A Bronsted-lowry Base Is Defined As
Muz Play
Apr 02, 2025 · 6 min read
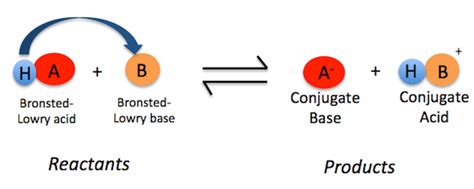
Table of Contents
A Brønsted-Lowry Base: A Deep Dive into Proton Acceptance
The world of chemistry is filled with fascinating concepts, and one that forms the bedrock of acid-base chemistry is the definition of a Brønsted-Lowry base. While seemingly simple at first glance, a thorough understanding of this definition unlocks a deeper appreciation for chemical reactions and their implications. This comprehensive article delves into the intricacies of Brønsted-Lowry bases, exploring their properties, reactions, and significance across various chemical domains.
Defining the Brønsted-Lowry Base
Unlike the simpler Arrhenius definition which limits bases to hydroxide ion (OH⁻) producers in aqueous solutions, the Brønsted-Lowry definition provides a broader, more encompassing perspective. A Brønsted-Lowry base is defined as a substance that accepts a proton (H⁺). This definition transcends the limitations of aqueous solutions, allowing us to classify a much wider range of compounds as bases. The key here is the acceptance of a proton; this proton transfer is the defining characteristic of a Brønsted-Lowry acid-base reaction.
Contrasting with Arrhenius and Lewis Bases
It's crucial to understand how the Brønsted-Lowry definition differs from other acid-base theories. The Arrhenius definition is limited to aqueous solutions and defines a base as a substance that increases the hydroxide ion (OH⁻) concentration. While many Brønsted-Lowry bases also fit the Arrhenius definition, many don't. Ammonia (NH₃), for example, is a Brønsted-Lowry base because it accepts a proton, but it doesn't directly produce OH⁻ ions in water.
The Lewis definition expands the concept further. A Lewis base is defined as an electron pair donor. While all Brønsted-Lowry bases are also Lewis bases (because they donate an electron pair to accept the proton), not all Lewis bases are Brønsted-Lowry bases. For instance, trimethylamine (N(CH₃)₃) acts as a Lewis base by donating an electron pair but doesn't necessarily accept a proton in the same way.
Therefore, the Brønsted-Lowry definition occupies a middle ground, providing a broader scope than Arrhenius while maintaining a focus on proton transfer as the central characteristic of acid-base reactions.
Identifying Brønsted-Lowry Bases: Common Examples
Numerous substances exhibit Brønsted-Lowry base behavior. Recognizing these patterns helps in predicting their reactivity and understanding their role in chemical processes. Here are some common examples:
1. Hydroxide Ions (OH⁻):
The quintessential example of a Brønsted-Lowry base, hydroxide ions readily accept protons to form water. This is the foundation of many familiar acid-base reactions. For example, the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH):
HCl + NaOH → NaCl + H₂O
Here, the OH⁻ ion acts as a Brønsted-Lowry base by accepting a proton from the HCl, which acts as a Brønsted-Lowry acid.
2. Ammonia (NH₃):
Ammonia is a classic example of a weak Brønsted-Lowry base. It readily accepts a proton from water in a reversible reaction, forming ammonium ions (NH₄⁺) and hydroxide ions (OH⁻):
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
This equilibrium reflects the relatively weak base strength of ammonia, meaning only a fraction of ammonia molecules accept protons in an aqueous solution.
3. Amines (RNH₂, R₂NH, R₃N):
Amines, organic derivatives of ammonia, also act as Brønsted-Lowry bases. The nitrogen atom, with its lone pair of electrons, readily accepts protons. The strength of the amine as a base depends on the nature of the R groups attached to the nitrogen atom. For example, methylamine (CH₃NH₂) is a stronger base than ammonia because the methyl group donates electron density to the nitrogen, increasing its basicity.
4. Carbonates (CO₃²⁻) and Bicarbonates (HCO₃⁻):
Carbonate and bicarbonate ions are important Brønsted-Lowry bases in many biological and environmental systems. They accept protons, acting as buffers to maintain pH stability. For example, in blood, the bicarbonate buffer system helps regulate blood pH.
5. Water (H₂O):
Water itself can act as both a Brønsted-Lowry acid and a Brønsted-Lowry base, a property known as amphiprotic behavior. It can donate a proton to a stronger base, like ammonia, or accept a proton from a stronger acid, like hydrochloric acid. This dual nature makes water crucial in many acid-base reactions.
Conjugate Acid-Base Pairs
A critical concept within the Brønsted-Lowry framework is the concept of conjugate acid-base pairs. When a base accepts a proton, it forms its conjugate acid. Conversely, when an acid donates a proton, it forms its conjugate base. Consider the ammonia-water reaction again:
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
In this reaction, NH₃ is the base, and NH₄⁺ is its conjugate acid (formed by accepting a proton). H₂O is the acid, and OH⁻ is its conjugate base (formed by donating a proton). Conjugate pairs always differ by a single proton (H⁺).
Factors Influencing Brønsted-Lowry Base Strength
The strength of a Brønsted-Lowry base is determined by its ability to accept a proton. Several factors influence this ability:
-
Electronegativity: Less electronegative atoms are generally better at accepting protons because they hold onto the electron pair less tightly. This is why amines are generally stronger bases than alcohols.
-
Size: Larger atoms can more effectively accommodate the added proton, leading to stronger basicity. This is evident in the trend of increasing basicity down a group in the periodic table.
-
Resonance: Resonance stabilization can either enhance or diminish basicity depending on the structure of the base. If resonance stabilizes the conjugate acid more than the base, the base's strength will be reduced.
-
Inductive Effects: Electron-donating groups increase the electron density on the atom accepting the proton, thereby increasing its basicity. Conversely, electron-withdrawing groups decrease basicity.
Brønsted-Lowry Bases in Everyday Life and Industrial Applications
Brønsted-Lowry bases play crucial roles in various aspects of daily life and industrial processes:
-
Antacids: Many antacids contain Brønsted-Lowry bases, like carbonates and hydroxides, to neutralize excess stomach acid (HCl).
-
Soaps and Detergents: Soaps and detergents often contain Brønsted-Lowry bases to increase the pH and enhance their cleaning power.
-
Fertilizers: Ammonia, a Brønsted-Lowry base, is a crucial component of many fertilizers, providing nitrogen to plants.
-
Chemical Synthesis: Brønsted-Lowry bases are widely used as catalysts and reagents in various chemical synthesis reactions.
-
Pharmaceuticals: Many pharmaceuticals function as Brønsted-Lowry bases, participating in biological processes and drug action.
Conclusion: The Importance of Understanding Brønsted-Lowry Bases
The Brønsted-Lowry definition of a base provides a powerful and versatile framework for understanding acid-base reactions. By focusing on proton transfer, this definition expands the scope of acid-base chemistry beyond aqueous solutions and encompasses a vast array of substances. Understanding the factors influencing base strength and the concept of conjugate acid-base pairs allows for the prediction and interpretation of chemical behavior across diverse fields, from everyday life to advanced chemical research. The ubiquitous presence and significance of Brønsted-Lowry bases underscore their fundamental importance in chemistry and related disciplines. Their role as essential components in biological systems, industrial processes, and chemical synthesis makes a deep understanding of their properties and reactions indispensable for anyone working in or studying the sciences. Further exploration into specific applications and more complex examples of Brønsted-Lowry bases will only deepen appreciation for their crucial role in the chemical world.
Latest Posts
Latest Posts
-
Difference Between A Somatic Cell And A Gamete
Apr 03, 2025
-
Match The Structure Process To The Letter
Apr 03, 2025
-
Find The Basis Of The Subspace
Apr 03, 2025
-
Why Is Immersion Oil Used With The 100x Objective
Apr 03, 2025
-
What Is Shared In A Covalent Bond
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about A Bronsted-lowry Base Is Defined As . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
