous dipoles created by the movement of electrons within a molecule. Larger molecules with more electrons generally experience stronger LDFs due to increased polarizability.
2. Dipole-Dipole Interactions:
These forces occur between polar molecules possessing permanent dipoles. The positive end of one molecule is attracted to the negative end of another, leading to a stronger attraction than LDFs.
3. Hydrogen Bonding:
This is a special type of dipole-dipole interaction involving hydrogen atoms bonded to highly electronegative atoms like oxygen, nitrogen, or fluorine. Hydrogen bonds are considerably stronger than typical dipole-dipole interactions.
The Role of Double Bonds in Intermolecular Forces
Double bonds, by their nature, affect molecular geometry and electron distribution, indirectly influencing IMFs. Let's explore how:
1. Molecular Shape and Packing:
Double bonds introduce rigidity into a molecule, affecting its overall shape. This altered shape influences how effectively molecules pack together in the liquid phase. Closer packing generally leads to stronger LDFs because of increased surface contact between molecules. A more compact, linear molecule will often have stronger LDFs than a branched isomer with the same molecular formula. This can lead to a higher boiling point, regardless of the presence of double bonds.
2. Polarity and Dipole-Dipole Interactions:
The presence of a double bond can affect the polarity of a molecule, leading to stronger or weaker dipole-dipole interactions. If the double bond introduces asymmetry in the electron distribution, it can create a dipole moment, enhancing dipole-dipole interactions and increasing the boiling point.
3. Conjugation and Resonance:
In molecules with conjugated double bonds (alternating single and double bonds), resonance structures can significantly alter the electron distribution. This delocalization of electrons can strengthen LDFs, leading to a higher boiling point compared to molecules lacking conjugation. The increased electron cloud mobility allows for stronger, more easily induced instantaneous dipoles.
4. Increased Molecular Weight:
Often, the introduction of a double bond increases the molecular weight of a compound. As molecular weight increases, the number of electrons also increases, enhancing LDFs and thus the boiling point. This effect is often prominent and can overshadow other factors.
Examples and Comparative Analysis
Let's examine some examples to illustrate the impact of double bonds on boiling points:
1. Alkanes vs. Alkenes:
Consider butane (C₄H₁₀) and 1-butene (C₄H₈). Butane, an alkane, boils at -0.5 °C, while 1-butene, an alkene with one double bond, boils at -6.3 °C. In this case, the double bond doesn't significantly raise the boiling point; the difference is minimal, and other factors are likely more influential.
2. Isomerism:
Compare 1-butene and 2-methylpropene (both C₄H₈). Both have a double bond, yet 1-butene boils at -6.3°C, and 2-methylpropene boils at -6.9 °C. The difference stems from the shape and packing: 1-butene's linear shape facilitates closer packing and slightly stronger LDFs.
3. Conjugated Systems:
Compare 1,3-butadiene (C₄H₆) with 1-butene (C₄H₈). 1,3-butadiene, with its conjugated double bonds, has a boiling point of -4.5°C, higher than 1-butene (-6.3°C), demonstrating the effect of conjugation on LDFs.
4. Polarity and Double Bonds:
Consider acetaldehyde (CH₃CHO) and ethane (C₂H₆). Acetaldehyde has a carbonyl group (C=O) containing a polar double bond, leading to a higher boiling point (20.2°C) compared to non-polar ethane (-88.6°C). The significant difference showcases the strong influence of dipole-dipole interactions.
Factors Complicating the Relationship
The relationship between double bonds and boiling points isn't straightforward. Other factors can significantly influence the boiling point, sometimes overriding the effects of double bonds:
- Branching: Branched molecules tend to have lower boiling points than their linear isomers due to less efficient packing and weaker LDFs.
- Hydrogen Bonding: The presence of hydrogen bonding often dominates other IMFs, significantly raising the boiling point, regardless of double bonds.
- Molecular Weight: The overall molecular weight has a major impact on boiling point due to its effect on LDFs.
Conclusion
The presence of a double bond doesn't automatically guarantee a higher boiling point. Its influence is indirect and depends heavily on several factors, including molecular shape, polarity, conjugation, and the interplay with other IMFs. While double bonds can sometimes lead to higher boiling points through enhanced LDFs or the introduction of stronger dipole-dipole interactions, other structural features, such as branching, hydrogen bonding, and molecular weight, often play a more dominant role. Careful analysis of all these aspects is crucial when predicting the boiling point of a compound. A thorough understanding of intermolecular forces is essential for interpreting the observed trends and making accurate predictions. The effect of a double bond should be considered within the context of the entire molecular structure and its influence on the overall intermolecular interactions. It's not a simple cause-and-effect relationship, but rather a complex interplay of various factors.
Latest Posts
Latest Posts
-
What Is The Difference Between Sensory Neurons And Motor Neurons
Apr 04, 2025
-
In What Order Do Events Occur During Mitosis
Apr 04, 2025
-
One Of The Geographical Advantages Of Early Rome
Apr 04, 2025
-
What Are Some Disadvantages Of Sexual Reproduction
Apr 04, 2025
-
M 1 And M 2 Peaks In Mass Spectrometry
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Do Double Bonds Increase Boiling Point . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
