Which State Of Matter Has A Definite Shape
Muz Play
Apr 03, 2025 · 5 min read
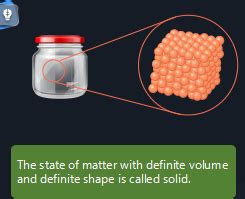
Table of Contents
Which State of Matter Has a Definite Shape? A Deep Dive into Solids, Liquids, and Gases
The question of which state of matter possesses a definite shape is a fundamental concept in science, particularly in chemistry and physics. Understanding the properties of solids, liquids, and gases is crucial to grasping the behavior of matter around us. This article will delve into the characteristics of each state of matter, explaining why only one definitively holds a fixed shape. We'll explore the underlying molecular structure and forces that dictate these properties, and touch upon some exceptions and complexities within these classifications.
Understanding the States of Matter
Before answering our central question, let's establish a firm understanding of the three primary states of matter: solid, liquid, and gas. These states are differentiated primarily by the arrangement and movement of their constituent particles (atoms, molecules, or ions).
Solids: The Rigid State
Solids are characterized by their definite shape and volume. Their particles are tightly packed together in a highly ordered, fixed arrangement. This strong intermolecular attraction and close proximity result in a rigid structure that resists deformation. The particles vibrate in place, but their movement is restricted to small oscillations around their equilibrium positions. This explains why solids maintain their shape regardless of the container they occupy.
Examples of Solids: Rocks, ice, wood, metals, and crystals all demonstrate the characteristic rigid structure and definite shape of the solid state.
Liquids: The Flowing State
Liquids, unlike solids, lack a definite shape but possess a definite volume. Their particles are close together but not rigidly fixed in a lattice structure as in solids. They exhibit a degree of freedom of movement, allowing them to flow and conform to the shape of their container. While intermolecular forces are still significant in liquids, they are weaker than in solids, allowing for the particles to slide past one another. This fluidity is a key defining characteristic of liquids.
Examples of Liquids: Water, oil, mercury, and molten metals are common examples of liquids. Note how they adapt their shape to the container they are in, while retaining a constant volume.
Gases: The Expansive State
Gases possess neither a definite shape nor a definite volume. Their particles are widely dispersed and move randomly at high speeds, with only weak intermolecular forces acting between them. Gases readily expand to fill any available space, completely taking the shape and volume of their container. This is a direct consequence of the high kinetic energy and minimal intermolecular attractions of their particles.
Examples of Gases: Air, oxygen, helium, and carbon dioxide are all examples of gases. Their ability to fill any space they are placed in is a hallmark of their state.
The Answer: Solids Have a Definite Shape
To definitively answer our question, solids are the state of matter that has a definite shape. This is a direct consequence of the strong intermolecular forces and the fixed arrangement of their particles. The particles are locked in place, unable to move freely, resulting in a rigid structure that maintains its shape regardless of external influences.
Delving Deeper: Factors Influencing Shape and Structure
While the above provides a general overview, several factors can influence the shape and structure of solids:
-
Crystalline Structure: Many solids exhibit a highly ordered, crystalline structure. This regular arrangement of particles contributes significantly to their definite shape. The type of crystal lattice (e.g., cubic, hexagonal) dictates the macroscopic shape of the solid.
-
Amorphous Solids: Some solids, known as amorphous solids (like glass), lack a well-defined crystalline structure. Their particles are arranged randomly, although still relatively close together. While they maintain a definite shape at a macroscopic level, their internal structure is less ordered than crystalline solids. This can lead to subtle differences in properties.
-
Allotropes: Certain elements can exist in different solid forms, called allotropes. For example, carbon can exist as diamond (highly ordered, hard) and graphite (layered structure, softer). These different arrangements lead to different shapes and properties.
-
External Factors: Temperature and pressure can influence the shape of solids. High temperatures can weaken intermolecular forces, leading to softening or even melting. High pressure can induce phase transitions, altering the solid's structure and consequently its shape.
Beyond the Three Primary States: Plasma and Bose-Einstein Condensates
It's important to note that the discussion of states of matter often extends beyond solids, liquids, and gases. Two additional states deserve mention:
-
Plasma: This state is characterized by ionized particles, meaning that electrons are stripped from atoms, resulting in a mixture of ions and free electrons. Plasma does not have a definite shape or volume, and it behaves differently than gases.
-
Bose-Einstein Condensate: This exotic state occurs at extremely low temperatures, where a large fraction of atoms occupy the lowest quantum state. The particles behave as a single entity and show remarkable properties, defying typical classifications of states of matter. Shape considerations are complex here, due to the quantum nature of the system.
Conclusion: A Definitive Answer with Nuances
In conclusion, while several factors can influence the precise shape and structure of a solid, the fundamental answer to the question remains: solids are the state of matter that possesses a definite shape. The strong intermolecular forces and ordered arrangement of their particles prevent free movement and maintain a rigid, consistent form. Understanding this fundamental difference between solids, liquids, and gases is critical for comprehending the physical world around us. Further exploration of the more exotic states of matter, like plasma and Bose-Einstein condensates, expands our understanding of the diverse behaviors of matter under varying conditions. This exploration emphasizes the rich and complex nature of the states of matter and how the behavior of their constituent particles determines their macroscopic properties.
Latest Posts
Latest Posts
-
The Ul For Folate Refers Only To Folic Acid Because
Apr 04, 2025
-
Artists Of The Early 20th Century
Apr 04, 2025
-
How To Find The Resistance Of A Circuit
Apr 04, 2025
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Which State Of Matter Has A Definite Shape . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
