Iron Iii Chloride Test For Aspirin
Muz Play
Apr 05, 2025 · 5 min read
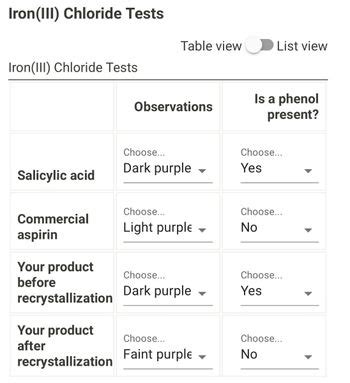
Table of Contents
Iron(III) Chloride Test for Aspirin: A Comprehensive Guide
The iron(III) chloride test, also known as the ferric chloride test, is a common qualitative test used to detect the presence of phenols and enols. While not specific to aspirin (acetylsalicylic acid) itself, it can be a useful tool in assessing the purity and potential degradation of aspirin samples. This comprehensive guide delves into the intricacies of this test, exploring its mechanism, procedure, limitations, and interpretation, particularly within the context of aspirin analysis.
Understanding the Chemistry Behind the Test
The iron(III) chloride test relies on the reaction between the ferric ion (Fe³⁺) and phenolic compounds. Phenols possess a hydroxyl group (-OH) directly attached to an aromatic ring. This hydroxyl group is weakly acidic and can donate a proton to the ferric ion, forming a colored complex. The color produced varies depending on the specific phenol involved, but often ranges from a deep purple or violet to a reddish-brown or green.
Aspirin, in its pure form, does not directly participate in this reaction because it lacks a free phenolic hydroxyl group. The acetyl group (-COCH₃) protects the hydroxyl group on the salicylic acid moiety. However, aspirin can degrade into salicylic acid, particularly under conditions of hydrolysis (reaction with water). Salicylic acid, possessing a free phenolic hydroxyl group, reacts readily with ferric chloride. Therefore, a positive iron(III) chloride test for an aspirin sample often indicates the presence of salicylic acid, either as an impurity in the original aspirin or as a product of its degradation.
The Reaction Mechanism
The reaction involves the formation of a complex between the ferric ion and the phenolic hydroxyl group. The phenolic oxygen donates a lone pair of electrons to the ferric ion, forming a coordinate covalent bond. This coordination complex exhibits intense color due to charge transfer interactions between the iron and the aromatic ring. The exact structure of the complex depends on the phenol's structure and the reaction conditions, explaining the variation in observed colors.
Simplified Reaction (with Salicylic Acid):
3 Salicylic Acid + FeCl₃ → [Fe(Salicylate)₃]³⁻ + 3 H⁺ + 3 Cl⁻
Conducting the Iron(III) Chloride Test for Aspirin
The test itself is relatively simple to perform, requiring minimal equipment and materials. Here's a step-by-step guide:
Materials Required:
- Aspirin sample (powder or tablet)
- 1% aqueous solution of ferric chloride (FeCl₃)
- Distilled water
- Test tubes or small beakers
- Stirring rod
Procedure:
-
Prepare the Aspirin Solution: Dissolve a small amount of aspirin (approximately 50-100 mg) in a small volume (around 2-3 mL) of distilled water. Aspirin has limited solubility in water, so gentle heating might be necessary to aid dissolution. Ensure the solution is clear or only slightly cloudy before proceeding.
-
Add Ferric Chloride Solution: Add several drops (approximately 5-10 drops) of the 1% ferric chloride solution to the aspirin solution.
-
Observe the Color Change: Gently stir the mixture and observe any color change. A positive result is indicated by the appearance of a characteristic color, ranging from violet, purple, red-brown, to green. The intensity of the color is often related to the concentration of the phenolic compound present.
-
Control Experiment: It's crucial to conduct a control experiment using only distilled water and ferric chloride solution. This helps to eliminate any potential interference from the water or reagents.
Interpreting the Results
The interpretation of the iron(III) chloride test requires careful consideration.
-
Positive Result (Color Change): A positive test (color change) indicates the presence of a phenolic compound, most likely salicylic acid in the context of aspirin analysis. This could mean:
- Impure Aspirin: The original aspirin sample contained salicylic acid as an impurity. This is a common issue in aspirin production, and the presence of salicylic acid can affect the therapeutic effectiveness and potentially lead to unwanted side effects.
- Degraded Aspirin: The aspirin sample has undergone hydrolysis, converting some of the aspirin into salicylic acid. This degradation can be accelerated by exposure to moisture, heat, or light.
-
Negative Result (No Color Change): A negative test (no color change) suggests the absence of significant amounts of phenolic compounds, which can indicate pure aspirin. However, it's important to remember that this doesn't guarantee absolute purity. Other impurities might be present that don't react with ferric chloride.
Limitations of the Iron(III) Chloride Test
The iron(III) chloride test has some limitations:
- Lack of Specificity: The test is not specific to salicylic acid. Other phenolic compounds will also yield positive results.
- Sensitivity: The test's sensitivity can be limited, meaning it might not detect very small amounts of salicylic acid.
- Interference: Certain substances can interfere with the test, leading to false positive or false negative results.
- Qualitative Nature: The test is qualitative; it indicates the presence or absence of a phenol, not its exact quantity.
Advanced Techniques for Aspirin Purity Analysis
While the iron(III) chloride test provides a quick and simple assessment of aspirin purity, more sophisticated techniques are necessary for accurate quantitative analysis. These include:
- High-Performance Liquid Chromatography (HPLC): HPLC allows for the precise separation and quantification of aspirin and salicylic acid, providing an accurate measure of purity.
- Titration: Titration methods can be employed to determine the aspirin content, providing a quantitative measure of its concentration.
- Spectrophotometry: Spectrophotometric techniques can measure the absorbance of aspirin solutions at specific wavelengths, which can be correlated to its concentration.
Conclusion
The iron(III) chloride test serves as a valuable preliminary screening tool for assessing the potential presence of salicylic acid in an aspirin sample. Its simplicity and ease of performance make it a useful educational tool and a rapid check for significant levels of degradation or impurity. However, its limitations should be acknowledged. For accurate and comprehensive purity analysis of aspirin, advanced techniques such as HPLC or titration are essential. Understanding both the capabilities and limitations of the iron(III) chloride test allows for its effective use as part of a broader analytical strategy. Remember to always follow appropriate safety procedures when handling chemicals in any laboratory setting. The use of protective eyewear and gloves is highly recommended. Proper disposal of chemical waste is also crucial for environmental protection. Further research into aspirin synthesis, degradation pathways, and quality control methods can enhance understanding and improve the quality and safety of aspirin products available to consumers. Proper storage conditions, including protection from moisture, heat, and light, play a vital role in maintaining the integrity of aspirin products. Continuous monitoring and improvement of manufacturing processes and analytical techniques are critical for ensuring the consistent quality and safety of pharmaceutical products.
Latest Posts
Latest Posts
-
All Viruses Leave A Cell By Exocytosis
Apr 06, 2025
-
When Two Monosaccharides Undergo A Dehydration Synthesis
Apr 06, 2025
-
Area Enclosed By A Parametric Curve
Apr 06, 2025
-
Diagram On How Geothermal Energy Works
Apr 06, 2025
-
How To Calculate The Heat Capacity Of A Calorimeter
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Iron Iii Chloride Test For Aspirin . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
