How Many Elements Are Gases At Room Temperature
Muz Play
Apr 02, 2025 · 6 min read
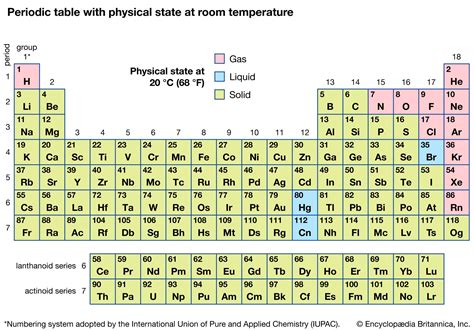
Table of Contents
How Many Elements Exist as Gases at Room Temperature?
The seemingly simple question, "How many elements are gases at room temperature?" leads us down a fascinating path exploring the periodic table, atomic structure, and the properties that determine a substance's state of matter. While the answer itself is relatively straightforward, understanding why certain elements exist as gases under standard conditions reveals a wealth of scientific principles. This article delves into this question, exploring the elements, their properties, and the underlying reasons for their gaseous nature.
Defining "Room Temperature" and "Standard Conditions"
Before we delve into the list of gaseous elements, it's crucial to define our terms. "Room temperature" is often loosely defined, but for scientific purposes, we typically use a standard temperature of 25° Celsius (298 Kelvin). Similarly, "standard conditions" often encompass a pressure of 1 atmosphere (atm). These parameters are essential because the state of matter of a substance can change with temperature and pressure variations. An element that's a gas at room temperature and standard pressure might become a liquid or even a solid at lower temperatures or higher pressures.
The Noble Gases: A Gaseous Family
The most straightforward group of elements that are gases at room temperature are the noble gases. Located in Group 18 of the periodic table, these elements—helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn)—are all monatomic gases. This means they exist as single atoms, not bound together in molecules.
Why are Noble Gases Gases?
The noble gases' gaseous nature stems from their electronic configuration. Their outermost electron shells are completely filled, giving them exceptional stability. They have little tendency to gain, lose, or share electrons, meaning they don't readily form chemical bonds with other atoms. This lack of bonding is a key factor in their existence as monatomic gases. The weak interatomic forces between these atoms make it easy for them to overcome their attractive forces and exist in a gaseous phase at room temperature.
Beyond the Noble Gases: Other Gaseous Elements
While the noble gases are the most well-known gaseous elements, several others exist in the gaseous state at room temperature and standard pressure.
-
Hydrogen (H₂): Hydrogen is the lightest element and exists as a diatomic molecule (H₂). Its small size and weak intermolecular forces contribute to its gaseous nature. The covalent bond between two hydrogen atoms is relatively weak, further facilitating its gaseous state at room temperature.
-
Nitrogen (N₂): Nitrogen is another diatomic gas, forming a strong triple bond (N≡N). Despite the strong bond, the relatively weak intermolecular forces between nitrogen molecules allow it to remain a gas at room temperature. Its abundance in the atmosphere is a testament to its stability in this state.
-
Oxygen (O₂): Similar to nitrogen, oxygen exists as a diatomic molecule (O₂) with a double bond. The relatively weak intermolecular forces allow it to remain a gas under standard conditions, making it crucial for respiration and many other biological processes.
-
Fluorine (F₂): Fluorine is a diatomic halogen gas. Its reactivity is exceptionally high, but at standard conditions, it exists as a gas due to weak intermolecular forces between the F₂ molecules.
-
Chlorine (Cl₂): Chlorine is another diatomic halogen gas with properties similar to fluorine, though less reactive. It exists as a gas at room temperature due to relatively weak intermolecular forces.
-
Bromine (Br₂): While the other halogens mentioned above are gases, bromine exists as a liquid at room temperature. However, it has a high vapor pressure, meaning it readily evaporates, and significant amounts of bromine vapor are present in equilibrium with the liquid at room temperature.
Understanding Intermolecular Forces: The Key to Gaseous States
The key to understanding why certain elements are gases at room temperature lies in the strength of intermolecular forces. These forces are the attractions between molecules (or atoms in the case of noble gases). Weak intermolecular forces allow the constituent particles to move freely, leading to the gaseous state. Stronger intermolecular forces can lead to liquids or solids, as the attractions hold the particles closer together.
Types of Intermolecular Forces
Several types of intermolecular forces influence the state of matter:
-
London Dispersion Forces (LDFs): These are the weakest intermolecular forces and are present in all molecules. They arise from temporary fluctuations in electron distribution, creating temporary dipoles. The strength of LDFs increases with the size and mass of the molecule.
-
Dipole-Dipole Forces: These forces exist between polar molecules—molecules with a permanent dipole moment due to unequal sharing of electrons. They are stronger than LDFs.
-
Hydrogen Bonding: This is a special type of dipole-dipole interaction that occurs when a hydrogen atom is bonded to a highly electronegative atom (such as oxygen, nitrogen, or fluorine). Hydrogen bonds are relatively strong and are crucial in the properties of water and many biological molecules.
In the case of gaseous elements at room temperature, the intermolecular forces are predominantly weak London Dispersion Forces, or in some cases, even weaker than typical LDFs due to extremely small size and negligible polarity.
The Periodic Table and Gaseous Elements: Trends and Predictions
The periodic table provides a valuable framework for understanding the trends in elemental properties, including the state of matter. While it's not possible to definitively predict the state of matter solely based on position in the periodic table, certain trends emerge:
-
Noble Gases (Group 18): All noble gases are gases at room temperature due to their stable electron configurations.
-
Halogens (Group 17): Halogens generally exhibit increasing boiling points as you go down the group, reflecting increasing LDF strength with increasing size. Fluorine and Chlorine are gases, while Bromine is a liquid, and Iodine is a solid at room temperature.
-
Hydrogen (Group 1): Despite its position, hydrogen's unique properties lead to its gaseous state at room temperature.
Factors Affecting State Transitions
It's important to remember that temperature and pressure significantly influence the state of matter. While the elements listed above are gases at room temperature and standard pressure, changes in these conditions can alter their state. For example, lowering the temperature can condense gases into liquids or solids, while increasing the pressure can force gases into a liquid or solid state, even at higher temperatures. This highlights the dynamic nature of matter and the interplay between intermolecular forces, temperature, and pressure.
Conclusion: A Summary of Gaseous Elements at Room Temperature
In conclusion, seven elements exist as gases under standard room temperature and pressure conditions: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) (noble gases); and hydrogen (H₂), nitrogen (N₂), oxygen (O₂), fluorine (F₂), and chlorine (Cl₂). Bromine (Br₂) while a liquid, has a substantial vapor pressure, contributing a significant gaseous component at room temperature. The primary factor determining this gaseous state is the weakness of intermolecular forces between the constituent atoms or molecules, allowing them to overcome attractive forces and exist freely in the gaseous phase. Understanding these intermolecular forces, atomic structures, and the influence of temperature and pressure provides a comprehensive understanding of why these elements exist as gases at room temperature. This knowledge is essential in various scientific disciplines, from atmospheric science to materials science and beyond.
Latest Posts
Latest Posts
-
The Heterozygote Expresses Phenotype Of Both Homozygotes
Apr 03, 2025
-
Difference Between A Somatic Cell And A Gamete
Apr 03, 2025
-
Match The Structure Process To The Letter
Apr 03, 2025
-
Find The Basis Of The Subspace
Apr 03, 2025
-
Why Is Immersion Oil Used With The 100x Objective
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How Many Elements Are Gases At Room Temperature . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
