The Chemistry Of Living Organisms Is Called Chemistry
Muz Play
Apr 03, 2025 · 6 min read
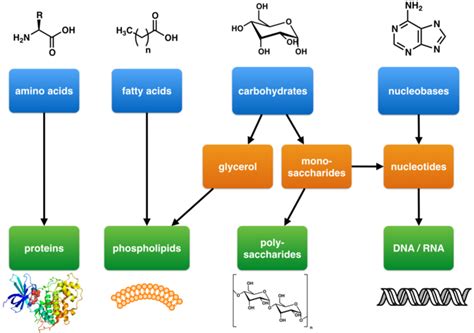
Table of Contents
The Chemistry of Living Organisms: A Deep Dive into Biochemistry
The chemistry of living organisms is called biochemistry. It's a fascinating and complex field that explores the intricate chemical processes occurring within and relating to living organisms. From the smallest bacteria to the largest whales, life is fundamentally driven by chemical reactions. Understanding these reactions is key to understanding life itself, and biochemistry provides the framework for doing so. This comprehensive article delves into the core principles of biochemistry, exploring its key components and their vital roles in maintaining life.
The Building Blocks of Life: Biomolecules
Life is constructed from a remarkable array of molecules, collectively known as biomolecules. These molecules, although diverse in structure and function, are largely composed of just six elements: carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S). The unique properties of carbon, particularly its ability to form four stable covalent bonds, are central to the formation of the complex organic molecules that characterize living systems.
1. Carbohydrates: The Energy Source
Carbohydrates are the primary source of energy for living organisms. They are composed of carbon, hydrogen, and oxygen, often in a ratio of 1:2:1 (hence the name "carbohydrate"). The simplest carbohydrates are monosaccharides, such as glucose, fructose, and galactose. These monosaccharides can link together to form disaccharides (like sucrose, lactose, and maltose) and polysaccharides (like starch, glycogen, and cellulose).
- Monosaccharides: These are the building blocks of carbohydrates and serve as immediate energy sources. Glucose, in particular, is crucial for cellular respiration.
- Disaccharides: Formed by the dehydration synthesis of two monosaccharides, disaccharides are also energy sources, though they must first be broken down into monosaccharides.
- Polysaccharides: These are complex carbohydrates composed of long chains of monosaccharides. Starch is the storage form of glucose in plants, glycogen in animals, and cellulose provides structural support in plant cell walls.
2. Lipids: The Structural and Energy Storage Molecules
Lipids are a diverse group of hydrophobic (water-insoluble) biomolecules that play crucial roles in energy storage, cell membrane structure, and hormone signaling. They include fats, oils, phospholipids, and steroids.
- Fats and Oils: These are triglycerides, composed of a glycerol molecule and three fatty acids. Saturated fatty acids have no double bonds between carbon atoms, while unsaturated fatty acids contain one or more double bonds. Saturated fats are typically solid at room temperature (e.g., butter), while unsaturated fats are liquid (e.g., vegetable oil).
- Phospholipids: These are crucial components of cell membranes. They have a hydrophilic (water-loving) head and two hydrophobic tails, forming a bilayer that separates the cell's interior from its surroundings.
- Steroids: These are lipids with a characteristic four-ring structure. Cholesterol, a crucial component of cell membranes, is a steroid, as are various hormones like testosterone and estrogen.
3. Proteins: The Workhorses of the Cell
Proteins are the most diverse and versatile biomolecules. They are composed of chains of amino acids linked together by peptide bonds. The sequence of amino acids determines a protein's unique three-dimensional structure, which, in turn, dictates its function.
- Amino Acids: The building blocks of proteins, there are 20 common amino acids, each with a unique side chain that contributes to the protein's overall properties.
- Protein Structure: Proteins have four levels of structure: primary (amino acid sequence), secondary (alpha-helices and beta-sheets), tertiary (three-dimensional folding), and quaternary (arrangement of multiple polypeptide chains).
- Protein Function: Proteins perform a vast array of functions, including catalysis (enzymes), transport (hemoglobin), structural support (collagen), defense (antibodies), and movement (actin and myosin).
4. Nucleic Acids: The Information Carriers
Nucleic acids, DNA and RNA, are the molecules that carry genetic information. They are composed of long chains of nucleotides.
- Nucleotides: The building blocks of nucleic acids, each nucleotide consists of a sugar (deoxyribose in DNA, ribose in RNA), a phosphate group, and a nitrogenous base (adenine, guanine, cytosine, and thymine in DNA; adenine, guanine, cytosine, and uracil in RNA).
- DNA (Deoxyribonucleic Acid): The primary genetic material, DNA stores the instructions for building and maintaining an organism. Its double-helix structure allows for accurate replication and transmission of genetic information.
- RNA (Ribonucleic Acid): Plays various roles in gene expression, including mRNA (messenger RNA), which carries genetic information from DNA to ribosomes, tRNA (transfer RNA), which carries amino acids to ribosomes for protein synthesis, and rRNA (ribosomal RNA), which is a structural component of ribosomes.
Metabolic Processes: The Chemical Engine of Life
Metabolism encompasses all the chemical reactions occurring within a living organism. These reactions are highly organized and regulated, allowing for the efficient acquisition and utilization of energy and the synthesis and breakdown of biomolecules. Metabolic pathways can be categorized into two main types:
1. Catabolism: Breaking Down Molecules
Catabolism involves the breakdown of complex molecules into simpler ones, releasing energy in the process. Cellular respiration, the process by which cells extract energy from glucose, is a prime example of catabolism. Other catabolic pathways break down proteins into amino acids, lipids into fatty acids and glycerol, and nucleic acids into nucleotides. This released energy is often stored in the form of ATP (adenosine triphosphate), the cell's primary energy currency.
2. Anabolism: Building Up Molecules
Anabolism involves the synthesis of complex molecules from simpler ones, requiring energy input. Examples of anabolic processes include protein synthesis, DNA replication, and the synthesis of carbohydrates and lipids. These processes are essential for growth, repair, and maintenance of the organism.
Enzymes: The Catalysts of Life
Enzymes are biological catalysts, primarily proteins, that accelerate the rate of chemical reactions within cells. They do this by lowering the activation energy required for a reaction to occur. Enzymes are highly specific, each catalyzing a particular reaction or a group of closely related reactions. Their activity is often regulated by factors such as temperature, pH, and the presence of inhibitors or activators. The remarkable efficiency and specificity of enzymes are critical for the orderly and efficient functioning of metabolic pathways.
The Importance of Water in Biochemistry
Water plays a central role in biochemistry. Its unique properties, stemming from its polar nature and ability to form hydrogen bonds, are essential for life. Water acts as a solvent, facilitating chemical reactions; it participates in many metabolic reactions; it plays a crucial role in maintaining cell structure and function; and it helps regulate temperature. The cohesive and adhesive properties of water are vital for the transport of substances within organisms.
The Future of Biochemistry
Biochemistry continues to be a dynamic and rapidly evolving field. Advances in genomics, proteomics, and metabolomics are providing unprecedented insights into the complexity of living systems. These advances are leading to breakthroughs in areas such as drug discovery, disease diagnosis, and the development of new biotechnologies. The understanding of biochemical processes is crucial for addressing global challenges such as food security, environmental sustainability, and human health.
Keywords: Biochemistry, biomolecules, carbohydrates, lipids, proteins, nucleic acids, metabolism, catabolism, anabolism, enzymes, water, DNA, RNA, cellular respiration, ATP, amino acids, nucleotides, molecular biology, genetics, cell biology, organic chemistry
This article provides a broad overview of biochemistry. Each of the topics discussed above could be the subject of an entire book. The incredible intricacy and beauty of the chemistry of life continue to inspire research and discovery, promising further exciting advancements in our understanding of the living world.
Latest Posts
Latest Posts
-
M 1 And M 2 Peaks In Mass Spectrometry
Apr 04, 2025
-
The Storage Capacity Of Long Term Memory Is
Apr 04, 2025
-
The Hormone Of Hypersecretion Is
Apr 04, 2025
-
How Many Electrons Can The Fourth Energy Level Hold
Apr 04, 2025
-
What Did The Angel Gabriel Say To Muhammad
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Chemistry Of Living Organisms Is Called Chemistry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
