Where Is The Energy Stored In Glucose
Muz Play
Apr 03, 2025 · 6 min read
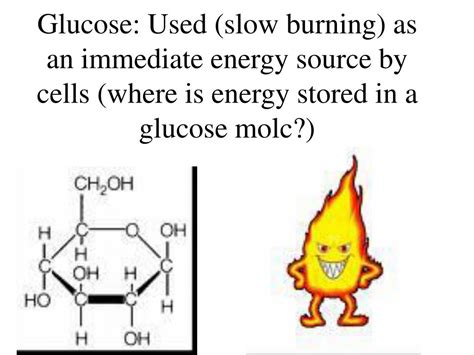
Table of Contents
Where is the Energy Stored in Glucose? Unpacking the Powerhouse of Life
Glucose, a simple sugar, is the primary source of energy for most living organisms. But where exactly is this energy stored within its seemingly simple molecular structure? It's not a matter of glucose containing energy in some readily accessible form, but rather that its chemical bonds hold the key to unlocking a substantial amount of usable energy. Understanding this requires delving into the intricacies of its molecular structure and the chemical reactions that release this stored potential.
The Structure of Glucose: A Blueprint for Energy
Glucose, a monosaccharide with the chemical formula C₆H₁₂O₆, exists in various isomeric forms, the most common being D-glucose. Its structure is crucial to understanding energy storage. The molecule is characterized by:
- Six carbon atoms: Forming the backbone of the molecule.
- Multiple hydroxyl (-OH) groups: These contribute to glucose's water solubility and its ability to participate in various chemical reactions.
- One aldehyde group (-CHO): In the open-chain form, this group is vital for the formation of crucial energy-releasing molecules. Note that in the ring form (the predominant form in solution), the aldehyde group is incorporated into a hemiacetal structure.
This seemingly simple structure belies the complex energetic relationships within its bonds. The energy isn't stored in individual atoms but rather in the arrangement and type of bonds connecting these atoms.
The Role of Chemical Bonds
The energy stored in glucose is primarily found in its covalent bonds, specifically the bonds between carbon atoms and between carbon and oxygen atoms. These bonds are high-energy bonds, meaning that a significant amount of energy is released when they are broken. This energy release is harnessed by cells through a series of carefully controlled chemical reactions.
The high-energy bonds aren't uniformly distributed. Certain bonds contain more energy than others due to factors like bond polarity and resonance stabilization. The breakage of these high-energy bonds during metabolism triggers the release of energy, which can then be used to perform cellular work, such as muscle contraction, protein synthesis, and active transport.
The Process of Energy Release: Cellular Respiration
The energy stored in glucose isn't directly accessible to the cell. It must be released through a series of carefully orchestrated steps known as cellular respiration. This process occurs in three main stages:
1. Glycolysis: Breaking Down Glucose
Glycolysis, the first stage, occurs in the cytoplasm and doesn't require oxygen. It involves a series of enzyme-catalyzed reactions that break down glucose into two molecules of pyruvate. During this process, some energy is released and stored in the form of ATP (adenosine triphosphate) and NADH (nicotinamide adenine dinucleotide). ATP is the cell's primary energy currency, while NADH is an electron carrier that will play a critical role in later stages. Crucially, this stage involves breaking specific high-energy bonds within the glucose molecule, liberating some energy in a controlled fashion.
2. The Krebs Cycle (Citric Acid Cycle): Further Energy Extraction
If oxygen is available, pyruvate enters the mitochondria, the cell's powerhouses, where the Krebs cycle takes place. Here, pyruvate is further broken down, releasing more energy that is captured as ATP, NADH, and FADH₂ (flavin adenine dinucleotide), another electron carrier. The Krebs cycle is a cyclical series of reactions that gradually oxidizes the carbon atoms from pyruvate, releasing carbon dioxide as a byproduct. Each step involves the breaking and forming of specific chemical bonds, releasing energy with each bond transformation.
3. Oxidative Phosphorylation: The Electron Transport Chain and ATP Synthase
The final stage, oxidative phosphorylation, also occurs in the mitochondria. The NADH and FADH₂ molecules generated in the previous stages deliver their electrons to the electron transport chain, a series of protein complexes embedded in the inner mitochondrial membrane. As electrons move down the chain, energy is released, and this energy is used to pump protons (H⁺ ions) across the inner mitochondrial membrane, creating a proton gradient.
This proton gradient represents a form of stored energy – potential energy. The protons flow back across the membrane through ATP synthase, a molecular machine that uses the energy from this proton flow to synthesize a large amount of ATP. This is called chemiosmosis, and it's the primary way that energy is harvested from glucose during cellular respiration. The oxygen we breathe acts as the final electron acceptor in the electron transport chain, forming water as a byproduct.
Beyond Glucose: Other Energy Storage Molecules
While glucose is the primary energy source, cells also store energy in other forms:
- Glycogen: In animals, glucose is stored as glycogen, a branched polymer of glucose molecules. Glycogen serves as a readily accessible energy reserve, especially in the liver and muscles.
- Starch: Plants store glucose as starch, another polymer of glucose, but with a different branching pattern compared to glycogen. Starch is a crucial energy source for plants and a significant food source for humans and animals.
- Fats (Triglycerides): Fats are much more efficient energy storage molecules than carbohydrates. They store significantly more energy per gram than glucose or glycogen. Fats are broken down during processes like beta-oxidation, releasing large amounts of ATP.
The Importance of Energy Storage and Release
The carefully controlled release of energy from glucose and its storage in other forms are essential for life. The ability to store energy allows organisms to survive periods of food scarcity and to meet the fluctuating energy demands of different activities. The tightly regulated process of cellular respiration ensures that energy is released gradually and efficiently, preventing damage to cellular components from sudden energy surges.
Conclusion: Glucose – A Complex Energy Package
In summary, the energy in glucose isn't stored in a single location or in a readily usable form. Instead, it's held within the high-energy covalent bonds that connect its carbon, hydrogen, and oxygen atoms. Cellular respiration is a remarkably efficient process that systematically breaks down these bonds, releasing energy that is carefully captured in the form of ATP and used to power cellular processes. Understanding the intricate details of glucose's structure and the mechanisms of energy release offers a deeper appreciation for the fundamental processes that underpin life itself. The entire process, from the structural intricacies of glucose to the complex machinery of cellular respiration, is a testament to the elegance and efficiency of biological systems. This intricate dance of molecular interactions ensures the continuous supply of energy needed to maintain life, from the simplest single-celled organism to the most complex multicellular life forms. Further research continues to unveil the complexities of energy metabolism and its importance in health and disease.
Latest Posts
Latest Posts
-
The Ul For Folate Refers Only To Folic Acid Because
Apr 04, 2025
-
Artists Of The Early 20th Century
Apr 04, 2025
-
How To Find The Resistance Of A Circuit
Apr 04, 2025
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Where Is The Energy Stored In Glucose . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
