How Do Intermolecular Forces Affect The Shape Of A Drop
Muz Play
Apr 06, 2025 · 6 min read
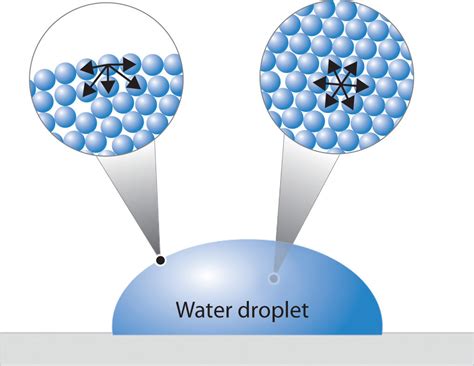
Table of Contents
How Do Intermolecular Forces Affect the Shape of a Drop?
The seemingly simple shape of a liquid drop—a sphere, or at least a close approximation—is a fascinating consequence of the interplay between intermolecular forces and external forces. Understanding this relationship provides insights into a wide range of phenomena, from the behavior of rain droplets to the design of inkjet printers. This article will delve into the intricacies of these forces and how they dictate the shape, size, and stability of liquid drops.
The Dominating Force: Surface Tension
At the heart of a liquid drop's shape is surface tension, a manifestation of intermolecular forces. These forces, primarily van der Waals forces and in some cases hydrogen bonding, act between molecules within the liquid. Molecules in the bulk of the liquid are surrounded by other molecules, experiencing attractive forces in all directions. This results in a net force of zero. However, molecules at the surface (the interface between the liquid and the surrounding medium, usually air) experience a net inward force. This inward pull minimizes the surface area, leading to the tendency of liquids to form spherical shapes—the shape that minimizes surface area for a given volume.
Types of Intermolecular Forces: A Deeper Dive
Several types of intermolecular forces contribute to surface tension and, consequently, the shape of a drop:
-
Van der Waals Forces: These are weak, short-range forces that arise from temporary fluctuations in electron distribution around atoms and molecules. They are ubiquitous and affect all substances, though their strength varies. Larger molecules with more electrons tend to exhibit stronger van der Waals forces.
-
Hydrogen Bonding: A stronger type of dipole-dipole interaction, hydrogen bonding occurs when a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) is attracted to another electronegative atom in a nearby molecule. Water, with its strong hydrogen bonding, exhibits exceptionally high surface tension.
-
Dipole-Dipole Forces: These forces occur between polar molecules, which have a permanent separation of charge. The positive end of one molecule attracts the negative end of another, leading to an attractive interaction.
-
Ion-Dipole Forces: These forces arise between ions and polar molecules. The charged ion attracts the oppositely charged end of the polar molecule. This interaction is particularly strong when the ion carries a high charge.
The strength of these intermolecular forces directly influences the surface tension. Higher surface tension means a stronger inward pull on the surface molecules, resulting in a more tightly curved surface and a more spherical drop.
External Forces Shaping the Drop
While surface tension is the primary force determining a liquid drop's shape, other external forces can significantly influence its form:
Gravity: The Downward Pull
Gravity exerts a downward force on the drop, counteracting the inward pull of surface tension. For small drops, surface tension dominates, resulting in a nearly perfect sphere. However, as the drop's size increases, the gravitational force becomes more significant, causing the drop to deform and flatten. This is why large raindrops are not perfectly spherical but rather somewhat flattened at the bottom.
External Pressure: Compressing the Drop
Changes in external pressure can also affect the drop's shape. Increased pressure can compress the drop, reducing its volume and potentially changing its overall shape. Conversely, a decrease in pressure can allow the drop to expand. This effect is more pronounced for drops with lower surface tension.
Electric Fields: Polarization and Deformation
The application of an electric field can significantly distort a liquid drop's shape. If the liquid is polar, the molecules align themselves with the field, leading to polarization and deformation of the drop. This effect is exploited in various applications, including inkjet printing and electrospinning.
Substrate Interactions: Wetting and Contact Angle
The interaction between the liquid drop and the substrate (the surface on which it rests) plays a crucial role in determining its shape. This interaction is quantified by the contact angle, the angle between the tangent to the liquid surface at the three-phase boundary (liquid-gas-solid) and the solid surface.
A low contact angle indicates good wetting, meaning the liquid spreads out on the surface. Conversely, a high contact angle indicates poor wetting, and the liquid tends to form a more spherical drop. The contact angle is determined by the balance of adhesive forces (between the liquid and the solid) and cohesive forces (between the liquid molecules).
The Mathematics of Drop Shape: Young's Equation
The relationship between surface tension, contact angle, and the forces at the three-phase boundary is described by Young's equation:
γ<sub>SG</sub> = γ<sub>SL</sub> + γ<sub>LG</sub>cosθ
where:
- γ<sub>SG</sub> is the surface tension between the solid and gas phases.
- γ<sub>SL</sub> is the surface tension between the solid and liquid phases.
- γ<sub>LG</sub> is the surface tension between the liquid and gas phases.
- θ is the contact angle.
This equation highlights the interplay of forces and demonstrates how the contact angle is a direct consequence of the balance between these interfacial tensions.
Applications and Further Considerations
Understanding how intermolecular forces affect the shape of a drop has far-reaching implications across various scientific disciplines and engineering applications:
-
Inkjet Printing: The precise control of drop formation and trajectory is crucial in inkjet printing. Manipulating the surface tension and charge of the ink allows for the precise deposition of ink droplets onto the substrate.
-
Spray Coating: The atomization and deposition of liquids in spray coating processes relies heavily on understanding surface tension and the forces that govern drop formation and breakup.
-
Microfluidics: The manipulation and control of fluids at the microscale are greatly influenced by surface tension and wetting properties. Microfluidic devices often rely on the precise control of droplet formation and movement.
-
Meteorology: The shape and size of raindrops are directly related to the balance between surface tension, gravity, and aerodynamic forces. Understanding these factors is crucial for modeling precipitation and weather patterns.
-
Material Science: The wetting behavior of liquids on different surfaces is critical in many material science applications, including adhesion, coating, and self-assembly.
Beyond the considerations discussed above, other factors such as temperature, viscosity, and the presence of impurities can also influence a liquid drop's shape. Temperature changes affect the strength of intermolecular forces, thereby affecting surface tension. Viscosity influences the drop's response to external forces, and impurities can alter the surface tension and wetting behavior.
Conclusion
The shape of a liquid drop is a beautiful example of the macroscopic manifestation of microscopic forces. The balance between the inward pull of surface tension, driven by intermolecular forces, and the various external forces determines the drop's shape, size, and stability. A deep understanding of these forces and their interactions is crucial across numerous scientific disciplines and technological applications, allowing for precise control and manipulation of liquid drops for a wide array of purposes. Further research into this complex interplay of forces continues to uncover new insights and expand our ability to leverage the unique properties of liquid drops.
Latest Posts
Latest Posts
-
Test Statistic For Hypothesis Test Calculator
Apr 06, 2025
-
What Is The Driving Force For An Acid Base Neutralization Reaction
Apr 06, 2025
-
How To Find Basis Of A Subspace
Apr 06, 2025
-
How To Tell If A Transformation Is Linear
Apr 06, 2025
-
How To Find The Kernel Of A Linear Transformation
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Do Intermolecular Forces Affect The Shape Of A Drop . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
