Salt Bridge Function In Galvanic Cell
Muz Play
Apr 01, 2025 · 6 min read
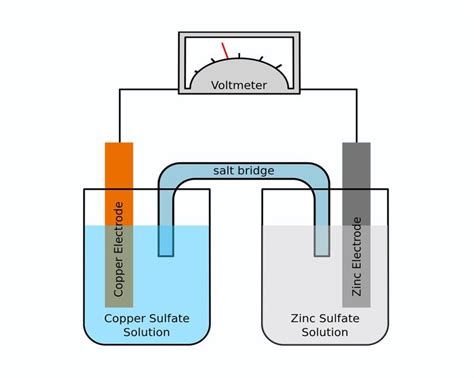
Table of Contents
- Salt Bridge Function In Galvanic Cell
- Table of Contents
- Salt Bridge: The Unsung Hero of Galvanic Cells
- Understanding the Galvanic Cell: A Quick Recap
- The Crucial Role of the Salt Bridge
- 1. Maintaining Electrical Neutrality
- 2. Completing the Electrical Circuit
- 3. Preventing Mixing of Electrolyte Solutions
- Consequences of Absence of Salt Bridge
- Choosing the Right Salt Bridge Electrolyte: Inertness is Key
- Alternative Methods to Salt Bridge: A Look Beyond the Traditional
- Conclusion: The Salt Bridge's Indispensable Role
- Latest Posts
- Latest Posts
- Related Post
Salt Bridge: The Unsung Hero of Galvanic Cells
The galvanic cell, also known as a voltaic cell, is a marvel of electrochemical engineering, silently powering countless devices in our daily lives. From the humble battery in your remote to the sophisticated systems in electric vehicles, these cells harness the power of spontaneous redox reactions to generate electrical energy. While the electrodes – the sites of oxidation and reduction – often take center stage, a less glamorous but equally crucial component plays a vital role: the salt bridge. This article delves deep into the function of the salt bridge in galvanic cells, exploring its mechanisms, importance, and the consequences of its absence.
Understanding the Galvanic Cell: A Quick Recap
Before we dive into the intricacies of the salt bridge, let's briefly revisit the fundamental principles of a galvanic cell. A galvanic cell consists of two half-cells: an anode (where oxidation occurs) and a cathode (where reduction occurs). These half-cells are typically connected by an external circuit allowing electron flow and a salt bridge completing the internal circuit.
- Anode: The electrode where oxidation occurs. Electrons are released as a species loses electrons. This electrode is negatively charged.
- Cathode: The electrode where reduction occurs. Electrons are consumed as a species gains electrons. This electrode is positively charged.
- Electrolyte Solutions: Each half-cell contains an electrolyte solution containing ions that participate in the redox reaction.
- External Circuit: The wire connecting the anode and cathode allows the flow of electrons, generating an electric current.
The overall cell potential (voltage) is determined by the difference in potential between the two half-cells, which is a function of the reduction potentials of the individual half-reactions. This potential difference drives the flow of electrons through the external circuit.
The Crucial Role of the Salt Bridge
The salt bridge is a crucial component that connects the two half-cells internally, completing the electrical circuit and preventing the buildup of charge that would otherwise halt the redox reaction. Its primary function is to maintain electrical neutrality within each half-cell.
1. Maintaining Electrical Neutrality
As the redox reaction proceeds, electrons flow from the anode (oxidation) to the cathode (reduction) through the external circuit. This electron flow would lead to a buildup of negative charge in the anode compartment and positive charge in the cathode compartment. This charge imbalance would create an opposing electric field, counteracting the potential difference and ultimately stopping the reaction.
The salt bridge prevents this charge buildup by allowing the flow of ions between the two half-cells. It contains a solution of an inert electrolyte, typically a salt with highly mobile ions (e.g., potassium nitrate, potassium chloride). These ions migrate to neutralize the charge imbalance:
- Anions (negatively charged ions) migrate towards the anode compartment: These anions neutralize the excess positive charge accumulating due to the oxidation reaction at the anode.
- Cations (positively charged ions) migrate towards the cathode compartment: These cations neutralize the excess negative charge accumulating due to the reduction reaction at the cathode.
This ion migration maintains electrical neutrality in both half-cells, allowing the redox reaction to continue smoothly and generate a sustained electric current.
2. Completing the Electrical Circuit
While the external circuit facilitates the flow of electrons, the internal circuit – completed by the salt bridge – facilitates the flow of ions. The flow of ions through the salt bridge completes the circuit, ensuring a continuous flow of electrons and ions, which is essential for the galvanic cell to function effectively.
3. Preventing Mixing of Electrolyte Solutions
Another crucial function of the salt bridge is to prevent the direct mixing of the electrolyte solutions in the two half-cells. If the solutions were to mix, it could lead to unwanted side reactions, affecting the overall cell potential and reducing the efficiency of the galvanic cell. The salt bridge acts as a barrier, allowing ion flow but preventing bulk mixing of the solutions.
Consequences of Absence of Salt Bridge
The absence of a salt bridge would have catastrophic consequences for the operation of a galvanic cell:
- Immediate Cessation of Current Flow: Without a salt bridge, the charge imbalance would quickly build up in the half-cells, creating a strong opposing electric field that would halt the flow of electrons and stop the redox reaction almost immediately.
- Reduced Cell Potential: The buildup of charge would significantly reduce the potential difference between the two half-cells, leading to a decrease in the cell potential and thus a weaker current.
- Irreversible Reactions: Without the neutralizing effect of the salt bridge, the cell's conditions will favor undesirable side reactions.
- Electrode Polarization: The anode and cathode would become polarized, meaning their surfaces would accumulate an excess charge that significantly alters the kinetics of the electrode reactions.
Choosing the Right Salt Bridge Electrolyte: Inertness is Key
The choice of electrolyte in the salt bridge is critical. The electrolyte must be chosen carefully to ensure it does not react with any of the components in the half-cells. Inert electrolytes, such as potassium nitrate (KNO₃) and potassium chloride (KCl), are commonly used because their ions are relatively unreactive and do not interfere with the primary redox reaction.
The selection criteria for the electrolyte also involve considerations such as solubility, mobility (high ionic mobility is preferable), and cost. The ions should be able to move freely through the salt bridge, facilitating efficient charge neutralization.
Alternative Methods to Salt Bridge: A Look Beyond the Traditional
While the salt bridge is the most common method for completing the internal circuit in galvanic cells, other methods can be used in certain scenarios. These alternatives often offer advantages in specific applications, such as miniaturized devices or those requiring high conductivity.
- Porous Membrane: A porous membrane can replace the salt bridge, allowing ion flow while preventing bulk mixing of the electrolytes. These membranes are often used in specialized galvanic cells or fuel cells.
- Direct Contact: In some cases, particularly in fuel cells, the two half-cells might be in direct contact, allowing for ion flow without a separate salt bridge or membrane. However, maintaining separation of reacting species remains a challenge.
Conclusion: The Salt Bridge's Indispensable Role
The salt bridge, often overlooked, is a critical component of any galvanic cell. Its function in maintaining electrical neutrality, completing the circuit, and preventing mixing of electrolytes is crucial for the cell's proper operation and efficient energy generation. Without the salt bridge, the galvanic cell would quickly cease to function, highlighting its indispensable role in electrochemical energy conversion. The understanding of salt bridge function and its implications extends beyond simple galvanic cell applications, impacting the design and optimization of more complex electrochemical systems, including batteries, fuel cells, and various types of sensors. Further research into the properties and advancements of salt bridge materials continues to drive improvements in the efficiency and longevity of these vital electrochemical devices. The seemingly simple salt bridge, therefore, holds a key position in the development of more sustainable and efficient energy technologies.
Latest Posts
Latest Posts
-
What Is An Inner Shell Electron
Apr 04, 2025
-
The Ul For Folate Refers Only To Folic Acid Because
Apr 04, 2025
-
Artists Of The Early 20th Century
Apr 04, 2025
-
How To Find The Resistance Of A Circuit
Apr 04, 2025
-
Identify The Features Associated With Waxes
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Salt Bridge Function In Galvanic Cell . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
