What Does Insoluble Mean In Chemistry
Muz Play
Apr 03, 2025 · 6 min read
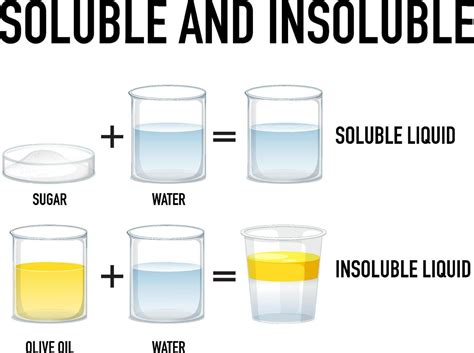
Table of Contents
What Does Insoluble Mean in Chemistry? A Comprehensive Guide
In the fascinating world of chemistry, understanding solubility is crucial. It's a fundamental concept that governs countless chemical reactions and processes. This comprehensive guide delves into the meaning of "insoluble" in chemistry, exploring its nuances, practical implications, and the factors influencing a substance's solubility. We will examine different types of insolubility, techniques for determining solubility, and the significance of this property in various fields.
Understanding Solubility: The Basics
Before we delve into the intricacies of insolubility, let's establish a firm understanding of solubility itself. Solubility refers to the maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature and pressure. The solute is the substance being dissolved, while the solvent is the substance doing the dissolving. The resulting homogeneous mixture is called a solution.
Solubility is often expressed in terms of molarity (moles of solute per liter of solution), molality (moles of solute per kilogram of solvent), or simply as grams of solute per 100 grams of solvent. These expressions help quantify the solubility of a substance under defined conditions.
What Does Insoluble Mean?
When we say a substance is insoluble, it means that it has a very low solubility in a particular solvent. This doesn't imply absolute insolubility – even the most "insoluble" compounds will dissolve to some minuscule extent. However, for practical purposes, a substance is considered insoluble if the amount that dissolves is negligible and insignificant for the application at hand. This is typically defined as less than 0.1 grams of solute per 100 grams of solvent. This threshold can vary depending on the context and the specific needs of the chemical process or experiment.
It's crucial to understand that insolubility is relative. A substance might be insoluble in water but readily soluble in another solvent like ethanol or benzene. The solubility of a compound is highly dependent on the interaction between the solute and solvent molecules.
Factors Affecting Insolubility
Several factors influence the solubility, and consequently the insolubility, of a substance. These include:
1. Nature of the Solute and Solvent: The "Like Dissolves Like" Rule
This fundamental principle dictates that polar solvents tend to dissolve polar solutes, while nonpolar solvents dissolve nonpolar solutes. This is due to the strength of intermolecular forces. Polar molecules possess permanent dipoles and interact strongly with other polar molecules through dipole-dipole interactions and hydrogen bonding. Nonpolar molecules, on the other hand, interact primarily through weaker London dispersion forces.
- Examples: NaCl (sodium chloride), a highly polar ionic compound, is readily soluble in water, a polar solvent. However, it's insoluble in nonpolar solvents like hexane. Conversely, oil (nonpolar) is insoluble in water but soluble in other nonpolar solvents.
2. Temperature
Temperature significantly affects solubility. For many solids dissolving in liquids, increasing the temperature increases solubility. This is because higher temperatures provide the molecules with more kinetic energy, making it easier to overcome the intermolecular forces holding the solid together. However, the effect of temperature on solubility is not universal; some substances exhibit decreased solubility with increasing temperature. Gases, on the other hand, generally show decreased solubility with increasing temperature.
3. Pressure
Pressure primarily affects the solubility of gases. Increasing the pressure on a gas above a liquid increases its solubility. This is described by Henry's Law, which states that the solubility of a gas is directly proportional to the partial pressure of that gas above the liquid. The effect of pressure on the solubility of solids and liquids is generally negligible.
4. Particle Size
Smaller particle sizes generally lead to increased dissolution rate, although the overall solubility (the maximum amount that dissolves) remains unchanged. Smaller particles have a larger surface area to volume ratio, providing more contact points for the solvent to interact with the solute.
Types of Insoluble Compounds
Insoluble compounds encompass a broad range of chemical substances. Some common examples include:
-
Metal hydroxides: Many metal hydroxides, such as aluminum hydroxide (Al(OH)₃) and iron(III) hydroxide (Fe(OH)₃), are insoluble in water.
-
Metal sulfides: Several metal sulfides, including copper(II) sulfide (CuS) and lead(II) sulfide (PbS), are characterized by low solubility in water.
-
Metal carbonates: Many metal carbonates, such as calcium carbonate (CaCO₃) and barium carbonate (BaCO₃), are insoluble or sparingly soluble in water.
-
Metal phosphates: Similar to carbonates, numerous metal phosphates, like calcium phosphate (Ca₃(PO₄)₂) and silver phosphate (Ag₃PO₄), are insoluble in water.
-
Organic compounds: Many organic compounds, particularly those with long nonpolar hydrocarbon chains, are insoluble in water.
Determining Insoluble Substances: Analytical Techniques
Several techniques are employed to determine whether a substance is insoluble or to quantify its low solubility. These include:
-
Qualitative observation: Simple visual inspection can often indicate insolubility. If a solid remains undissolved after thorough mixing, it is likely insoluble. However, this method is not quantitative and should be supplemented by more rigorous techniques.
-
Gravimetric analysis: This quantitative technique involves weighing the undissolved solute after a saturation experiment. By comparing this weight to the initial amount of solute, the solubility can be calculated.
-
Spectrophotometry: This method can be used to determine the concentration of a dissolved substance, even at very low concentrations. If the concentration is very low after attempting to dissolve the substance, it suggests insolubility.
-
Titration: This technique can be applied if the insoluble substance reacts with a known solution. By measuring the amount of titrant needed to react with the dissolved part, one can calculate the dissolved amount and infer solubility.
Practical Implications of Insolubility
The concept of insolubility has significant implications across various fields:
-
Pharmaceutical industry: Insolubility can affect drug absorption and bioavailability. Researchers often use techniques to improve the solubility of poorly soluble drugs to enhance their effectiveness.
-
Environmental science: Insolubility plays a role in the fate and transport of pollutants in the environment. Insoluble pollutants can accumulate in sediments and soil, posing potential long-term risks.
-
Materials science: Insolubility is utilized in the synthesis and characterization of many materials. Precipitates formed from insoluble compounds are often used to create specific materials or to purify substances.
-
Chemical analysis: The formation of insoluble precipitates is a cornerstone of many analytical methods, including gravimetric analysis and qualitative tests.
Overcoming Insolubility: Strategies and Techniques
While insolubility can sometimes be a problem, various strategies can be employed to overcome it or to utilize it effectively:
-
Changing the solvent: Using a different solvent in which the compound is more soluble can be a straightforward solution.
-
Heating the solution: Increasing temperature can increase the solubility of many solids (but not gases).
-
Using surfactants: Surfactants can reduce the surface tension of the solvent, allowing more solute to dissolve.
-
Particle size reduction: Grinding the solid into smaller particles increases the surface area available for dissolution.
-
Chemical modification: The chemical structure of the solute can be modified to enhance its solubility. This is a common strategy used in pharmaceutical science.
Conclusion: The Significance of Insolubility in Chemistry
Insolubility, despite often being viewed as the opposite of solubility, is a fundamental concept in chemistry with far-reaching implications. Understanding what insoluble means, the factors influencing it, and the various techniques used to determine and overcome it, is essential for numerous scientific and industrial applications. From drug development to environmental remediation, the concept of insolubility plays a critical role in shaping our understanding and manipulation of the chemical world. The relative nature of insolubility, its dependence on various parameters, and the practical strategies to manage it highlight its importance in the ever-evolving landscape of chemical research and technology. Further research continues to expand our understanding of this fundamental chemical property and its impact on our world.
Latest Posts
Latest Posts
-
Use The Graph To Determine The Following
Apr 04, 2025
-
Is Meiosis Asexual Or Sexual Reproduction
Apr 04, 2025
-
Why Is Organization Important To A Speech
Apr 04, 2025
-
How To Find The Mass Of An Isotope
Apr 04, 2025
-
What Is A Verbal Expression In Math
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Does Insoluble Mean In Chemistry . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
