What Does A Chemical Equation Describe
Muz Play
Mar 31, 2025 · 6 min read
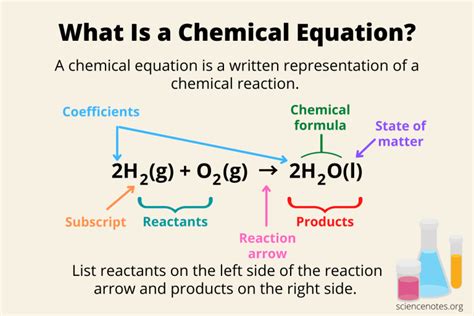
Table of Contents
- What Does A Chemical Equation Describe
- Table of Contents
- What Does a Chemical Equation Describe? A Comprehensive Guide
- Decoding the Components of a Chemical Equation
- 1. Reactants: The Starting Materials
- 2. Products: The Resulting Substances
- 3. The Arrow: Indicating Transformation
- 4. Coefficients: Balancing the Equation
- Beyond the Basics: What Chemical Equations Truly Describe
- 1. The Stoichiometry of the Reaction
- 2. The Type of Reaction
- 3. The State of the Reactants and Products
- 4. Energy Changes in the Reaction
- Advanced Concepts and Applications
- 1. Ionic Equations: Focusing on Ions
- 2. Net Ionic Equations: Even Greater Simplification
- 3. Redox Reactions and Half-Reactions: Understanding Electron Transfer
- 4. Equilibrium Reactions: Dynamic Balance
- 5. Reaction Mechanisms: Unveiling the Steps
- The Importance of Chemical Equations
- Latest Posts
- Latest Posts
- Related Post
What Does a Chemical Equation Describe? A Comprehensive Guide
Chemical equations are the language of chemistry, concisely conveying crucial information about chemical reactions. Understanding what a chemical equation describes is fundamental to grasping the principles of chemistry. This comprehensive guide will delve deep into the components, interpretation, and significance of chemical equations, equipping you with a thorough understanding of this essential tool.
Decoding the Components of a Chemical Equation
A chemical equation is a symbolic representation of a chemical reaction. It uses chemical formulas and symbols to depict the reactants (starting materials) transforming into products (resulting substances). Let's break down the key components:
1. Reactants: The Starting Materials
Reactants are the substances that undergo a chemical change during a reaction. They are written on the left-hand side of the equation, separated by plus (+) signs if multiple reactants are involved. For example, in the equation for the combustion of methane:
CH₄ + 2O₂ → CO₂ + 2H₂O
Methane (CH₄) and oxygen (O₂) are the reactants.
2. Products: The Resulting Substances
Products are the new substances formed as a result of the chemical reaction. They are written on the right-hand side of the equation, also separated by plus signs if multiple products are formed. In the methane combustion example above, carbon dioxide (CO₂) and water (H₂O) are the products.
3. The Arrow: Indicating Transformation
The arrow (→) signifies the direction of the chemical reaction. It indicates the transformation of reactants into products. Sometimes, a double arrow (⇌) is used to represent reversible reactions, where the products can react to reform the reactants.
4. Coefficients: Balancing the Equation
Coefficients are the numbers placed before chemical formulas. They indicate the relative number of molecules or moles of each substance involved in the reaction. Balancing a chemical equation is crucial because it ensures that the law of conservation of mass is obeyed—the total number of atoms of each element remains the same on both sides of the equation. In the methane combustion example, the coefficient '2' before O₂ and H₂O ensures that the number of oxygen and hydrogen atoms are equal on both sides.
Beyond the Basics: What Chemical Equations Truly Describe
While the basic components are essential, chemical equations convey much more than just the identities of reactants and products. They provide insights into:
1. The Stoichiometry of the Reaction
Stoichiometry refers to the quantitative relationships between reactants and products in a chemical reaction. Chemical equations provide the stoichiometric ratios, which are crucial for:
-
Predicting the amounts of products formed: Knowing the amount of reactants used, you can calculate the theoretical yield of the products using the stoichiometric ratios.
-
Determining the limiting reactant: When multiple reactants are involved, the limiting reactant is the one that is completely consumed first, limiting the amount of product formed. The stoichiometric ratios help identify the limiting reactant.
-
Calculating percent yield: The percent yield compares the actual yield of the product obtained in an experiment to the theoretical yield calculated from the stoichiometric ratios.
2. The Type of Reaction
The chemical equation provides clues about the type of reaction taking place. For instance:
-
Combination reactions: Two or more reactants combine to form a single product (e.g., A + B → AB).
-
Decomposition reactions: A single reactant breaks down into two or more products (e.g., AB → A + B).
-
Single displacement reactions: One element replaces another in a compound (e.g., A + BC → AC + B).
-
Double displacement reactions: Two compounds exchange ions to form two new compounds (e.g., AB + CD → AD + CB).
-
Combustion reactions: A substance reacts rapidly with oxygen, often producing heat and light (e.g., CxHy + O₂ → CO₂ + H₂O).
-
Acid-base reactions (neutralization): An acid reacts with a base to form water and a salt (e.g., HCl + NaOH → NaCl + H₂O).
3. The State of the Reactants and Products
Often, chemical equations include symbols indicating the physical state of the reactants and products:
- (s) – solid
- (l) – liquid
- (g) – gas
- (aq) – aqueous (dissolved in water)
This information is crucial for understanding the reaction conditions and the properties of the substances involved.
4. Energy Changes in the Reaction
While not always explicitly shown, chemical equations can implicitly describe energy changes. Exothermic reactions release energy (usually as heat), while endothermic reactions absorb energy. This information might be incorporated by adding "+ heat" or "- heat" to the equation or by indicating ΔH (change in enthalpy), a measure of the heat change during the reaction.
Advanced Concepts and Applications
The use and interpretation of chemical equations extend beyond basic stoichiometry and reaction types. More advanced concepts include:
1. Ionic Equations: Focusing on Ions
Ionic equations represent reactions involving ions in solution. They show only the ions that participate in the reaction, omitting spectator ions (ions that do not change during the reaction). This simplifies the representation and highlights the actual chemical changes occurring.
2. Net Ionic Equations: Even Greater Simplification
Net ionic equations are further simplified versions of ionic equations, showing only the species that directly participate in the reaction. They exclude all spectator ions.
3. Redox Reactions and Half-Reactions: Understanding Electron Transfer
Redox (reduction-oxidation) reactions involve the transfer of electrons between species. Chemical equations can be split into half-reactions, one for oxidation (loss of electrons) and one for reduction (gain of electrons), to better understand the electron transfer process.
4. Equilibrium Reactions: Dynamic Balance
For reversible reactions, the chemical equation can represent the equilibrium state, where the rates of the forward and reverse reactions are equal. The equilibrium constant (K) quantifies the relative amounts of reactants and products at equilibrium.
5. Reaction Mechanisms: Unveiling the Steps
Reaction mechanisms describe the sequence of elementary steps involved in a complex reaction. While the overall chemical equation shows the net change, the mechanism reveals the individual steps and intermediates involved.
The Importance of Chemical Equations
Chemical equations are not simply abstract representations; they are essential tools for:
-
Understanding chemical reactions: They provide a clear and concise way to represent the transformation of matter.
-
Predicting the outcome of reactions: Based on the stoichiometry and reaction type, one can predict the amounts of products formed.
-
Designing and optimizing chemical processes: Chemical equations are fundamental to designing industrial chemical processes and optimizing reaction conditions for maximum yield and efficiency.
-
Analyzing chemical systems: Equations help analyze the composition and properties of chemical systems, including equilibrium and kinetics.
-
Communication in chemistry: They provide a universal language for chemists worldwide to communicate about chemical reactions.
In conclusion, chemical equations are much more than simple formulas and symbols. They are powerful tools encapsulating vast amounts of information about chemical reactions, including stoichiometry, reaction type, energy changes, and the physical states of reactants and products. Mastering the interpretation of chemical equations is crucial for any serious student or practitioner of chemistry. Their applications span from basic laboratory experiments to complex industrial processes, underpinning our understanding of the chemical world around us.
Latest Posts
Latest Posts
-
Is Blood Clotting Negative Or Positive Feedback
Apr 03, 2025
-
Evaluating Functions Linear And Quadratic Or Cubic
Apr 03, 2025
-
What Are The Reactants In The Calvin Cycle
Apr 03, 2025
-
What Happened To Islam After The Death Of Muhammad
Apr 03, 2025
-
Which Of These Diagrams Is A Convex Mirror
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Does A Chemical Equation Describe . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
