Three Factors That Affect Enzyme Activity
Muz Play
Apr 01, 2025 · 6 min read
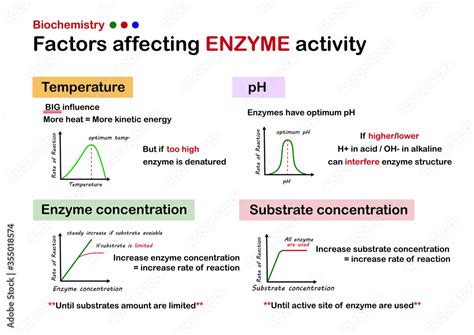
Table of Contents
Three Key Factors Affecting Enzyme Activity: A Deep Dive
Enzymes are the workhorses of life, biological catalysts that accelerate the rate of virtually every chemical reaction within living organisms. Their remarkable efficiency and specificity are crucial for maintaining cellular homeostasis and enabling life's complex processes. However, the activity of these crucial molecules isn't constant; it's delicately balanced and influenced by a multitude of factors. Understanding these influences is critical to comprehending biological processes, developing new medical treatments, and even optimizing industrial applications of enzymes. This article delves into three key factors that profoundly affect enzyme activity: temperature, pH, and substrate concentration.
1. Temperature: The Goldilocks Principle of Enzyme Activity
Temperature exerts a significant impact on enzyme activity, adhering to a principle often described as "Goldilocks": it needs to be "just right." Enzymes are proteins, and their three-dimensional structure is critical to their function. This intricate structure, with precisely positioned active sites, is maintained by a complex network of weak bonds (hydrogen bonds, van der Waals forces).
The Optimal Temperature Range
At low temperatures, enzyme activity is low. The kinetic energy of molecules is reduced, meaning that enzyme-substrate collisions are less frequent, slowing down the reaction rate. The enzyme molecules themselves move sluggishly, reducing their effectiveness in binding to substrates. Think of it like trying to play a fast-paced game with your hands frozen stiff – it's just not efficient.
As temperature increases, enzyme activity generally increases. Higher temperatures lead to more frequent and energetic enzyme-substrate collisions, resulting in an accelerated reaction rate. The molecules possess more kinetic energy, facilitating faster binding and catalysis. This increase continues until the enzyme reaches its optimum temperature.
Denaturation: The Point of No Return
However, exceeding the optimum temperature leads to a drastic decline in enzyme activity. This is because high temperatures disrupt the weak bonds maintaining the enzyme's tertiary structure. The enzyme undergoes denaturation, losing its specific three-dimensional shape and, consequently, its active site. This renders the enzyme non-functional, irreversibly damaging its catalytic ability. Imagine crumpling a perfectly folded origami crane – you can't easily refold it to its original pristine state.
Different enzymes have different optimum temperatures, reflecting their natural environments. For instance, enzymes found in thermophilic bacteria, which thrive in hot springs, have much higher optimum temperatures compared to enzymes found in human cells. This adaptation allows them to function effectively in their extreme environments.
Practical Implications of Temperature Sensitivity
The temperature sensitivity of enzymes has widespread implications. In industrial processes utilizing enzymes, such as in brewing or laundry detergents, maintaining the optimal temperature is crucial for efficiency. In medicine, understanding the temperature dependence of enzymes is vital in the design and development of therapies, particularly those involving enzyme-based drugs. Furthermore, this knowledge is crucial in understanding how environmental changes, such as global warming, might affect ecosystems by altering enzyme activity in various organisms.
2. pH: Maintaining the Right Balance for Enzyme Function
pH, a measure of the acidity or alkalinity of a solution, significantly impacts enzyme activity. Enzymes possess ionizable amino acid side chains in their active sites, and the charge of these groups is heavily influenced by the surrounding pH. This charge influences the enzyme's ability to bind to its substrate and catalyze the reaction.
The Importance of the Active Site's Charge
The optimal pH for an enzyme is the pH at which the enzyme exhibits its maximal activity. At this pH, the amino acid side chains in the active site possess the appropriate charge to optimally bind the substrate. Deviations from this optimum pH can alter the charge distribution, affecting substrate binding and catalytic efficiency.
The Impact of Extreme pH Values
Both high and low pH values can cause enzyme denaturation. Extreme pH levels disrupt the balance of charges within the enzyme molecule, leading to the disruption of non-covalent interactions (like hydrogen bonds and ionic bonds) that maintain its three-dimensional structure. This results in a conformational change, affecting the active site and rendering the enzyme inactive. This denaturation is often irreversible.
Just like temperature, different enzymes have different optimal pH values, reflecting their specific physiological roles. For example, pepsin, a digestive enzyme found in the stomach, functions optimally at a highly acidic pH (around 2), whereas trypsin, a digestive enzyme found in the small intestine, works best at a more neutral pH (around 8). These differing pH optima reflect the distinct environments in which these enzymes operate.
Practical Implications of pH Sensitivity
The pH sensitivity of enzymes has crucial implications in various fields. In industrial applications, the pH of the reaction medium needs careful control to maintain optimal enzyme activity. In medicine, maintaining the proper pH balance in the body is crucial for numerous physiological processes, as many enzyme-driven reactions depend on it. Disruptions in pH, such as acidosis or alkalosis, can have severe consequences by affecting the activity of vital enzymes. Therefore, understanding and managing pH is essential for maintaining health and achieving optimal performance in many biological and industrial contexts.
3. Substrate Concentration: The Law of Diminishing Returns
Substrate concentration, the amount of substrate available for the enzyme to act upon, directly influences the rate of enzyme-catalyzed reactions. However, this relationship isn't linear; it follows a pattern described by the Michaelis-Menten kinetics.
Michaelis-Menten Kinetics: Understanding the Relationship
At low substrate concentrations, the reaction rate increases proportionally with the substrate concentration. This is because there are many free enzyme molecules available to bind to the substrate. Essentially, more substrate means more frequent enzyme-substrate encounters and hence, a faster reaction rate.
However, as the substrate concentration continues to increase, the rate of reaction eventually plateaus. This is because all the enzyme molecules become saturated; they are all bound to substrate molecules, and there are no more free enzymes available to bind to additional substrate. Increasing the substrate concentration beyond this saturation point doesn't lead to a further increase in the reaction rate. This point represents the enzyme's maximum velocity (Vmax).
The Michaelis Constant (Km): A Measure of Affinity
The Michaelis constant (Km) is a valuable parameter characterizing the enzyme-substrate interaction. It represents the substrate concentration at which the reaction rate is half of its maximum velocity (Vmax). A low Km value indicates a high affinity between the enzyme and its substrate, meaning that the enzyme can effectively catalyze the reaction even at low substrate concentrations. Conversely, a high Km value indicates a low affinity, requiring a higher substrate concentration to achieve half of the maximum reaction rate.
Practical Implications of Substrate Concentration
Substrate concentration is a critical factor in many biological and industrial processes. In metabolic pathways, the regulation of substrate concentration is a key mechanism for controlling the overall rate of the pathway. In industrial applications, optimizing substrate concentration is vital for maximizing enzyme efficiency and product yield. Understanding the relationship between substrate concentration, enzyme activity, and reaction kinetics allows for the optimization of processes across numerous fields.
Conclusion: A Delicate Balance
Enzyme activity is a delicate balance, finely tuned by numerous factors. Temperature, pH, and substrate concentration are three crucial parameters influencing enzyme function. Understanding the impact of these factors is fundamental to comprehending biological processes, designing medical treatments, and optimizing industrial applications of enzymes. The interplay of these factors highlights the remarkable complexity and elegance of enzymatic systems and their vital role in sustaining life itself. Further research continues to unravel the intricate details of enzyme regulation and the multifaceted ways these molecules orchestrate life's chemical symphony. The knowledge we gain will continue to shape our understanding of biology and drive innovations in various fields, from medicine and biotechnology to environmental science and industrial processes.
Latest Posts
Latest Posts
-
Who Is The First Person To See Cells
Apr 03, 2025
-
Which Of The Following Are Examples Of Homologous Structures
Apr 03, 2025
-
Which Plasma Component Is Not Present In Serum
Apr 03, 2025
-
Physical Features Map Of South America
Apr 03, 2025
-
What Is The Basic Idea Behind Disengagement Theory
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Three Factors That Affect Enzyme Activity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
