Which Amino Acid Has A Positively Charged R Group
Muz Play
Apr 03, 2025 · 5 min read
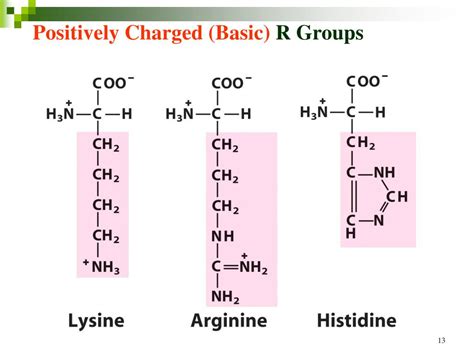
Table of Contents
Which Amino Acid Has a Positively Charged R Group? Understanding the Basics of Positively Charged Amino Acids
Amino acids are the fundamental building blocks of proteins, and their unique properties significantly influence protein structure and function. One crucial aspect of amino acid characterization is the nature of their side chains, also known as R groups. These R groups can be categorized based on their charge at physiological pH (around 7.4). This article delves into the fascinating world of amino acids with positively charged R groups, exploring their chemical properties, roles in protein structure, and significance in biological processes.
Understanding Amino Acid Structure and R Groups
Before focusing on positively charged amino acids, let's revisit the basic structure of an amino acid. Every amino acid consists of:
- A central carbon atom (α-carbon): This carbon atom is chiral (except for glycine), meaning it's bonded to four different groups.
- An amino group (-NH₂): This group is positively charged at physiological pH.
- A carboxyl group (-COOH): This group is negatively charged at physiological pH.
- A hydrogen atom (-H): A simple hydrogen atom.
- A variable side chain (R group): This is the unique part of each amino acid, dictating its properties and influencing protein structure and function.
The R group is the key determinant of an amino acid's chemical characteristics. R groups can be:
- Nonpolar (hydrophobic): These R groups tend to cluster together in the interior of proteins, away from water.
- Polar (hydrophilic): These R groups are attracted to water and often found on the protein's surface.
- Positively charged (basic): These are the focus of this article.
- Negatively charged (acidic): These R groups carry a negative charge at physiological pH.
Positively Charged Amino Acids: The Basic Trio
Three amino acids possess positively charged R groups at physiological pH:
-
Lysine (Lys, K): Lysine has a long aliphatic side chain terminating in a primary amino group (-NH₂). This amino group is readily protonated at physiological pH, resulting in a positive charge. Its fully protonated form is -NH₃⁺.
-
Arginine (Arg, R): Arginine has a more complex guanidinium group as its side chain. This group has three nitrogen atoms capable of resonance, making it exceptionally stable and strongly basic. The positive charge is delocalized across the guanidinium group. This delocalization contributes to its strong positive charge.
-
Histidine (His, H): Histidine has an imidazole ring as its side chain. The imidazole ring contains a nitrogen atom that can be protonated or deprotonated depending on the pH. While its pKa is close to physiological pH (around 6.0), at pH 7.4, histidine often carries a partial positive charge. This makes it unique among the positively charged amino acids, as its charge can vary depending on the environment.
Detailed Examination of Each Positively Charged Amino Acid
Let's delve deeper into the individual characteristics of each positively charged amino acid:
1. Lysine (Lys, K):
- Structure: A linear aliphatic chain ending in a primary amino group.
- Charge: Strongly positive at physiological pH.
- Properties: Hydrophilic due to the charged amino group. It participates in ionic interactions (salt bridges) and hydrogen bonding within proteins.
- Functions: Involved in enzymatic reactions, protein-protein interactions, and histone modification (plays a crucial role in gene regulation). Its positive charge often facilitates interaction with negatively charged molecules.
2. Arginine (Arg, R):
- Structure: A side chain containing a guanidinium group.
- Charge: Strongly positive at physiological pH due to resonance stabilization of the guanidinium group.
- Properties: Hydrophilic; strongly basic. It forms strong hydrogen bonds and participates in ionic interactions.
- Functions: Often found in active sites of enzymes, particularly those involving phosphate groups (e.g., kinases). Its strong positive charge is essential for substrate binding and catalysis.
3. Histidine (His, H):
- Structure: A side chain containing an imidazole ring.
- Charge: Partially positive at physiological pH. Its pKa is close to 7, making its charge sensitive to environmental changes.
- Properties: Hydrophilic. Can act as both an acid and a base (amphoteric). Its pKa near physiological pH allows it to act as a buffer and participate in proton transfer reactions within enzymes.
- Functions: Often found in active sites of enzymes, where its ability to accept or donate protons is crucial for catalysis. Its ability to change charge depending on pH and environment makes it highly versatile in biological processes.
The Role of Positively Charged Amino Acids in Protein Structure and Function
Positively charged amino acids play several critical roles in protein structure and function:
-
Protein Folding: The interactions between positively and negatively charged amino acids contribute to the overall three-dimensional structure of proteins. These ionic interactions (salt bridges) stabilize protein folding and contribute to protein stability.
-
Enzyme Activity: Many enzymes rely on the positive charges of these amino acids for substrate binding and catalysis. The charged side chains can attract and orient substrates correctly for reaction.
-
Protein-Protein Interactions: Positively charged amino acids frequently participate in protein-protein interactions. They can interact with negatively charged regions on other proteins, mediating protein complex formation and signaling pathways.
-
DNA Binding: Many proteins that bind DNA contain positively charged amino acids which interact with the negatively charged phosphate backbone of DNA. This interaction is essential for processes like transcription and replication.
-
Membrane Proteins: Positively charged amino acids can interact with negatively charged lipids in cell membranes, influencing the positioning and function of membrane proteins.
Clinical Significance and Applications
The properties of positively charged amino acids have far-reaching clinical implications:
-
Enzyme Deficiency: Deficiencies in enzymes dependent on specific positively charged amino acids can lead to metabolic disorders.
-
Drug Design: The positive charge of these amino acids is exploited in drug design. Many drugs interact with proteins through ionic interactions with positively charged residues.
-
Diagnostics: The presence or absence of specific positively charged amino acids can be used as diagnostic markers for certain diseases.
-
Gene Therapy: Understanding the roles of positively charged amino acids in gene regulation is crucial for developing gene therapies.
Conclusion: The Importance of Positively Charged Amino Acids
Positively charged amino acids – lysine, arginine, and histidine – are indispensable components of proteins. Their unique chemical properties, particularly their positive charge at physiological pH, play crucial roles in protein structure, function, and interactions. Their involvement in diverse biological processes, from enzymatic catalysis to gene regulation, highlights their fundamental importance in maintaining cellular life. Further research continues to unravel the multifaceted roles of these essential amino acids and their implications in health and disease. Understanding their characteristics is key to advancements in fields ranging from drug development to gene therapy.
Latest Posts
Latest Posts
-
Physical And Chemical Properties And Changes Answers
Apr 04, 2025
-
Which Process Of Cellular Respiration Produces The Most Atp
Apr 04, 2025
-
How To Fill In An Anova Table
Apr 04, 2025
-
What Are The Basic Building Blocks Of Nucleic Acids
Apr 04, 2025
-
Does Wedge Mean Axial Or Equatorial
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which Amino Acid Has A Positively Charged R Group . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
