What Factors Affect The Geometry Of A Molecule
Muz Play
Apr 05, 2025 · 6 min read
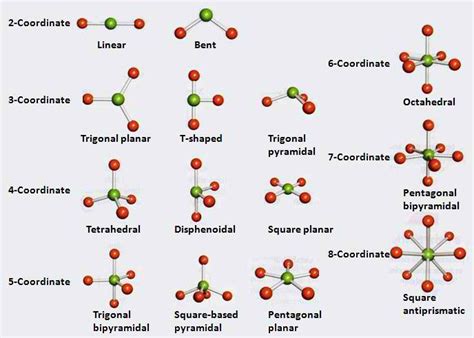
Table of Contents
What Factors Affect the Geometry of a Molecule?
Molecular geometry, the three-dimensional arrangement of atoms in a molecule, profoundly influences a molecule's physical and chemical properties. Understanding the factors that dictate this geometry is crucial in various fields, from predicting reactivity to designing new materials. This article delves deep into the key determinants of molecular geometry, explaining the concepts with clarity and providing illustrative examples.
Valence Shell Electron Pair Repulsion (VSEPR) Theory: The Cornerstone
The Valence Shell Electron Pair Repulsion (VSEPR) theory serves as the foundational model for predicting molecular geometry. This theory postulates that electron pairs, both bonding and lone pairs, in the valence shell of a central atom repel each other and arrange themselves to minimize this repulsion. This arrangement dictates the overall geometry of the molecule.
Key Concepts within VSEPR Theory:
- Electron Domains: These are regions of high electron density surrounding the central atom. They can be either bonding pairs (electrons shared between the central atom and another atom) or lone pairs (electrons not involved in bonding).
- Minimizing Repulsion: The electron domains arrange themselves as far apart as possible to minimize electrostatic repulsion. This leads to specific geometric arrangements.
- Lone Pair vs. Bonding Pair Repulsion: Lone pairs exert a stronger repulsive force than bonding pairs. This is because lone pairs are closer to the central atom and occupy a larger volume of space. This difference in repulsion strength can slightly distort the ideal geometry predicted by the theory.
Predicting Geometry using VSEPR: A Step-by-Step Guide
- Draw the Lewis Structure: Determine the number of valence electrons for each atom and construct the Lewis structure, showing all bonding and lone pairs.
- Count Electron Domains: Count the total number of electron domains around the central atom (both bonding and lone pairs).
- Determine the Electron Domain Geometry: Based on the number of electron domains, determine the electron domain geometry. Common geometries include linear (2 domains), trigonal planar (3 domains), tetrahedral (4 domains), trigonal bipyramidal (5 domains), and octahedral (6 domains).
- Consider Lone Pairs: If lone pairs are present, consider their stronger repulsive effect. This might distort the ideal geometry, leading to a different molecular geometry. For example, a molecule with four electron domains (tetrahedral electron domain geometry) could have a bent or trigonal pyramidal molecular geometry if lone pairs are involved.
- Name the Molecular Geometry: Based on the arrangement of atoms only (ignoring lone pairs), assign the final molecular geometry.
Examples:
- Methane (CH₄): Four bonding pairs, tetrahedral electron domain geometry, and tetrahedral molecular geometry.
- Water (H₂O): Two bonding pairs and two lone pairs, tetrahedral electron domain geometry, and bent molecular geometry.
- Ammonia (NH₃): Three bonding pairs and one lone pair, tetrahedral electron domain geometry, and trigonal pyramidal molecular geometry.
Beyond VSEPR: Other Influencing Factors
While VSEPR provides a good first approximation, other factors can subtly or significantly influence molecular geometry:
1. Hybridization: Shaping the Orbitals
Hybridization is the concept of mixing atomic orbitals to form new hybrid orbitals with different shapes and energies. The type of hybridization significantly impacts the geometry. Common hybridization types include:
- sp: Linear geometry (two hybrid orbitals)
- sp²: Trigonal planar geometry (three hybrid orbitals)
- sp³: Tetrahedral geometry (four hybrid orbitals)
- sp³d: Trigonal bipyramidal geometry (five hybrid orbitals)
- sp³d²: Octahedral geometry (six hybrid orbitals)
The hybridization of the central atom is often directly related to the VSEPR electron domain geometry. For example, a molecule with a tetrahedral electron domain geometry often involves sp³ hybridization.
2. Bond Order and Bond Length: The Influence of Multiple Bonds
Multiple bonds (double or triple bonds) influence geometry. Double and triple bonds occupy more space than single bonds, leading to increased repulsion and potentially altering the ideal geometry predicted by VSEPR. A double bond acts as a single electron domain, but its increased electron density leads to a slightly greater repulsive effect than a single bond.
3. Steric Effects: Bulky Groups and Crowding
Steric effects arise from the spatial interactions between atoms or groups of atoms. Bulky substituents can cause distortions in the ideal geometry by increasing steric hindrance. These groups repel each other, forcing a deviation from the predicted geometry to minimize steric strain.
4. Resonance: Delocalization and Geometry Averaging
Resonance occurs when a molecule can be represented by multiple Lewis structures, with delocalized electrons. The actual molecular geometry is often an average of the structures predicted by each resonance contributor. The molecule adopts a geometry that minimizes the overall energy of the resonance hybrid.
5. Intermolecular Forces: Weak Interactions, Big Impact
While primarily influencing bulk properties, intermolecular forces can subtly affect the conformation (the specific arrangement of atoms in space) of a molecule. Hydrogen bonding, dipole-dipole interactions, and van der Waals forces can influence the preferred orientation and overall three-dimensional structure, especially in larger molecules.
6. Ring Strain: Cyclic Molecules and Geometry Constraints
In cyclic molecules, the bond angles may be constrained by the ring structure. This can lead to deviations from the ideal bond angles predicted by VSEPR. Ring strain, the energy associated with deviations from ideal bond angles, significantly affects the stability and geometry of cyclic compounds. Smaller rings experience greater ring strain.
7. Crystal Packing Forces: Influence in the Solid State
In the solid state, crystal packing forces play a role in determining the overall arrangement of molecules. These intermolecular forces can influence the molecular conformation and even subtly alter the bond angles within the molecule to optimize packing within the crystal lattice.
Advanced Techniques for Determining Molecular Geometry
While VSEPR provides a valuable predictive tool, more sophisticated techniques are employed for accurate determination of molecular geometries:
- X-ray crystallography: This technique uses X-rays to diffract off the atoms in a crystal, providing a detailed three-dimensional map of electron density, which can be used to determine bond lengths and angles with high precision.
- Gas-phase electron diffraction: This method analyzes the scattering of electrons by gas-phase molecules to determine molecular structure.
- Neutron diffraction: Similar to X-ray diffraction, but uses neutrons instead of X-rays, allowing for the location of hydrogen atoms more easily.
- Computational chemistry: Advanced computer simulations using quantum mechanics can predict molecular geometries with high accuracy.
Conclusion: A Holistic Perspective
The geometry of a molecule is a complex interplay of various factors. While VSEPR theory provides a robust starting point, a complete understanding requires considering hybridization, bond order, steric effects, resonance, intermolecular forces, ring strain, and crystal packing effects. The interplay of these factors determines the precise three-dimensional arrangement of atoms, which in turn dictates the molecule's reactivity, physical properties, and biological function. Advanced techniques like X-ray crystallography and computational chemistry offer powerful tools for confirming and refining the predicted geometries. By understanding these factors and employing appropriate techniques, scientists can accurately determine and predict molecular geometry, paving the way for advancements in various scientific disciplines.
Latest Posts
Latest Posts
-
How Do Gametes Differ From Somatic Cells
Apr 06, 2025
-
Magnetic Field Of A Loop Formula
Apr 06, 2025
-
Characteristic Polynomial Of A 2x2 Matrix
Apr 06, 2025
-
Which Is The Best Example Of Immiscible Liquids
Apr 06, 2025
-
Is Bf3 An Acid Or Base
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about What Factors Affect The Geometry Of A Molecule . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
