The Nucleus Of An Atom Consists Of
Muz Play
Apr 04, 2025 · 7 min read
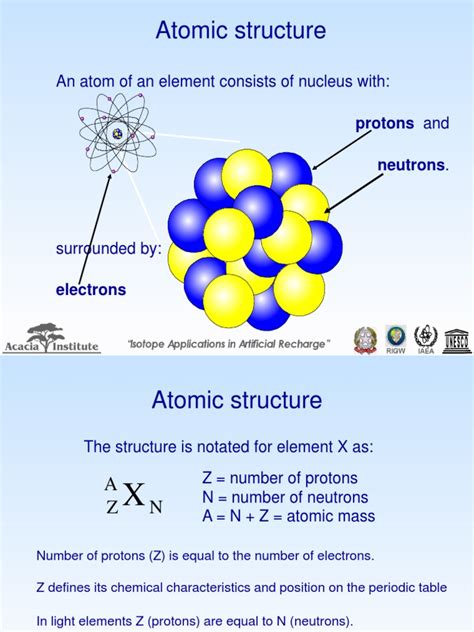
Table of Contents
The Nucleus of an Atom: A Deep Dive into Protons, Neutrons, and the Strong Force
The atom, the fundamental building block of matter, is a fascinating microcosm of physics. While often depicted as a simple solar system model with electrons orbiting a central nucleus, the reality is far more intricate and captivating. This article delves into the heart of the atom – its nucleus – exploring its composition, properties, and the fundamental forces governing its existence. We'll uncover the roles of protons and neutrons, the strong nuclear force, nuclear stability, isotopes, and the implications for various scientific fields.
The Composition of the Atomic Nucleus: Protons and Neutrons
The atomic nucleus is a densely packed region residing at the center of the atom, comprising two primary types of particles: protons and neutrons. These particles, collectively known as nucleons, are bound together by the incredibly powerful strong nuclear force.
Protons: The Positively Charged Core
Protons carry a positive electrical charge, equal in magnitude to the negative charge of an electron. The number of protons in an atom's nucleus defines its atomic number, which uniquely identifies the element. For instance, hydrogen (H) has one proton (atomic number 1), helium (He) has two (atomic number 2), and uranium (U) has 92 (atomic number 92). This proton count dictates the atom's chemical properties and its place on the periodic table.
Neutrons: The Neutral Stabilizers
Neutrons, as their name suggests, carry no electrical charge. They contribute significantly to the nucleus's mass but don't directly participate in chemical reactions. The number of neutrons in a nucleus, along with the number of protons, determines the mass number of the atom. The mass number is approximately the total mass of the atom in atomic mass units (amu), as the mass of electrons is negligible in comparison.
Isotopes: Variations in Neutron Count
Atoms of the same element can have varying numbers of neutrons. These variations are called isotopes. Isotopes of an element have the same atomic number (same number of protons) but different mass numbers (different numbers of neutrons). For example, carbon-12 (¹²C) has 6 protons and 6 neutrons, while carbon-14 (¹⁴C) has 6 protons and 8 neutrons. While chemically similar, isotopes can have significantly different physical properties and applications. Carbon-14, for instance, is radioactive and used in radiocarbon dating.
The Strong Nuclear Force: The Glue That Holds the Nucleus Together
The protons within the nucleus are positively charged and would, according to Coulomb's law, repel each other with immense force, causing the nucleus to fly apart. However, the nucleus remains remarkably stable, thanks to the strong nuclear force. This is one of the four fundamental forces in nature, far stronger than the electromagnetic force but acting over an extremely short range—only within the nucleus itself.
Understanding the Strong Force's Strength and Range
The strong force is approximately 100 times stronger than the electromagnetic force. However, its influence diminishes rapidly with distance, making it essentially negligible beyond the confines of the nucleus. This short-range nature explains why the electromagnetic repulsion between protons doesn't completely overcome the strong force's attraction at the nuclear scale. The precise mechanism of the strong force is governed by quantum chromodynamics (QCD), a complex area of particle physics.
The Role of Gluons and Quarks
The strong force isn't a simple attraction between protons and neutrons; it's mediated by particles called gluons. Protons and neutrons, in turn, are composed of even smaller particles called quarks, which are bound together by gluons. This intricate interplay of quarks and gluons is responsible for the strong force's binding effect within the nucleus.
Nuclear Stability and Radioactive Decay
Not all combinations of protons and neutrons result in stable nuclei. The stability of a nucleus depends on the balance between the strong nuclear force, which attracts nucleons, and the electromagnetic force, which repels protons. Nuclei with a certain optimal neutron-to-proton ratio tend to be more stable.
Radioactive Decay: Unstable Nuclei's Transformation
Nuclei with an imbalanced neutron-to-proton ratio are unstable and undergo radioactive decay. This involves the spontaneous emission of particles (alpha, beta, or gamma radiation) or energy to achieve a more stable configuration. Radioactive decay is a crucial process in various scientific applications, including medical imaging, cancer treatment, and geological dating.
Types of Radioactive Decay: Alpha, Beta, and Gamma
- Alpha decay: involves the emission of an alpha particle (two protons and two neutrons), effectively reducing the atomic number by 2 and the mass number by 4.
- Beta decay: involves the conversion of a neutron into a proton (or vice versa), accompanied by the emission of an electron (beta minus decay) or a positron (beta plus decay). Beta decay changes the atomic number but not the mass number significantly.
- Gamma decay: involves the emission of gamma rays, which are high-energy photons. Gamma decay doesn't change the atomic number or mass number but releases excess energy from an excited nucleus.
Nuclear Fission and Fusion: Harnessing Nuclear Energy
The immense energy stored within the atomic nucleus can be harnessed through two primary processes: nuclear fission and nuclear fusion.
Nuclear Fission: Splitting the Atom
Nuclear fission involves splitting a heavy, unstable nucleus (such as uranium or plutonium) into two smaller, more stable nuclei. This process releases a tremendous amount of energy, as some of the mass is converted into energy according to Einstein's famous equation, E=mc². Nuclear fission is the basis for nuclear power plants and nuclear weapons.
Nuclear Fusion: Combining Nuclei
Nuclear fusion, in contrast, involves combining two light nuclei (such as isotopes of hydrogen) into a heavier nucleus. This process also releases a vast amount of energy, even more than fission, but requires extremely high temperatures and pressures to overcome the electrostatic repulsion between the positively charged nuclei. Nuclear fusion is the process powering the sun and other stars, and is the subject of intense research for future clean energy sources.
Applications of Nuclear Science
The study of the atomic nucleus and its properties has far-reaching applications across diverse scientific and technological fields:
Nuclear Medicine: Diagnosis and Treatment
Radioactive isotopes are extensively used in nuclear medicine for both diagnostic and therapeutic purposes. Radioactive tracers are injected into the body, allowing doctors to visualize organs and tissues, detect tumors, and monitor physiological processes. Radiotherapy uses radioactive materials to target and destroy cancerous cells.
Archaeology and Geology: Dating Techniques
Radioactive decay is crucial for radiometric dating, enabling scientists to determine the age of artifacts, fossils, and geological formations. Carbon-14 dating is a widely used technique for dating organic materials, while other isotopes (like uranium and potassium) are used for dating older rocks and minerals.
Nuclear Power: Generating Electricity
Nuclear fission is employed in nuclear power plants to generate electricity. The heat generated from fission is used to boil water, producing steam that drives turbines to generate electricity. While nuclear power provides a carbon-free energy source, it also involves risks related to radioactive waste disposal and potential accidents.
Material Science and Engineering: Modifying Properties
Nuclear techniques are used in material science to modify the properties of materials. Ion implantation, for instance, involves bombarding materials with ions to alter their electrical, mechanical, or optical properties.
Conclusion: The Ongoing Exploration of the Atomic Nucleus
The atomic nucleus, with its intricate interplay of protons, neutrons, and the strong force, remains a subject of intense scientific inquiry. Understanding the nucleus's structure and behavior is crucial for advancing numerous fields, from medicine and energy production to materials science and archaeology. Ongoing research continues to reveal new insights into this fundamental component of matter, promising further technological advancements and a deeper understanding of the universe. The ongoing quest to unravel the mysteries of the nucleus ensures its continued importance in shaping our future.
Latest Posts
Latest Posts
-
What Is The Difference Between Religion And Culture
Apr 04, 2025
-
What Are Indicators Of Chemical Change
Apr 04, 2025
-
How To Find Empirical Formula From Grams
Apr 04, 2025
-
How To Make A Thesis Statement For A Narrative Essay
Apr 04, 2025
-
Describe Two Variables That Affect The Rate Of Diffusion
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about The Nucleus Of An Atom Consists Of . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
