The Coefficients In A Chemical Equation Represent The
Muz Play
Apr 03, 2025 · 6 min read
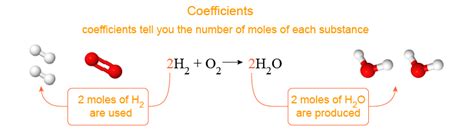
Table of Contents
The Coefficients in a Chemical Equation Represent the Moles of Reactants and Products
Chemical equations are the shorthand language of chemistry, providing a concise way to describe chemical reactions. They show us what substances react (reactants) and what substances are formed (products). But the true power of a chemical equation lies not just in identifying the reactants and products, but also in the coefficients that precede each chemical formula. These coefficients are crucial because they represent the relative number of moles of each substance involved in the reaction. Understanding this fundamental concept is key to mastering stoichiometry, the quantitative relationship between reactants and products in a chemical reaction.
Understanding Moles: The Foundation of Stoichiometry
Before delving into the significance of coefficients, let's solidify our understanding of the mole. A mole (mol) is the cornerstone of chemical calculations, representing Avogadro's number (6.022 x 10²³) of particles. These particles can be atoms, molecules, ions, or any other specified entity. The mole provides a bridge between the macroscopic world (grams) and the microscopic world (atoms and molecules). The molar mass of a substance, expressed in grams per mole (g/mol), is the mass of one mole of that substance.
For instance, the molar mass of water (H₂O) is approximately 18 g/mol. This means that one mole of water contains 6.022 x 10²³ water molecules and weighs 18 grams.
The Role of Coefficients: Balancing Equations and Mole Ratios
A balanced chemical equation is crucial because it adheres to the law of conservation of mass. This law dictates that matter cannot be created or destroyed in a chemical reaction; the total mass of reactants must equal the total mass of products. Coefficients are the tools we use to achieve this balance.
Consider the following unbalanced equation for the combustion of methane:
CH₄ + O₂ → CO₂ + H₂O
This equation is unbalanced because the number of atoms of each element is not equal on both sides. To balance it, we need to adjust the coefficients:
CH₄ + 2O₂ → CO₂ + 2H₂O
Now the equation is balanced. We have one carbon atom, four hydrogen atoms, and four oxygen atoms on both the reactant and product sides. The coefficients tell us the relative number of moles of each substance involved. This balanced equation tells us that:
- One mole of methane (CH₄) reacts with two moles of oxygen (O₂)
- To produce one mole of carbon dioxide (CO₂) and two moles of water (H₂O)
This ratio of moles is incredibly important for stoichiometric calculations.
Applying Coefficients in Stoichiometric Calculations
The coefficients in a balanced chemical equation provide the mole ratios necessary for solving various stoichiometry problems. These problems often involve determining the amount of product formed from a given amount of reactant (or vice versa), limiting reactants, and percent yield.
Example 1: Determining the moles of product
Let's say we react 5 moles of methane (CH₄) with excess oxygen. How many moles of carbon dioxide (CO₂) will be produced?
Using the balanced equation (CH₄ + 2O₂ → CO₂ + 2H₂O), we can establish the mole ratio between methane and carbon dioxide as 1:1. Therefore, if we react 5 moles of methane, we will produce 5 moles of carbon dioxide.
Example 2: Determining the mass of product
Let's say we react 16 grams of methane (CH₄) with excess oxygen. How many grams of water (H₂O) will be produced?
-
Convert grams of methane to moles: The molar mass of CH₄ is approximately 16 g/mol. Therefore, 16 g of CH₄ is equal to 1 mole.
-
Use the mole ratio: From the balanced equation, the mole ratio of CH₄ to H₂O is 1:2. This means that 1 mole of CH₄ produces 2 moles of H₂O.
-
Convert moles of water to grams: The molar mass of H₂O is approximately 18 g/mol. Therefore, 2 moles of H₂O is equal to 36 grams.
Therefore, reacting 16 grams of methane with excess oxygen will produce 36 grams of water.
Example 3: Limiting Reactants
Let's consider a reaction where we have a limited amount of both reactants. For instance, if we react 2 moles of methane with 3 moles of oxygen, which reactant is limiting, and how many moles of carbon dioxide will be produced?
-
Determine the mole ratio: From the balanced equation, the mole ratio of CH₄ to O₂ is 1:2. This means that 1 mole of CH₄ requires 2 moles of O₂ for complete reaction.
-
Identify the limiting reactant: With 2 moles of CH₄, we would need 4 moles of O₂ for complete reaction. However, we only have 3 moles of O₂. Therefore, oxygen is the limiting reactant.
-
Calculate moles of product: The mole ratio of O₂ to CO₂ is 2:1. With 3 moles of O₂, we can produce 1.5 moles of CO₂.
Therefore, only 1.5 moles of carbon dioxide will be produced.
Beyond Simple Mole Ratios: More Complex Reactions
While the examples above focus on relatively simple reactions, the principle remains the same for more complex reactions involving multiple reactants and products. The coefficients always represent the relative number of moles. For instance, consider the reaction:
2Fe₂O₃ + 3C → 4Fe + 3CO₂
This balanced equation tells us:
- 2 moles of iron(III) oxide (Fe₂O₃) react with 3 moles of carbon (C)
- To produce 4 moles of iron (Fe) and 3 moles of carbon dioxide (CO₂)
The coefficients remain fundamental in calculating the amount of reactants needed or the amount of products formed. The mole ratios derived from these coefficients are the key to performing accurate stoichiometric calculations.
Practical Applications and Real-World Significance
The understanding and application of coefficients in chemical equations have far-reaching consequences across various scientific and industrial fields. Some key applications include:
-
Industrial Chemical Processes: The efficient production of chemicals relies heavily on precise stoichiometric calculations to optimize the use of reactants and maximize product yield. This is crucial in industries like fertilizers, pharmaceuticals, and plastics.
-
Environmental Monitoring: Understanding the stoichiometry of reactions is essential for analyzing environmental pollution and designing effective remediation strategies.
-
Medical Applications: In medicine, stoichiometry plays a critical role in drug dosage calculations, ensuring patients receive the correct amount of medication.
-
Analytical Chemistry: Quantitative analysis of chemical samples relies on the precise determination of the amounts of reactants and products involved in reactions.
Conclusion: Coefficients – The Key to Understanding Chemical Reactions
In conclusion, the coefficients in a balanced chemical equation are far more than just numbers; they represent the relative number of moles of each substance involved in the reaction. They are fundamental to understanding and quantifying chemical reactions. By mastering the interpretation and application of these coefficients, we gain the ability to perform stoichiometric calculations, predict reaction outcomes, and solve a wide range of problems in chemistry and related fields. A deep understanding of this concept is essential for anyone studying or working with chemistry. From simple laboratory experiments to large-scale industrial processes, the coefficients in a chemical equation are the key to unlocking a precise and quantitative understanding of the chemical world. Without them, our understanding of chemical reactions would be incomplete and largely qualitative. Therefore, understanding and applying the principles of stoichiometry, rooted in the meaning of coefficients, remains crucial for continued advancement in various scientific disciplines.
Latest Posts
Latest Posts
-
Maslows Hierarchy Of Needs For Kids
Apr 04, 2025
-
How Many Electrons In A Covalent Bond
Apr 04, 2025
-
A Stick Model Of A Water Molecule
Apr 04, 2025
-
Critical Value Of Wilcoxon Signed Rank Test
Apr 04, 2025
-
What Are The 3 Properties Of Ionic Compounds
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Coefficients In A Chemical Equation Represent The . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
