What Is Shared In A Covalent Bond
Muz Play
Apr 03, 2025 · 6 min read
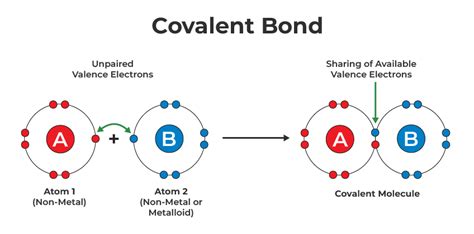
Table of Contents
What is Shared in a Covalent Bond? A Deep Dive into Chemical Bonding
Covalent bonds are the fundamental building blocks of countless molecules, forming the basis of organic chemistry and much of inorganic chemistry. Understanding what's shared in a covalent bond is crucial to grasping the properties of matter and the behavior of chemical reactions. This article delves deep into the intricacies of covalent bonding, exploring the nature of shared electrons, the types of covalent bonds, and their influence on molecular geometry and properties.
The Essence of Covalent Bonding: Shared Electrons
At the heart of a covalent bond lies the sharing of electrons between atoms. Unlike ionic bonds, where electrons are transferred from one atom to another, in a covalent bond, atoms achieve a stable electron configuration by sharing one or more pairs of valence electrons. This sharing creates a strong attractive force that holds the atoms together, forming a molecule.
Why Share Electrons? The Octet Rule
The driving force behind covalent bond formation is the octet rule, which states that atoms tend to gain, lose, or share electrons in order to achieve a full outer electron shell (valence shell) containing eight electrons. This configuration is exceptionally stable, mirroring the electron configuration of noble gases, which are chemically inert. However, it's important to note that the octet rule is a guideline, not an absolute law; exceptions exist, especially for elements in periods beyond the second row.
The Role of Valence Electrons
Only the valence electrons, those located in the outermost electron shell, participate in covalent bond formation. These electrons are the most loosely held and are most readily available for interaction with other atoms. The number of valence electrons an atom possesses dictates its bonding capacity – the number of covalent bonds it can form.
Types of Covalent Bonds: Exploring the Variations
Covalent bonds aren't monolithic; they exhibit variations based on the number of electron pairs shared and the nature of the participating atoms.
Single Covalent Bonds: One Pair Shared
A single covalent bond, also known as a sigma (σ) bond, involves the sharing of one pair of electrons between two atoms. This results in a relatively weak bond compared to multiple bonds, but it's still strong enough to hold atoms together in many stable molecules. Examples abound, from the simple hydrogen molecule (H₂) to complex organic molecules like methane (CH₄).
Double Covalent Bonds: Two Pairs Shared
A double covalent bond involves the sharing of two pairs of electrons. This results in a stronger and shorter bond than a single bond. One bond is a sigma (σ) bond, and the other is a pi (π) bond, formed by the sideways overlap of p orbitals. Ethylene (C₂H₄) is a classic example, with a carbon-carbon double bond. Double bonds are often associated with increased reactivity due to the presence of the pi bond, which is more exposed to electrophilic attack.
Triple Covalent Bonds: Three Pairs Shared
A triple covalent bond represents the sharing of three pairs of electrons between two atoms. This is the strongest type of covalent bond, with the shortest bond length. One bond is a sigma (σ) bond, and two are pi (π) bonds. Nitrogen gas (N₂) is a prime example, showcasing the exceptional stability conferred by the triple bond. Acetylene (C₂H₂) is another illustrative example.
Polar and Nonpolar Covalent Bonds: The Electronegativity Factor
The nature of the atoms involved significantly impacts the properties of the covalent bond. Electronegativity, the ability of an atom to attract electrons towards itself in a chemical bond, plays a pivotal role.
-
Nonpolar Covalent Bonds: When two atoms with similar electronegativities share electrons, the electron pair is shared equally. This leads to a nonpolar covalent bond, with no significant difference in charge distribution. The classic example is the bond between two hydrogen atoms in H₂.
-
Polar Covalent Bonds: When two atoms with differing electronegativities share electrons, the electron pair is pulled more strongly towards the atom with the higher electronegativity. This creates a polar covalent bond, resulting in a partial negative charge (δ-) on the more electronegative atom and a partial positive charge (δ+) on the less electronegative atom. Water (H₂O) is a quintessential example, with oxygen being significantly more electronegative than hydrogen. The resulting dipole moment influences the molecule's properties, making water a powerful solvent.
The Influence of Covalent Bonding on Molecular Geometry and Properties
The way atoms are bonded together through covalent linkages directly impacts the three-dimensional structure (geometry) of a molecule. Molecular geometry significantly influences various physical and chemical properties, including:
-
Melting and Boiling Points: Molecules with strong covalent bonds and larger surface areas tend to have higher melting and boiling points due to stronger intermolecular forces.
-
Solubility: Polar molecules generally dissolve well in polar solvents (like water), while nonpolar molecules dissolve better in nonpolar solvents.
-
Reactivity: The presence of double or triple bonds, along with polar bonds, can significantly influence a molecule's reactivity, making it more susceptible to certain types of chemical reactions.
-
Optical Properties: The arrangement of atoms can affect a molecule's ability to interact with light, influencing its color and other optical properties.
VSEPR Theory: Predicting Molecular Geometry
The Valence Shell Electron Pair Repulsion (VSEPR) theory provides a simple yet powerful model for predicting the three-dimensional arrangement of atoms in a molecule based on the repulsion between electron pairs in the valence shell. By considering the number of bonding and non-bonding electron pairs, VSEPR theory helps predict molecular geometries such as linear, bent, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
Beyond Simple Covalent Bonds: Coordinate Covalent Bonds
In a coordinate covalent bond (also called a dative covalent bond), both electrons shared in the bond originate from the same atom. This is in contrast to typical covalent bonds where each atom contributes one electron to the shared pair. The atom donating the electron pair is called the donor, and the atom accepting the electron pair is called the acceptor. Coordinate covalent bonds are often found in complex ions and certain organic molecules. Ammonia (NH₃) can act as a donor, forming a coordinate covalent bond with a hydrogen ion (H⁺) to create the ammonium ion (NH₄⁺).
Covalent Bonds in Organic Chemistry: The Foundation of Life
Covalent bonds are the cornerstone of organic chemistry, the chemistry of carbon-containing compounds. Carbon's unique ability to form four covalent bonds, along with its ability to form chains and rings, allows for the immense diversity of organic molecules found in living organisms. From simple hydrocarbons to complex proteins and DNA, the intricate network of covalent bonds governs the structure and function of biological systems.
Conclusion: The Ubiquitous Nature of Covalent Bonds
Covalent bonds are ubiquitous in nature, forming the backbone of countless molecules that shape our world. Understanding the principles of covalent bonding—the sharing of electrons, the variations in bond strength and polarity, and the influence on molecular geometry—is essential for comprehending the properties and reactivity of matter. This article has provided a comprehensive overview, from the fundamental principles to the complexities of different bond types and their influence on molecular properties, establishing a strong foundation for further exploration in the fascinating world of chemistry. The intricate interplay of shared electrons, electronegativity, and molecular geometry creates the diverse range of compounds that govern the physical and biological world around us. Continued research and advancements in our understanding of covalent bonding will undoubtedly unlock further insights into the intricate workings of the universe.
Latest Posts
Latest Posts
-
Does Km Increase With Competitive Inhibition
Apr 03, 2025
-
Right Lateral Vs Left Lateral Fault
Apr 03, 2025
-
The Measure Of The Amount Of Matter In An Object
Apr 03, 2025
-
How To Calculate Hydronium Ion Concentration
Apr 03, 2025
-
How Many Valence Electrons Do Alkali Metals Have
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Is Shared In A Covalent Bond . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
