Is Oxidation Gaining Or Losing Electrons
Muz Play
Apr 05, 2025 · 5 min read
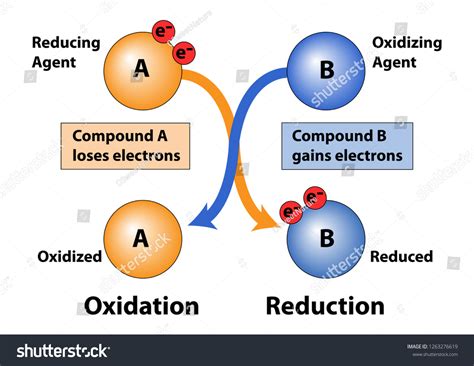
Table of Contents
Is Oxidation Gaining or Losing Electrons? Understanding Redox Reactions
The question of whether oxidation involves gaining or losing electrons is a fundamental concept in chemistry, often causing confusion for beginners. The simple answer is: oxidation is the loss of electrons. However, a deeper understanding requires exploring the broader context of redox reactions and the nuances surrounding electron transfer. This comprehensive guide will delve into the intricacies of oxidation and reduction, providing a clear and concise explanation backed by examples.
Understanding Oxidation and Reduction: The Core Concepts
Oxidation and reduction are always coupled processes; you can't have one without the other. This interconnectedness is why they are often referred to as redox reactions. To remember which is which, many students use the mnemonic devices:
- OIL RIG: Oxidation Is Losing, Reduction Is Gaining (electrons)
- LEO the lion says GER: Lose Electrons Oxidation, Gain Electrons Reduction
Let's break down each term individually:
Oxidation: The Loss of Electrons
Oxidation, at its core, is a chemical process where an atom, molecule, or ion loses one or more electrons. This loss results in an increase in the oxidation state of the species involved. The oxidation state, or oxidation number, represents the hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic. It's a crucial tool for tracking electron transfer in redox reactions.
Key characteristics of oxidation:
- Loss of electrons: This is the defining feature. The atom undergoing oxidation becomes more positively charged or less negatively charged.
- Increase in oxidation state: The oxidation number of the atom increases.
- Often involves the addition of oxygen or removal of hydrogen: While not always the case, these are common scenarios associated with oxidation.
Reduction: The Gain of Electrons
Reduction is the exact opposite of oxidation. It's a chemical process where an atom, molecule, or ion gains one or more electrons. This gain leads to a decrease in the oxidation state of the species.
Key characteristics of reduction:
- Gain of electrons: The atom undergoing reduction becomes more negatively charged or less positively charged.
- Decrease in oxidation state: The oxidation number of the atom decreases.
- Often involves the removal of oxygen or addition of hydrogen: Similar to oxidation, these are common but not exclusive characteristics.
Examples Illustrating Oxidation and Reduction
Let's examine some classic examples to solidify our understanding:
Example 1: The Reaction Between Iron and Oxygen
The rusting of iron is a quintessential example of a redox reaction. Iron (Fe) reacts with oxygen (O₂) in the presence of water to form iron(III) oxide (Fe₂O₃), commonly known as rust.
The reaction can be represented as:
4Fe(s) + 3O₂(g) → 2Fe₂O₃(s)
- Oxidation: Iron (Fe) loses electrons and its oxidation state increases from 0 to +3. The iron atoms are oxidized.
- Reduction: Oxygen (O₂) gains electrons and its oxidation state decreases from 0 to -2. The oxygen atoms are reduced.
Example 2: The Reaction Between Zinc and Copper(II) Sulfate
When a zinc strip is placed in a solution of copper(II) sulfate, a spontaneous redox reaction occurs. Zinc displaces copper from the solution, resulting in a coating of copper metal on the zinc strip and a change in the solution's color.
The reaction is:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
- Oxidation: Zinc (Zn) loses two electrons, increasing its oxidation state from 0 to +2. The zinc is oxidized.
- Reduction: Copper(II) ions (Cu²⁺) gain two electrons, decreasing their oxidation state from +2 to 0. The copper ions are reduced.
Example 3: Combustion Reactions
Combustion reactions, like burning wood or propane, are also redox reactions. The fuel (hydrocarbon) is oxidized, and oxygen is reduced.
For example, the combustion of methane (CH₄):
CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(g)
- Oxidation: Carbon in methane (oxidation state -4) loses electrons and increases its oxidation state to +4 in carbon dioxide. Methane is oxidized.
- Reduction: Oxygen (oxidation state 0) gains electrons and decreases its oxidation state to -2 in water and carbon dioxide. Oxygen is reduced.
Beyond Simple Electron Transfer: Oxidation States and Complex Reactions
While the simple loss and gain of electrons are central to oxidation and reduction, many reactions involve complex electron shifts and changes in oxidation states that aren't directly observable as a simple electron transfer.
For instance, consider the oxidation of ethanol to ethanal:
CH₃CH₂OH → CH₃CHO + 2H⁺ + 2e⁻
Here, two electrons are lost, and hydrogen ions are released. The carbon atom bonded to the hydroxyl group (-OH) undergoes oxidation as its oxidation state increases. While not a direct transfer to another atom, the overall process is still classified as oxidation.
Similarly, the reduction of a carbonyl group (C=O) to a hydroxyl group (-OH) isn't a simple addition of an electron. Instead, the overall change in oxidation state indicates a reduction.
Identifying Oxidation and Reduction in Complex Reactions
To confidently identify oxidation and reduction in more complex reactions, you'll need to:
- Assign oxidation states: Systematically determine the oxidation state of each atom in the reactants and products.
- Track changes in oxidation state: Observe the change in oxidation state for each atom. An increase indicates oxidation, and a decrease indicates reduction.
- Balance the redox reaction: Ensure that the number of electrons lost in oxidation equals the number of electrons gained in reduction.
Applications of Redox Reactions
Redox reactions are ubiquitous in nature and have numerous applications in various fields:
- Biological systems: Respiration, photosynthesis, and many other metabolic processes rely on redox reactions for energy transfer.
- Industrial processes: Extraction of metals from their ores, production of chemicals, and electroplating utilize redox reactions extensively.
- Batteries: The operation of batteries depends on redox reactions that generate an electric current.
- Corrosion: Rusting, tarnishing, and other forms of corrosion are redox reactions leading to material degradation.
Conclusion
In summary, oxidation is the loss of electrons, and reduction is the gain of electrons. These two processes are always coupled in redox reactions. Understanding oxidation and reduction is crucial in various fields, from biology and chemistry to materials science and engineering. By grasping the fundamental concepts and employing systematic approaches to analyze reactions, you can confidently identify oxidation and reduction processes in even the most complex chemical transformations. Remembering OIL RIG or LEO GER can serve as helpful mnemonics, but understanding the underlying principles of electron transfer and oxidation states is key to mastering this essential chemical concept.
Latest Posts
Latest Posts
-
How Many Electrons Are In A Single Bond
Apr 06, 2025
-
Is Plant Cells Prokaryotic Or Eukaryotic
Apr 06, 2025
-
All Viruses Leave A Cell By Exocytosis
Apr 06, 2025
-
When Two Monosaccharides Undergo A Dehydration Synthesis
Apr 06, 2025
-
Area Enclosed By A Parametric Curve
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is Oxidation Gaining Or Losing Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
