Where Does Reduction Occur In An Electrolytic Cell
Muz Play
Apr 04, 2025 · 6 min read
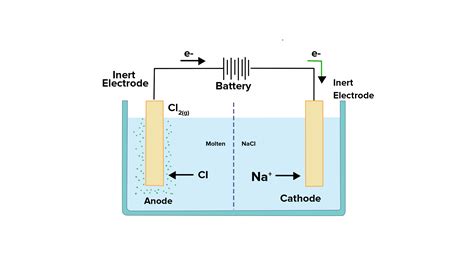
Table of Contents
Where Does Reduction Occur in an Electrolytic Cell? Understanding Cathodic Processes
Electrolytic cells are the heart of many industrial processes, from aluminum production to electroplating. Understanding their inner workings, specifically where and how reduction occurs, is crucial for anyone involved in electrochemistry or related fields. This comprehensive guide will delve deep into the cathodic processes within electrolytic cells, exploring the fundamental principles, influencing factors, and practical applications.
The Basics: Electrolytic Cells and Reduction
Before diving into the specifics of reduction location, let's establish a foundational understanding. An electrolytic cell is an electrochemical device that uses an external electric current to drive a non-spontaneous redox reaction. Unlike voltaic cells, which generate electricity from spontaneous reactions, electrolytic cells consume electricity to force a reaction to proceed. This process involves two key electrodes:
- Anode: The electrode where oxidation (loss of electrons) occurs.
- Cathode: The electrode where reduction (gain of electrons) occurs.
It's the cathode that is the focus of this article. Reduction, at its core, is the process where an atom, ion, or molecule gains electrons, resulting in a decrease in its oxidation state. In an electrolytic cell, the external power source provides the electrons necessary for this reduction process to occur at the cathode.
Why the Cathode? Understanding Electron Flow
The cathode's role as the site of reduction stems from the fundamental principles of electrochemistry and electron flow. The external power source, often a battery or power supply, establishes a potential difference (voltage) across the electrodes. This potential difference creates an electric field within the electrolytic cell. Electrons flow from the negative terminal of the power source to the cathode, making the cathode negatively charged.
This negative charge on the cathode is what attracts positively charged ions (cations) from the electrolyte solution. These cations are drawn to the negatively charged cathode where they accept electrons, undergoing reduction. The specific cation that is reduced depends on several factors which we will explore in detail below.
Analogy: A Magnet and Metal Shavings
Imagine a bar magnet attracting iron shavings. The magnet represents the negatively charged cathode, and the iron shavings represent the positively charged cations. The iron shavings are drawn to the magnet (cathode) where they "stick"—analogous to the cations gaining electrons and undergoing reduction.
Factors Influencing Reduction at the Cathode
The specific reduction process that occurs at the cathode is not arbitrary. Several factors influence which cation will be reduced and the overall efficiency of the process:
1. Standard Reduction Potential (E°)
Each ion has a characteristic standard reduction potential (E°), which indicates its tendency to gain electrons and undergo reduction. Ions with more positive E° values are more readily reduced than those with less positive (or negative) E° values. This is a crucial factor determining the order of reduction in a solution containing multiple cations.
2. Concentration of Ions
The concentration of ions in the electrolyte solution significantly influences the reduction process. Higher concentrations of a particular cation increase the probability of its reduction at the cathode, even if its standard reduction potential is less positive than another ion present in lower concentration. This is governed by the Nernst equation, which relates the cell potential to the concentrations of reactants and products.
3. Overpotential
Overpotential refers to the extra voltage required beyond the theoretical voltage predicted by the standard reduction potential to initiate the reduction process. This extra voltage is often necessary to overcome activation energy barriers, such as the formation of a new phase (e.g., the deposition of a metal). Overpotential can significantly affect the selectivity of the reduction process, favoring the reduction of ions with lower overpotentials.
4. Cathode Material
The material of the cathode itself can also play a role. The surface properties of the cathode, such as its roughness and crystal structure, can influence the adsorption of ions and the kinetics of the reduction process. Specific cathode materials might catalyze certain reduction reactions, making them more favorable than others. This is particularly relevant in heterogeneous catalysis within electrochemical processes.
5. pH of the Electrolyte
The pH of the electrolyte solution can impact the reduction process by affecting the speciation of ions. For example, certain metal ions may exist in different oxidation states or form complexes depending on the pH. These changes in speciation can alter the reduction potential and the overall reaction pathway.
Examples of Cathodic Reduction Reactions
Let's illustrate the principles discussed with some examples:
1. Electrolysis of Water
During the electrolysis of water, reduction occurs at the cathode:
2H₂O(l) + 2e⁻ → H₂(g) + 2OH⁻(aq)
Here, water molecules gain electrons, forming hydrogen gas and hydroxide ions.
2. Electroplating of Copper
In electroplating, a metal object is coated with a layer of another metal through electrolytic deposition. For copper electroplating, copper(II) ions from the electrolyte solution are reduced at the cathode:
Cu²⁺(aq) + 2e⁻ → Cu(s)
This results in the deposition of solid copper onto the object.
3. Aluminum Production (Hall-Héroult Process)
The industrial production of aluminum involves the electrolytic reduction of aluminum oxide (Al₂O₃) dissolved in molten cryolite. At the cathode, aluminum ions are reduced:
Al³⁺(l) + 3e⁻ → Al(l)
This process produces molten aluminum, which is then collected and solidified.
Practical Applications and Industrial Relevance
Understanding the reduction process at the cathode is critical in numerous industrial applications:
- Metal refining: Electrorefining uses electrolytic cells to purify metals by selectively reducing desired metal ions from impure solutions.
- Electroplating and electroforming: These techniques are used to deposit thin layers of metal onto objects or create three-dimensional metal structures.
- Metal extraction: Electrolysis is used to extract reactive metals, like aluminum, from their ores.
- Chlor-alkali process: This process uses electrolysis to produce chlorine gas and sodium hydroxide from brine. The reduction of water at the cathode is a crucial step in this process.
- Fuel cells: Although not strictly electrolytic cells, fuel cells utilize reduction reactions at the cathode to generate electricity. The reduction of oxygen is a key step in many fuel cell designs.
Conclusion: A Critical Understanding
The cathode in an electrolytic cell is the site of reduction, a process of paramount importance in electrochemistry and many industrial applications. The specific reduction reaction occurring at the cathode is determined by a complex interplay of factors, including standard reduction potentials, ion concentrations, overpotential, cathode material, and electrolyte pH. Understanding these factors is essential for designing and optimizing electrolytic processes to achieve desired outcomes. Continued research and advancements in this field will further refine our understanding and unlock new applications for electrolytic cells. This deep dive has offered a comprehensive understanding of the intricacies of cathodic reduction, its underlying principles, and its far-reaching practical implications.
Latest Posts
Latest Posts
-
Explain Why Each Function Is Continuous Or Discontinuous
Apr 05, 2025
-
Describe The Development Of The Modern Periodic Table
Apr 05, 2025
-
Which Is Most Likely A Physical Change
Apr 05, 2025
-
How To Find The Velocity Of A Vector
Apr 05, 2025
-
What Are The Tools To Measure Mass
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Where Does Reduction Occur In An Electrolytic Cell . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
