What Chemical Element Has The Highest Electron Affinity
Muz Play
Apr 03, 2025 · 5 min read
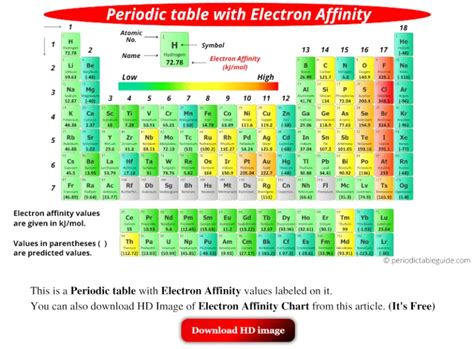
Table of Contents
What Chemical Element Has the Highest Electron Affinity?
Electron affinity, a fundamental concept in chemistry, measures the energy change when an atom gains an electron. It's a crucial property influencing an element's reactivity and its role in forming chemical bonds. While the exact value of electron affinity can be complex and dependent on factors like atomic size and electron configuration, a consistent trend reveals which element boasts the highest electron affinity. However, pinpointing the absolute highest isn't as straightforward as it might seem, due to variations in measurement techniques and theoretical calculations. This exploration delves into the nuances of electron affinity, explains the factors that influence it, and ultimately identifies the element generally considered to have the highest value.
Understanding Electron Affinity
Electron affinity (EA) is defined as the energy released when a neutral gaseous atom gains an electron to form a negative ion (anion). A higher electron affinity indicates a greater tendency for an atom to accept an electron, releasing more energy in the process. This energy release is exothermic, meaning it's accompanied by a negative value for electron affinity. Conversely, a positive value signifies that energy is required to add an electron, making the process endothermic. This is less common, particularly for elements in their ground state.
Key Factors Affecting Electron Affinity:
Several factors significantly influence an element's electron affinity:
-
Atomic Size: Smaller atoms generally have higher electron affinities. This is because the added electron experiences a stronger electrostatic attraction to the nucleus when it's closer. The increased nuclear charge effectively pulls the electron in more tightly.
-
Effective Nuclear Charge: The effective nuclear charge (Z<sub>eff</sub>) represents the net positive charge experienced by the outermost electrons. A higher Z<sub>eff</sub> leads to a stronger attraction for the incoming electron, resulting in a higher electron affinity. This is influenced by the number of protons and the shielding effect of inner electrons.
-
Electron Configuration: Half-filled and completely filled subshells are particularly stable electron configurations. Adding an electron to a half-filled or completely filled subshell disrupts this stability, resulting in a lower electron affinity (or even a positive value). For instance, adding an electron to a stable noble gas configuration requires significant energy.
-
Electron-Electron Repulsion: The added electron experiences repulsion from the existing electrons in the atom. This repulsion can counteract the attractive force from the nucleus, leading to a lower electron affinity, especially in atoms with many electrons.
The Contenders for Highest Electron Affinity
While chlorine (Cl) is often cited as having the highest electron affinity, the reality is more nuanced. The experimental determination of electron affinities is challenging, and slight discrepancies arise between different measurement techniques and theoretical calculations. However, several elements consistently appear near the top of the list:
-
Chlorine (Cl): Chlorine is generally accepted as having one of the highest electron affinities. Its relatively small size and high effective nuclear charge contribute to its strong attraction for an additional electron. Adding an electron to chlorine results in a stable configuration.
-
Fluorine (F): Fluorine, despite being the most electronegative element, displays a slightly lower electron affinity than chlorine. This unexpected finding is attributed to the smaller size of fluorine. Although it possesses a high nuclear charge, the close proximity of the electrons leads to increased electron-electron repulsion, partially offsetting the attractive force. This repulsion effect is stronger in fluorine than in chlorine, thus leading to a lower electron affinity.
-
Oxygen (O): Oxygen exhibits a relatively high electron affinity but less than chlorine or fluorine. Its electron configuration influences this; adding an electron fills a subshell, leading to a more stable configuration but a slightly lower energy release.
-
Other Halogens: Other halogens (bromine, iodine, etc.) also exhibit high electron affinities, although decreasing as you move down the group, due to increasing atomic size.
Experimental Challenges and Theoretical Calculations
Precisely measuring electron affinity experimentally is notoriously difficult. Several methods are used, including:
-
Photoelectron Spectroscopy: Measures the energy required to remove an electron from a negative ion, providing indirect information about electron affinity.
-
Collisional Ionization Methods: Involve studying the energy changes during collisions of ions and neutral atoms.
The results from these different techniques can sometimes vary slightly. Moreover, theoretical calculations, employing quantum mechanical methods, are often used to complement experimental data and provide more accurate predictions. The accuracy of these calculations depends heavily on the sophistication of the methods employed. Different levels of theory can yield slightly different results.
Why the Discrepancies? A Deeper Dive
The seemingly small differences in electron affinity between chlorine and fluorine highlight the complex interplay of various factors. While fluorine is the most electronegative element (meaning it strongly attracts electrons in a molecule), it doesn't necessarily have the highest electron affinity when considering isolated atoms in the gas phase. The key differences boil down to:
-
Electron-Electron Repulsion: As mentioned earlier, the small size of the fluorine atom results in significant electron-electron repulsion when another electron is added. This repulsion counteracts the attractive force of the nucleus. Chlorine, being larger, experiences less electron-electron repulsion.
-
Electron Correlation: Accurate theoretical calculations need to account for electron correlation, which describes the complex interactions between electrons. Ignoring these interactions can lead to inaccuracies in predicting electron affinity.
-
Polarizability: The ability of the electron cloud to distort in the presence of an electric field (polarizability) also plays a subtle role. Fluorine's smaller size gives it lower polarizability compared to chlorine.
Conclusion: Chlorine Takes the Crown (Generally)
Although fluorine is the most electronegative element and demonstrates a strong attraction for electrons in chemical bonds, chlorine (Cl) is generally considered to have the highest electron affinity among the elements when dealing with isolated gaseous atoms. This is due to the optimal balance between a relatively high effective nuclear charge and less significant electron-electron repulsion compared to fluorine. However, it's important to remember that the exact value can vary slightly depending on the measurement technique and the level of theoretical sophistication used in calculations. The differences between the top contenders are relatively small, highlighting the complexity of this fundamental atomic property. Further research and advancements in measurement and computational techniques may lead to more refined understandings of electron affinity values in the future. The ongoing refinements in our understanding underscore the intricate nature of electron interactions within atoms.
Latest Posts
Latest Posts
-
What Are Some Disadvantages Of Sexual Reproduction
Apr 04, 2025
-
M 1 And M 2 Peaks In Mass Spectrometry
Apr 04, 2025
-
The Storage Capacity Of Long Term Memory Is
Apr 04, 2025
-
The Hormone Of Hypersecretion Is
Apr 04, 2025
-
How Many Electrons Can The Fourth Energy Level Hold
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Chemical Element Has The Highest Electron Affinity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
