Is Cl A Metal Or Nonmetal
Muz Play
Apr 02, 2025 · 5 min read
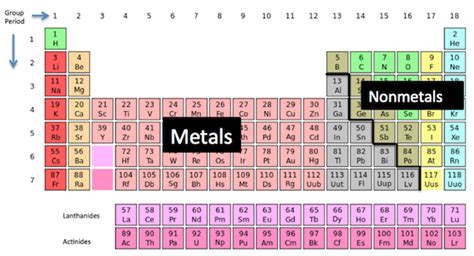
Table of Contents
Is Chlorine (Cl) a Metal or Nonmetal? A Deep Dive into its Properties and Reactivity
Chlorine (Cl), element number 17 on the periodic table, is a crucial element with a wide range of applications, from water purification to the production of plastics. A common question that arises, especially for students learning chemistry, is whether chlorine is a metal or a nonmetal. The answer is clear: chlorine is a nonmetal. But understanding why requires delving into its atomic structure, chemical properties, and behavior. This comprehensive article will explore these aspects in detail, providing a complete picture of chlorine's nature and its place within the broader context of the periodic table.
Understanding the Basics: Metals vs. Nonmetals
Before we dive into the specifics of chlorine, let's establish a clear understanding of the fundamental differences between metals and nonmetals. These differences are rooted in their atomic structures and how their electrons behave.
Metals, typically located on the left side of the periodic table, are characterized by:
- Low ionization energies: They readily lose electrons to form positive ions (cations).
- High electrical and thermal conductivity: Electrons are delocalized and can move freely throughout the metal structure.
- Malleability and ductility: They can be hammered into sheets (malleability) and drawn into wires (ductility).
- Metallic luster: They possess a shiny appearance.
Nonmetals, primarily located on the right side of the periodic table, exhibit contrasting properties:
- High ionization energies: They tend to gain electrons to form negative ions (anions).
- Poor electrical and thermal conductivity: Electrons are localized and not free to move easily.
- Brittle solids (generally): They lack the malleability and ductility of metals.
- Lack of metallic luster: They often appear dull or have various colors.
Chlorine: A Nonmetal's Characteristics
Chlorine's classification as a nonmetal is evident from its properties:
1. Electronic Configuration and Ionization Energy:
Chlorine's atomic number is 17, meaning it has 17 electrons. Its electronic configuration is [Ne] 3s²3p⁵. This means it has seven valence electrons in its outermost shell. To achieve a stable octet (eight electrons in its outermost shell), chlorine readily gains one electron, forming a chloride ion (Cl⁻). This electron gain is indicative of a nonmetal. The high ionization energy required to remove an electron from chlorine further supports its nonmetallic nature.
2. Chemical Reactivity and Compound Formation:
Chlorine is a highly reactive nonmetal. Its strong tendency to gain an electron makes it a powerful oxidizing agent. This reactivity is showcased in its numerous compounds:
- Ionic compounds: Chlorine readily forms ionic bonds with metals, resulting in compounds like sodium chloride (NaCl, table salt), where chlorine exists as the chloride ion (Cl⁻).
- Covalent compounds: Chlorine also forms covalent bonds with other nonmetals, sharing electrons to achieve a stable octet. Examples include hydrogen chloride (HCl), a strong acid, and carbon tetrachloride (CCl₄), a former industrial solvent. The existence of covalent compounds is a characteristic feature of nonmetals.
3. Physical Properties:
Chlorine's physical properties further cement its classification as a nonmetal:
- State at room temperature: Chlorine exists as a diatomic gas (Cl₂) at room temperature, unlike most metals which are solid.
- Color and Odor: Chlorine gas is a yellowish-green gas with a pungent, suffocating odor. This is atypical of metals.
- Electrical and Thermal Conductivity: Chlorine gas is a poor conductor of electricity and heat, another hallmark of nonmetals.
- Hardness and Brittleness: Solid chlorine (at very low temperatures) is brittle, unlike the malleable nature of metals.
Chlorine's Importance and Applications
Chlorine's nonmetallic nature underpins its versatility and importance in various applications:
1. Water Purification:
Chlorine is widely used in water treatment plants to disinfect drinking water and swimming pools. Its strong oxidizing properties effectively kill harmful bacteria and viruses, ensuring safe water consumption. This application relies on chlorine's chemical reactivity, a nonmetal characteristic.
2. Industrial Applications:
Chlorine is a key component in the production of a wide range of industrial chemicals, including:
- Plastics (PVC): Polyvinyl chloride (PVC) is a common plastic used in pipes, flooring, and other applications. Chlorine is a crucial ingredient in its synthesis.
- Solvents: Various chlorine-containing solvents are used in industrial processes, though their use is decreasing due to environmental concerns.
- Bleaches: Chlorine-based bleaches are commonly used in household cleaning and textile industries.
- Pharmaceuticals and pesticides: Chlorine is present in many pharmaceuticals and pesticides, highlighting its importance in chemical synthesis.
3. Other Applications:
Beyond these major applications, chlorine also finds use in:
- Metal processing: Chlorine is used in the extraction and purification of certain metals.
- Fire extinguishers: Some fire extinguishers utilize chlorine compounds.
The Periodic Table and Chlorine's Position
Chlorine's position in the periodic table, in Group 17 (also known as the halogens), strongly indicates its nonmetallic nature. Halogens are known for their high electronegativity (a measure of an atom's ability to attract electrons) and their tendency to form anions. This group consists entirely of nonmetals, further confirming chlorine's classification.
Addressing Potential Confusion: Halogens and their unique characteristics
While chlorine's nonmetallic nature is clear, it's important to note that halogens display some unique characteristics that might initially seem contradictory. For instance, iodine, another halogen, appears as a dark, shiny solid, which may seem metallic. However, this shiny appearance is not indicative of metallic bonding or properties; it’s a characteristic of the element itself and doesn't alter its nonmetallic classification. The key is to focus on the underlying chemical behavior: electron gain to achieve a stable octet and the lack of metallic bonding.
Conclusion: Chlorine - Undeniably a Nonmetal
In summary, chlorine's electronic configuration, chemical reactivity, physical properties, and its position in the periodic table conclusively demonstrate its classification as a nonmetal. Its importance in various industries and its role in everyday life highlight the significance of this highly reactive and versatile element, whose unique characteristics stem directly from its nonmetallic nature. Understanding the fundamental differences between metals and nonmetals and applying this understanding to the specific case of chlorine provides a solid foundation for comprehending its chemical behavior and diverse applications. The fact that chlorine readily forms anions, possesses high electronegativity, and exhibits poor electrical conductivity solidifies its place firmly within the nonmetal category.
Latest Posts
Latest Posts
-
The Echelon Form Of A Matrix Is Unique
Apr 03, 2025
-
How To Write An Equation For A Vertical Line
Apr 03, 2025
-
The Heterozygote Expresses Phenotype Of Both Homozygotes
Apr 03, 2025
-
Difference Between A Somatic Cell And A Gamete
Apr 03, 2025
-
Match The Structure Process To The Letter
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is Cl A Metal Or Nonmetal . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
