Is Corrosion A Physical Or Chemical Property
Muz Play
Apr 05, 2025 · 5 min read
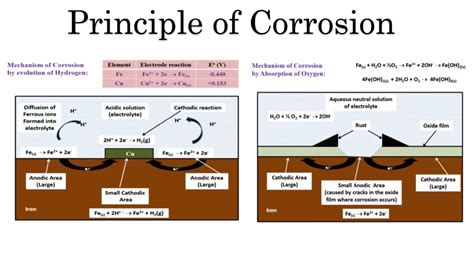
Table of Contents
Is Corrosion a Physical or Chemical Property? A Deep Dive
Corrosion, the deterioration of a material due to its reaction with its environment, is a topic of significant importance across numerous industries. Understanding its nature, however, often leads to the fundamental question: is corrosion a physical or chemical property? The short answer is chemical, but the nuanced answer involves a deeper exploration of the processes involved and a closer examination of the distinction between physical and chemical changes.
Understanding Physical vs. Chemical Changes
Before delving into the specifics of corrosion, let's establish a clear understanding of the difference between physical and chemical changes.
Physical changes affect the form of a substance, but not its chemical composition. These changes are often reversible. Examples include:
- Changes of state: Melting ice (solid to liquid), boiling water (liquid to gas).
- Shape changes: Bending a metal rod, cutting paper.
- Dissolution (in some cases): Dissolving sugar in water (sugar retains its chemical identity).
Chemical changes, on the other hand, involve a transformation of the substance's chemical composition, creating new substances with different properties. These changes are usually irreversible. Examples include:
- Burning: Wood reacting with oxygen to produce ash and gases.
- Rusting: Iron reacting with oxygen and water to form iron oxide (rust).
- Decomposition: Breaking down hydrogen peroxide into water and oxygen.
The Chemical Nature of Corrosion
Corrosion is fundamentally a chemical process because it involves the chemical transformation of a material due to its reaction with its environment. The process usually involves a redox (reduction-oxidation) reaction where electrons are transferred between the metal and its environment.
Let's examine the classic example of rusting, the corrosion of iron:
Rusting: A Detailed Look at a Chemical Reaction
The rusting of iron is a complex electrochemical process. It doesn't simply involve iron reacting directly with oxygen in the air. Instead, it requires the presence of both oxygen and water (or moisture). The process can be summarized as follows:
-
Oxidation: Iron atoms lose electrons (oxidation) to form iron(II) ions (Fe²⁺). This occurs at anodic sites on the iron surface. The equation is: Fe → Fe²⁺ + 2e⁻
-
Reduction: Oxygen molecules gain electrons (reduction) in the presence of water, forming hydroxide ions (OH⁻). This happens at cathodic sites on the iron surface. The simplified equation is: O₂ + 2H₂O + 4e⁻ → 4OH⁻
-
Formation of Iron Oxide: The iron(II) ions (Fe²⁺) react with hydroxide ions (OH⁻) to form iron(II) hydroxide, Fe(OH)₂. This can further react with oxygen to form iron(III) oxide-hydroxide (Fe₂O₃·xH₂O), which is what we commonly know as rust.
The overall reaction can be represented as:
4Fe + 3O₂ + 6H₂O → 4Fe(OH)₃ → 2Fe₂O₃·3H₂O + 3H₂O
Key Indicators of a Chemical Change in Corrosion
Several observations confirm that corrosion is a chemical change:
- Irreversibility: Rust cannot be easily converted back to iron. The original material is fundamentally altered.
- Formation of New Substances: The formation of rust (iron oxide) is a distinct substance with properties entirely different from those of iron.
- Energy Changes: Corrosion often involves the release of energy (exothermic reaction), as seen in the heat generated during the rusting process.
- Gas Evolution: In some corrosion processes, gases like hydrogen are evolved.
- Change in Mass: The corroded metal typically exhibits a change in mass, due to the addition of oxygen and other elements.
Types of Corrosion and Their Chemical Mechanisms
Corrosion manifests in various forms, each involving specific chemical reactions and influencing factors:
1. Uniform Corrosion
This is the most common type, where the corrosion occurs evenly across the metal surface. The chemical mechanism is generally a straightforward oxidation-reduction reaction as seen in the rusting of iron.
2. Galvanic Corrosion
This occurs when two dissimilar metals are in electrical contact in the presence of an electrolyte (like seawater). The more active metal corrodes more rapidly, acting as the anode, while the less active metal acts as the cathode.
3. Pitting Corrosion
Localized corrosion that leads to the formation of small pits or holes on the metal surface. This is often initiated by defects on the metal surface or by the presence of aggressive ions like chloride. The chemical reactions involved are similar to uniform corrosion but are concentrated in specific areas.
4. Crevice Corrosion
This type of corrosion occurs in confined spaces, like crevices or joints, where stagnant solutions can accumulate. The chemical environment within the crevice becomes highly concentrated and acidic, accelerating the corrosion process.
5. Stress Corrosion Cracking
This is a form of corrosion where tensile stresses in the metal accelerate the corrosion rate, leading to cracks in the material.
Factors Influencing Corrosion: A Chemical Perspective
Several factors influence the rate and type of corrosion, all having a direct chemical basis:
- Material Composition: Different metals possess varying electrochemical potentials, impacting their susceptibility to corrosion.
- Environment: The presence of oxygen, water, and various ions in the surrounding environment significantly influences corrosion rates. For example, acidic environments accelerate corrosion.
- Temperature: Higher temperatures generally increase the rate of chemical reactions, including corrosion processes.
- pH: The acidity or alkalinity of the environment affects the corrosion rate.
- Presence of Inhibitors: Certain chemicals can slow down or prevent corrosion by interfering with the chemical reactions involved.
Conclusion: Corrosion – An Irrefutably Chemical Process
While the physical appearance of a corroded material might change significantly – its shape, texture, and even its mass – the underlying process is undeniably chemical. It is the alteration of the material's chemical composition through reactions with its surrounding environment that defines corrosion. The numerous types of corrosion and the various factors influencing it all point to its chemical nature. Understanding this fundamental aspect is crucial for developing effective corrosion prevention and control strategies across various sectors. From safeguarding infrastructure to protecting valuable assets, managing corrosion requires a robust grasp of the underlying chemical principles.
Latest Posts
Latest Posts
-
Which Of The Following Determines The Length Of The Base
Apr 06, 2025
-
Carboxylic Acids Can Be Made By The Hydrolysis Of Nitriles
Apr 06, 2025
-
Electrons Are Found In The Nucleus Of An Atom
Apr 06, 2025
-
Subshells In Order Of Increasing Energy
Apr 06, 2025
-
The Enlightenment And The Great Awakening Caused
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is Corrosion A Physical Or Chemical Property . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
