What Are The Horizontal Columns On The Periodic Table Called
Muz Play
Apr 03, 2025 · 7 min read
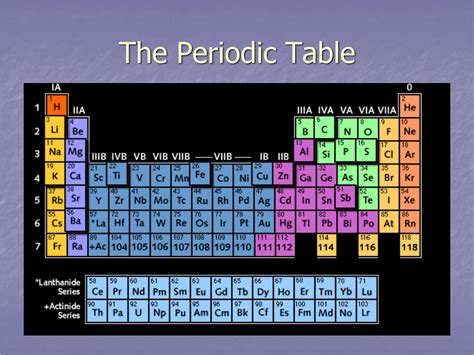
Table of Contents
What Are the Horizontal Columns on the Periodic Table Called? Understanding Periods and Their Significance
The periodic table, that iconic chart of chemical elements, is more than just a neatly organized list. Its structure, with its rows and columns, reflects fundamental properties and behaviors of atoms. While many are familiar with the vertical columns called groups or families, understanding the horizontal rows, known as periods, is crucial to grasping the periodic table's predictive power. This article delves deep into what these horizontal columns are called, their significance, and how understanding them unlocks a deeper understanding of chemistry.
Understanding Periods: A Horizontal Journey Through Atomic Structure
The horizontal rows in the periodic table are called periods. Each period represents a principal energy level or shell in an atom. As we move across a period from left to right, we're essentially adding one proton and one electron to the atom, increasing the atomic number. This addition affects the electron configuration, leading to predictable changes in the chemical properties of the elements.
Period 1: The Simplest Beginnings
Period 1, the shortest period, contains only two elements: hydrogen (H) and helium (He). These elements have electrons filling the first principal energy level (n=1), which can accommodate a maximum of two electrons. This level has only one subshell, the 1s subshell. Hydrogen, with one electron, is highly reactive, while helium, with its full 1s subshell, is exceptionally inert – a noble gas.
Period 2: Introducing Subshells and Reactivity
Period 2 is longer, containing eight elements, from lithium (Li) to neon (Ne). In this period, electrons begin filling the second principal energy level (n=2), which comprises two subshells: 2s and 2p. The increasing number of electrons in the 2s and 2p subshells significantly influences the reactivity and properties of the elements in this period. We observe a transition from highly reactive alkali metals (like lithium) to increasingly less reactive elements, culminating in the inert noble gas neon.
Period 3: Expanding Electron Configurations
Similar to Period 2, Period 3 also contains eight elements (sodium (Na) to argon (Ar)). Here, electrons fill the third principal energy level (n=3), with its 3s and 3p subshells. The pattern of reactivity mirrors Period 2, starting with a highly reactive alkali metal (sodium) and ending with an inert noble gas (argon).
Periods 4 and 5: The Introduction of d-block Elements
Periods 4 and 5 introduce a new complexity: the filling of the d-block subshells. These elements, known as transition metals, are characterized by their variable oxidation states and often exhibit colorful compounds due to the intricate electronic transitions within their d-orbitals. The addition of the d-orbitals increases the number of elements in periods 4 and 5 to 18. This expansion is due to the filling of the 3d and 4d subshells, respectively.
Periods 6 and 7: Lanthanides, Actinides, and the Expanding Table
Periods 6 and 7 are the longest periods, containing 32 elements each. This expansion is due to the filling of the f-block subshells, which house the lanthanides (rare earth elements) in Period 6 and the actinides in Period 7. These elements have complex electronic configurations and often display similar chemical properties due to the similar shielding effects within their f-orbitals. The placement of the lanthanides and actinides at the bottom of the periodic table is a matter of convenience to maintain the table's compactness and readability.
The Significance of Periods: Trends and Properties
Understanding periods is crucial for predicting the trends in various properties of elements:
-
Atomic Radius: Atomic radius generally increases as we move down a group (vertical column) due to the addition of electron shells. However, across a period (horizontal row), atomic radius generally decreases. This is because the increased nuclear charge pulls the electrons closer to the nucleus, outweighing the effect of adding electrons to the same shell.
-
Ionization Energy: Ionization energy, the energy required to remove an electron from an atom, generally increases across a period. This is due to the increased nuclear charge, which holds the electrons more tightly. It decreases down a group as the outer electrons are further from the nucleus.
-
Electronegativity: Electronegativity, the ability of an atom to attract electrons in a chemical bond, generally increases across a period. This is because elements on the right side of the periodic table have a stronger pull on electrons due to their higher nuclear charge. Electronegativity decreases down a group.
-
Metallic Character: Metallic character, the tendency of an element to lose electrons and form positive ions, generally decreases across a period. This is because the increased nuclear charge makes it harder for atoms to lose electrons. Metallic character increases down a group.
-
Chemical Reactivity: Chemical reactivity is intricately linked to the electron configuration and the tendency of an element to gain, lose, or share electrons. The reactivity patterns within periods are clearly observed, with alkali metals (Group 1) showing high reactivity and noble gases (Group 18) displaying near-zero reactivity.
Periods and the Quantum Mechanical Model
The organization of the periodic table into periods is a direct consequence of the quantum mechanical model of the atom. The quantum numbers (n, l, ml, ms) which define the orbitals within an atom directly correspond to the periodic table's arrangement.
-
Principal Quantum Number (n): This number corresponds to the energy level and determines the period number. For instance, n=1 corresponds to Period 1, n=2 to Period 2, and so on.
-
Azimuthal Quantum Number (l): This number describes the shape of the orbital (s, p, d, f). The filling of these subshells as we move across a period dictates the properties of the elements.
The quantum mechanical model beautifully explains the periodic trends observed in the properties of elements across periods and down groups. The systematic filling of electron subshells, driven by the quantum numbers, is the underlying reason for the periodic table's organization and its predictive power.
Beyond the Basics: Exploring the Nuances of Periodic Trends
While the general trends across periods are relatively straightforward, some subtle exceptions exist. These exceptions arise due to complex electron-electron interactions, shielding effects, and the relative stability of certain electron configurations. Understanding these nuances requires a deeper understanding of atomic structure and quantum mechanics.
For example, the slight irregularities in ionization energy across a period can be attributed to the different energy levels of subshells (s, p, d, f) within a particular principal energy level. Similarly, the electron configurations of certain transition metals may lead to unexpected variations in their properties compared to the general trends. Studying these exceptions enhances our grasp of the periodic table's intricacies.
The Periodic Table: A Powerful Tool for Chemical Prediction
The horizontal rows, known as periods, are an integral part of the periodic table’s design and power. Understanding periods gives us insight into:
-
Predicting Element Properties: Based on their position in a period, we can predict various properties like atomic radius, ionization energy, and electronegativity.
-
Understanding Chemical Reactions: The periodic trends help us understand why certain elements react readily with others and what types of compounds they will form.
-
Developing New Materials: The periodic table serves as a blueprint for designing and synthesizing new materials with tailored properties.
Conclusion: Periods as the Foundation of Chemical Understanding
In conclusion, the horizontal columns on the periodic table, known as periods, are fundamental to our understanding of chemical elements and their behavior. Each period represents a principal energy level, and the systematic filling of these energy levels as we move across a period leads to the predictable trends in atomic properties and chemical reactivity. By comprehending periods and their implications, we gain a deeper understanding of the periodic table's power as a tool for prediction, material design, and the advancement of chemical science. It’s a fundamental concept that underscores the elegance and predictability within the world of chemistry. The study of periods is not merely a rote memorization exercise; it's a journey into the heart of atomic structure and the fascinating interplay of forces that govern the chemical world.
Latest Posts
Latest Posts
-
What Does The Triangle Mean In Chemistry
Apr 04, 2025
-
Normal Force Is Equal To Weight
Apr 04, 2025
-
Is Sodium Carbonate An Acid Or Base
Apr 04, 2025
-
Are Hydrogen Bonds Stronger Than Covalent Bonds
Apr 04, 2025
-
Verify The Inverse Of A Function
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Are The Horizontal Columns On The Periodic Table Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
