What Is The Difference Between Intermolecular And Intramolecular Forces
Muz Play
Apr 03, 2025 · 6 min read
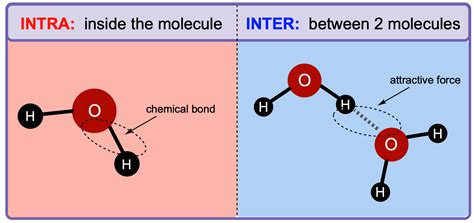
Table of Contents
Delving Deep: The Crucial Differences Between Intermolecular and Intramolecular Forces
Understanding the fundamental forces that govern the behavior of molecules is crucial in chemistry. This article dives deep into the differences between intermolecular forces and intramolecular forces, two distinct types of forces that shape the properties of matter. We'll explore their nature, strength, and impact on various physical and chemical properties.
What are Intramolecular Forces? – The Bonds That Hold Molecules Together
Intramolecular forces are the attractive forces that hold atoms together within a molecule. These forces are strong and are responsible for the formation of chemical bonds. The strength of these bonds dictates many of a molecule's characteristics, including its shape, reactivity, and stability.
Types of Intramolecular Forces:
-
Covalent Bonds: These bonds are formed by the sharing of electrons between two atoms. Covalent bonds are prevalent in organic molecules and many inorganic compounds. The strength of a covalent bond depends on factors like the electronegativity difference between the bonded atoms and the number of electron pairs shared (single, double, or triple bonds). Stronger bonds require more energy to break.
-
Ionic Bonds: These bonds are formed by the electrostatic attraction between oppositely charged ions. An ionic bond occurs when one atom donates an electron to another, creating a positively charged cation and a negatively charged anion. The strength of an ionic bond depends on the charge of the ions and the distance between them. Generally, ionic bonds are stronger than many covalent bonds.
-
Metallic Bonds: These bonds are found in metals and are characterized by a "sea" of delocalized electrons surrounding positively charged metal ions. This unique electron arrangement allows for excellent electrical and thermal conductivity and the malleability and ductility often observed in metals. The strength of a metallic bond depends on factors such as the number of valence electrons and the size of the metal atoms.
What are Intermolecular Forces? – Forces Between Molecules
Intermolecular forces (IMFs) are the attractive forces that exist between different molecules. Unlike intramolecular forces, which are strong and hold atoms together within a molecule, intermolecular forces are relatively weaker and hold molecules together in a larger assembly, such as a liquid or solid. However, the collective strength of numerous intermolecular forces can significantly influence a substance's physical properties.
Types of Intermolecular Forces:
The strength of intermolecular forces varies, impacting the physical properties of substances. The key types include:
-
London Dispersion Forces (LDFs): Also known as Van der Waals forces, these are the weakest type of intermolecular force. They result from temporary, instantaneous fluctuations in electron distribution around a molecule. These fluctuations create temporary dipoles, inducing dipoles in neighboring molecules, leading to weak attractive forces. LDFs are present in all molecules, regardless of polarity. The strength of LDFs increases with the size and surface area of the molecule. Larger molecules with more electrons experience stronger LDFs.
-
Dipole-Dipole Forces: These forces occur between polar molecules, which possess a permanent dipole moment due to an uneven distribution of electrons. The positive end of one polar molecule attracts the negative end of another, resulting in a dipole-dipole attraction. These forces are stronger than LDFs but weaker than hydrogen bonds.
-
Hydrogen Bonds: A special type of dipole-dipole force, hydrogen bonds are significantly stronger than typical dipole-dipole interactions. They occur when a hydrogen atom bonded to a highly electronegative atom (such as fluorine, oxygen, or nitrogen) is attracted to another electronegative atom in a nearby molecule. The highly electronegative atoms strongly pull the electron density away from the hydrogen atom, leaving it with a partial positive charge that can interact strongly with the lone pairs on the electronegative atom of another molecule. Hydrogen bonds play a crucial role in determining the properties of water and many biological molecules.
Key Differences Summarized: Intramolecular vs. Intermolecular Forces
| Feature | Intramolecular Forces | Intermolecular Forces |
|---|---|---|
| Nature | Strong attractive forces within a molecule | Weaker attractive forces between molecules |
| Strength | High (strong bonds) | Low (weak forces) |
| Bond Type | Covalent, Ionic, Metallic | London Dispersion, Dipole-Dipole, Hydrogen Bonding |
| Energy Changes | Large energy changes associated with bond breaking and formation | Smaller energy changes involved in overcoming forces |
| Properties Affected | Chemical properties (reactivity, stability) | Physical properties (boiling point, melting point, viscosity, surface tension) |
| Examples | Bonds within a water molecule (H-O-H) | Forces between water molecules in liquid water |
The Impact on Physical Properties: A Deeper Dive
The strength of intermolecular forces significantly influences a substance's physical properties:
-
Boiling Point and Melting Point: Substances with stronger intermolecular forces have higher boiling and melting points. This is because more energy is required to overcome the attractive forces holding the molecules together in the liquid or solid state. For instance, water has a relatively high boiling point due to its strong hydrogen bonds.
-
Viscosity: Viscosity refers to a liquid's resistance to flow. Liquids with stronger intermolecular forces have higher viscosity because the molecules are more strongly attracted to each other, making it more difficult for them to move past one another.
-
Surface Tension: Surface tension is the force that causes the surface of a liquid to contract. Stronger intermolecular forces lead to higher surface tension as the molecules at the surface are strongly attracted to each other.
-
Vapor Pressure: Vapor pressure is the pressure exerted by the vapor of a liquid in equilibrium with its liquid phase. Substances with weaker intermolecular forces have higher vapor pressures because the molecules escape the liquid phase more easily.
Examples Illustrating the Differences
Let's consider a few examples to solidify our understanding:
Example 1: Water (H₂O)
- Intramolecular forces: Covalent bonds hold the hydrogen and oxygen atoms together within each water molecule.
- Intermolecular forces: Strong hydrogen bonds exist between water molecules, explaining water's high boiling point, surface tension, and other unique properties.
Example 2: Methane (CH₄)
- Intramolecular forces: Covalent bonds link the carbon and hydrogen atoms.
- Intermolecular forces: Primarily London Dispersion Forces are present, resulting in a much lower boiling point than water due to weaker intermolecular attraction.
Example 3: Sodium Chloride (NaCl)
- Intramolecular forces: Strong ionic bonds exist between the sodium (Na⁺) and chloride (Cl⁻) ions.
- Intermolecular forces: In the solid state, the strong ionic bonds dominate, leading to a high melting point. However, once molten, the interactions between the ions are still strong, resulting in high viscosity.
Conclusion: Understanding the Interplay of Forces
Intramolecular and intermolecular forces are fundamental concepts in understanding the behavior of matter. While intramolecular forces determine the structure and stability of individual molecules, intermolecular forces govern the interactions between molecules and significantly influence a substance's physical properties. By understanding these forces, we can explain and predict the behavior of various substances across different states of matter. This knowledge is critical in diverse fields, from material science and engineering to biology and medicine. Further exploration into these concepts will undoubtedly reveal even more about the intricate world of molecular interactions.
Latest Posts
Latest Posts
-
Artists Of The Early 20th Century
Apr 04, 2025
-
How To Find The Resistance Of A Circuit
Apr 04, 2025
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
-
What Is The Net Change In Math
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is The Difference Between Intermolecular And Intramolecular Forces . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
