In Hydrolysis Reactions Compounds React With
Muz Play
Apr 05, 2025 · 6 min read
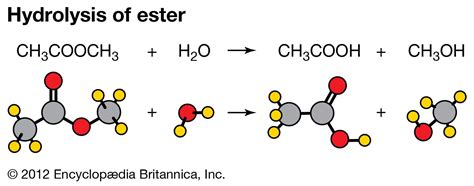
Table of Contents
In Hydrolysis Reactions, Compounds React With Water: A Deep Dive
Hydrolysis, derived from the Greek words "hydro" (water) and "lysis" (to break down), is a fundamental chemical process where water molecules break down a compound. This reaction is ubiquitous in nature and plays a vital role in various biological and industrial processes. Understanding how hydrolysis works, the different types of hydrolysis reactions, and their applications is crucial in various scientific fields. This comprehensive article delves deep into the intricacies of hydrolysis reactions, explaining what compounds react with water, the mechanisms involved, and their significant implications.
What is Hydrolysis? The Fundamental Reaction
At its core, hydrolysis is a chemical reaction where a water molecule (H₂O) reacts with another compound, breaking the compound's bonds and forming two or more new products. This process often involves the addition of a hydrogen ion (H⁺) from the water molecule to one fragment and a hydroxide ion (OH⁻) to another fragment of the original compound. The overall effect is the decomposition of the original substance. Think of it as water acting as a "cleaver," splitting larger molecules into smaller, more manageable components.
The general equation for a hydrolysis reaction can be represented as:
AB + H₂O → AH + BOH
Where:
- AB represents the compound undergoing hydrolysis.
- H₂O is the water molecule.
- AH and BOH are the products formed after the bond in AB is broken and the H⁺ and OH⁻ ions are added.
Types of Hydrolysis Reactions: A Categorical Overview
Hydrolysis reactions are diverse and can be classified based on several factors, including the type of compound undergoing hydrolysis and the reaction mechanism. Some major categories include:
1. Salt Hydrolysis: The Ionic Breakdown
Salt hydrolysis involves the reaction of a salt with water to produce an acidic or basic solution. This occurs when the salt is derived from a weak acid or a weak base (or both). When a salt of a weak acid and a strong base hydrolyzes, the resulting solution is basic. Conversely, the hydrolysis of a salt from a strong acid and a weak base results in an acidic solution. For example, the hydrolysis of sodium acetate (NaCH₃COO), the salt of a strong base (NaOH) and a weak acid (CH₃COOH), produces a basic solution.
Mechanism: The anion of the weak acid (e.g., CH₃COO⁻) reacts with water, accepting a proton (H⁺) to form the weak acid and hydroxide ions (OH⁻), increasing the solution's pH.
2. Ester Hydrolysis: Breaking Ester Bonds
Ester hydrolysis is a particularly important type of hydrolysis reaction involving the breakdown of esters. Esters are organic compounds commonly found in fats, oils, and fragrances. In this reaction, an ester reacts with water to produce a carboxylic acid and an alcohol. This reaction is often catalyzed by acids or bases, significantly accelerating the hydrolysis rate. For instance, the hydrolysis of ethyl acetate (CH₃COOCH₂CH₃) yields acetic acid (CH₃COOH) and ethanol (CH₂CH₂OH).
Mechanism: Acid-catalyzed ester hydrolysis involves protonation of the carbonyl oxygen in the ester, making it more susceptible to nucleophilic attack by water. Base-catalyzed ester hydrolysis involves the hydroxide ion directly attacking the carbonyl carbon.
3. Amide Hydrolysis: Breaking Peptide Bonds
Amide hydrolysis is crucial in biochemistry, specifically in the digestion of proteins. Amides, which are characterized by the –CONH– group, are broken down into a carboxylic acid and an amine. Peptide bonds, which link amino acids in proteins, are a type of amide bond. Amide hydrolysis, like ester hydrolysis, can be catalyzed by acids or bases. The hydrolysis of a peptide bond results in the formation of individual amino acids.
Mechanism: Similar to ester hydrolysis, acid-catalyzed amide hydrolysis involves protonation of the carbonyl oxygen, increasing susceptibility to nucleophilic attack. Base-catalyzed amide hydrolysis involves hydroxide ion attack on the carbonyl carbon. This process is slower than ester hydrolysis because the amide bond is less reactive.
4. Carbohydrate Hydrolysis: Breaking Down Sugars
Carbohydrates, like starch and cellulose, are composed of long chains of sugar molecules linked together. Carbohydrate hydrolysis involves breaking these glycosidic bonds, releasing individual sugar molecules (monosaccharides). This process is essential for the digestion of carbohydrates in living organisms. Enzymes, like amylase, play a critical role in catalyzing carbohydrate hydrolysis.
Mechanism: Specific enzymes catalyze the hydrolysis of glycosidic bonds, breaking them down into individual monosaccharide units. The mechanism involves a precise positioning of the substrate and the catalytic site of the enzyme.
5. Hydrolysis of Inorganic Compounds: Beyond Organics
Hydrolysis isn't limited to organic compounds. Many inorganic compounds also undergo hydrolysis. For example, the hydrolysis of metal ions, such as aluminum ions (Al³⁺), forms hydrated metal ions and changes the pH of the solution. Similarly, the hydrolysis of certain salts, as mentioned earlier, can produce acidic or basic solutions depending on the nature of the constituent ions.
Mechanism: Inorganic hydrolysis often involves the reaction of a metal cation with water, leading to the formation of a hydrated metal ion and the release of H⁺ ions.
Factors Affecting Hydrolysis Reactions: Speed and Efficiency
Several factors influence the rate and efficiency of hydrolysis reactions:
-
Temperature: Higher temperatures generally increase the rate of hydrolysis reactions. The increased kinetic energy facilitates the breaking of bonds.
-
pH: The acidity or basicity of the solution significantly impacts hydrolysis rates. Acidic or basic catalysts can substantially accelerate the reaction.
-
Catalyst: Enzymes and other catalysts can significantly speed up hydrolysis reactions by lowering the activation energy required for the reaction to proceed.
-
Concentration: Higher concentrations of reactants generally lead to faster reaction rates.
-
Nature of the compound: The specific chemical structure of the compound undergoing hydrolysis plays a vital role in determining the reaction's rate and mechanism. Stronger bonds, for instance, may require more energy to break.
Applications of Hydrolysis: A Wide Spectrum of Uses
Hydrolysis reactions have widespread applications across various fields:
1. Digestion and Metabolism: Life's Essential Chemistry
Hydrolysis is fundamental to digestion and metabolism in living organisms. The breakdown of carbohydrates, proteins, and fats into smaller, absorbable molecules relies heavily on hydrolysis reactions catalyzed by enzymes.
2. Industrial Processes: Synthesizing and Degrading
Hydrolysis plays a critical role in various industrial processes. It's used in the production of numerous chemicals, including the synthesis of esters and amides. Conversely, hydrolysis is also used for the degradation of polymers, such as the breakdown of cellulose in the paper industry.
3. Environmental Science: Waste Treatment and Remediation
Hydrolysis is employed in environmental remediation for breaking down pollutants and hazardous waste. The hydrolysis of certain compounds can transform them into less harmful substances.
4. Food and Beverage Industry: Processing and Preservation
Hydrolysis is essential in the food industry for processes like the production of sweeteners from starch and the modification of proteins for improved texture and functionality.
5. Pharmaceutical Industry: Drug Synthesis and Degradation
Hydrolysis reactions play a crucial role in drug synthesis and can also affect drug stability and degradation. Understanding the hydrolysis of drug molecules is essential for formulating stable and effective medications.
Conclusion: The Importance of Hydrolysis
Hydrolysis reactions are a cornerstone of chemistry, biology, and various industrial processes. Their profound impact stems from the ability of water to break down a wide range of compounds, leading to the formation of new substances with altered properties. Understanding the different types of hydrolysis reactions, the factors influencing their rates, and their vast applications is crucial for advancing knowledge in multiple scientific fields and developing innovative technological solutions. From digestion to industrial processes, hydrolysis remains an indispensable chemical process shaping our world.
Latest Posts
Latest Posts
-
Evidence Of Chemical Change Lab Answers
Apr 06, 2025
-
How Do Gametes Differ From Somatic Cells
Apr 06, 2025
-
Magnetic Field Of A Loop Formula
Apr 06, 2025
-
Characteristic Polynomial Of A 2x2 Matrix
Apr 06, 2025
-
Which Is The Best Example Of Immiscible Liquids
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about In Hydrolysis Reactions Compounds React With . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
