How To Read An Ir Graph
Muz Play
Apr 03, 2025 · 6 min read
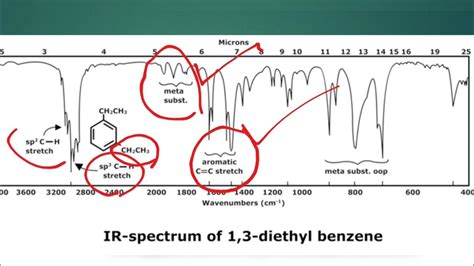
Table of Contents
How to Read an IR Graph: A Comprehensive Guide
Infrared (IR) spectroscopy is a powerful analytical technique used to identify and characterize molecules based on their vibrational modes. Understanding how to interpret an IR spectrum is crucial for chemists, material scientists, and anyone working with molecular compounds. This comprehensive guide will walk you through the essential elements of an IR graph, enabling you to confidently analyze and interpret the data.
Understanding the Fundamentals: The X and Y Axes
Before diving into complex spectra, let's establish a firm understanding of the axes. An IR spectrum is a plot of percent transmittance (%) on the Y-axis versus wavenumber (cm⁻¹) or wavelength (µm) on the X-axis.
The X-axis: Wavenumber and Wavelength
The X-axis represents the frequency of infrared radiation absorbed by the sample. This is typically expressed in wavenumbers (cm⁻¹), which is the reciprocal of the wavelength in centimeters. A higher wavenumber indicates a higher frequency and higher energy. While you might occasionally see wavelength (µm, micrometers) used, wavenumbers are far more common in IR spectroscopy. Remember this inverse relationship: higher wavenumber = shorter wavelength = higher energy.
The Y-axis: Percent Transmittance
The Y-axis depicts the percent transmittance of infrared radiation through the sample. Transmittance is the ratio of the intensity of the infrared light that passes through the sample to the intensity of the incident light. A 100% transmittance indicates that all the infrared light passed through the sample without being absorbed. Conversely, 0% transmittance means all the infrared light was absorbed by the sample. In practice, you'll see peaks representing absorbed frequencies. The deeper the dip (lower transmittance percentage), the stronger the absorption.
Deciphering the Peaks: Functional Group Identification
The key to interpreting an IR spectrum lies in identifying the characteristic absorption peaks associated with specific functional groups. Each functional group vibrates at a specific frequency, resulting in a unique absorption pattern. Understanding these patterns is crucial for identifying the components of an unknown sample. Let's examine some common functional groups and their corresponding absorption regions:
1. O-H Stretch (Alcohol, Carboxylic Acid)
- Wavenumber Range: 3200-3600 cm⁻¹ (broad peak for alcohols, sharper peak for carboxylic acids).
- Intensity: Strong
- Characteristics: The broadness of the O-H stretch in alcohols is due to hydrogen bonding. Carboxylic acids tend to exhibit a sharper peak due to dimerization through strong hydrogen bonding.
2. N-H Stretch (Amine, Amide)
- Wavenumber Range: 3300-3500 cm⁻¹
- Intensity: Medium to strong
- Characteristics: Primary amines (RNH₂) show two distinct N-H stretching bands, while secondary amines (R₂NH) display only one.
3. C-H Stretch (Alkane, Alkene, Alkyne)
- Wavenumber Range: 2850-3000 cm⁻¹ (alkanes), 3000-3100 cm⁻¹ (alkenes), 3300 cm⁻¹ (alkynes)
- Intensity: Strong
- Characteristics: The position of the C-H stretch can help distinguish between different types of carbon-hydrogen bonds. Alkenes and alkynes show C-H stretches at higher wavenumbers due to increased bond strength.
4. C≡N Stretch (Nitrile)
- Wavenumber Range: 2200-2300 cm⁻¹
- Intensity: Medium
- Characteristics: A sharp peak in this region is indicative of a nitrile group.
5. C=O Stretch (Ketone, Aldehyde, Carboxylic Acid, Ester, Amide)
- Wavenumber Range: 1680-1750 cm⁻¹ (varies depending on the functional group)
- Intensity: Strong
- Characteristics: This is arguably one of the most important peaks to identify. The exact position of the C=O stretch is highly dependent on the surrounding chemical environment.
6. C=C Stretch (Alkene)
- Wavenumber Range: 1620-1680 cm⁻¹
- Intensity: Medium
- Characteristics: The presence of this peak confirms the presence of a carbon-carbon double bond.
7. Fingerprint Region (Below 1500 cm⁻¹)
- Wavenumber Range: Below 1500 cm⁻¹
- Intensity: Variable
- Characteristics: This region is highly complex and contains many overlapping peaks. While difficult to interpret individually, the overall pattern in this region can be used to distinguish between different compounds. It's often compared to a fingerprint; unique to a specific molecule.
Analyzing the Spectrum: A Step-by-Step Approach
Analyzing an IR spectrum involves a systematic approach:
-
Identify prominent peaks: Begin by identifying the strong and medium intensity peaks above 1500 cm⁻¹. These peaks often correspond to key functional groups.
-
Assign functional groups: Based on the wavenumber and intensity of the prominent peaks, assign potential functional groups using the information provided in the previous section.
-
Consider peak shape and width: The shape and width of the peaks provide additional information. For instance, a broad peak often suggests hydrogen bonding.
-
Analyze the fingerprint region: Although challenging, the fingerprint region can provide crucial information to distinguish between isomers or closely related compounds. Comparing your spectrum to a spectral database can greatly help in this region.
-
Correlate with known spectra: If possible, compare your spectrum to known spectra of similar compounds. Databases like the NIST Chemistry WebBook can be incredibly helpful.
-
Consider the context: The information about the sample's origin and preparation is crucial. This context can help eliminate possibilities and refine your analysis.
Common Pitfalls and Troubleshooting
Even with careful analysis, several challenges can arise:
-
Overlapping peaks: Overlapping peaks can make it difficult to accurately assign functional groups. Advanced techniques like Fourier self-deconvolution can help resolve overlapping peaks.
-
Weak peaks: Weak peaks might be difficult to distinguish from baseline noise. Improving the signal-to-noise ratio by increasing the number of scans can alleviate this issue.
-
Solvent interference: The solvent used to prepare the sample can interfere with the spectrum. Choosing a suitable solvent is crucial for obtaining a clean spectrum. Consider using deuterated solvents when analyzing certain types of samples.
-
Sample preparation: Improper sample preparation, such as insufficient drying or poor sample handling, can lead to inaccurate results. Ensure a thorough understanding of correct sample preparation for your specific analysis.
-
Incorrect instrument calibration: An incorrectly calibrated instrument will lead to inaccurate wavenumbers and potentially misinterpretations of the data.
Advanced Techniques and Applications
Beyond the basic principles, several advanced techniques and applications further enhance the power of IR spectroscopy:
-
Attenuated Total Reflectance (ATR): ATR avoids the need for sample preparation such as creating KBr pellets and allows for direct analysis of solid samples. This is a huge advantage when dealing with limited or precious sample amounts.
-
Gas-phase IR: Analyzing gaseous samples provides unique information not available from liquid or solid-state analysis.
-
Difference Spectroscopy: Subtracting spectra can help highlight specific changes in a system, such as chemical reactions or structural modifications.
-
Quantitative IR: IR spectroscopy can be used for quantitative analysis, providing information about the concentration of specific components in a mixture.
-
Coupling with other techniques: IR spectroscopy is often coupled with other analytical techniques like Gas Chromatography (GC) or High-Performance Liquid Chromatography (HPLC) for comprehensive characterization of complex samples.
Conclusion
Interpreting IR spectra requires practice and a thorough understanding of the fundamental principles. By mastering the techniques outlined in this guide, you can confidently analyze IR graphs, identify functional groups, and gain valuable insights into the molecular structure and composition of various samples. Remember that the fingerprint region and correlation with known spectra are often crucial for precise identification. Consistent practice and a comprehensive understanding of spectral features will significantly enhance your proficiency in this powerful analytical method. Don't hesitate to explore advanced techniques and applications to further deepen your expertise.
Latest Posts
Latest Posts
-
Periodic Trends Worksheet Atomic Radius Answers
Apr 03, 2025
-
Completa Este Parrafo Con Las Preposiciones Por O Para
Apr 03, 2025
-
Sequence Of Events In Muscle Contraction
Apr 03, 2025
-
When Making An Occupied Bed The Nurse Aide Should
Apr 03, 2025
-
Solve An Inequality And Graph The Solution
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about How To Read An Ir Graph . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
