Does Km Increase With Competitive Inhibition
Muz Play
Apr 03, 2025 · 6 min read
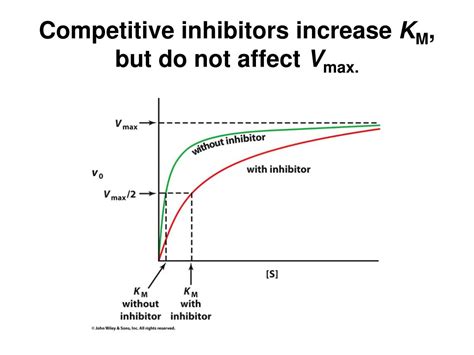
Table of Contents
Does Km Increase with Competitive Inhibition? A Deep Dive into Enzyme Kinetics
Enzyme kinetics is a cornerstone of biochemistry, providing crucial insights into how enzymes function and how they are regulated. Understanding enzyme kinetics is vital in various fields, from drug discovery to metabolic engineering. One of the key concepts in enzyme kinetics is the Michaelis-Menten constant (Km), a measure of an enzyme's affinity for its substrate. This article delves into the effects of competitive inhibition on the Km value, explaining the underlying mechanisms and their implications.
Understanding the Michaelis-Menten Equation and Km
The Michaelis-Menten equation describes the relationship between the initial reaction rate (v) of an enzyme-catalyzed reaction and the substrate concentration ([S]). The equation is:
v = Vmax[S] / (Km + [S])
Where:
- v represents the initial reaction velocity.
- Vmax represents the maximum reaction velocity when the enzyme is saturated with substrate.
- [S] represents the substrate concentration.
- Km represents the Michaelis constant.
Km is defined as the substrate concentration at which the reaction velocity is half of Vmax (v = Vmax/2). A low Km value indicates high substrate affinity, meaning the enzyme can achieve half its maximum velocity at a lower substrate concentration. Conversely, a high Km value indicates low substrate affinity. It's crucial to understand that Km is not simply a measure of binding affinity; it's a reflection of both the rate of substrate binding and the rate of product formation.
Competitive Inhibition: A Molecular Tug-of-War
Competitive inhibition occurs when an inhibitor molecule structurally resembles the substrate and competes with it for binding to the enzyme's active site. The inhibitor binds reversibly to the active site, preventing the substrate from binding and thus reducing the reaction rate. Crucially, the inhibitor doesn't alter the enzyme's structure in a way that affects its ability to bind substrate, only its availability to bind substrate.
Imagine the active site as a lock, the substrate as the correct key, and the competitive inhibitor as a similar but incorrect key. Both keys can fit into the lock (bind to the active site), but only the correct key (substrate) can open the lock (initiate the catalytic process). The more incorrect keys (inhibitor) present, the less frequently the correct key (substrate) can access the lock.
The Impact of Competitive Inhibitors on Enzyme Kinetics
The presence of a competitive inhibitor affects the Michaelis-Menten equation. The apparent Km value increases, while Vmax remains unchanged. This is because the inhibitor is competing with the substrate for binding. To achieve half of the Vmax, you need a higher substrate concentration because some of the enzyme molecules are occupied by the inhibitor. Therefore, the apparent Km value increases.
Increased apparent Km: The higher the inhibitor concentration, the higher the apparent Km. This signifies that a higher substrate concentration is required to achieve half-maximal velocity in the presence of the inhibitor.
Unchanged Vmax: At sufficiently high substrate concentrations, the substrate can outcompete the inhibitor for binding to the enzyme's active site. This means that even with the inhibitor present, the reaction can still reach its maximum velocity (Vmax). The inhibitor essentially dilutes the active enzyme concentration, not altering the potential catalytic activity of the enzyme itself.
Graphical Representation of Competitive Inhibition
The effects of competitive inhibition are clearly visualized using Lineweaver-Burk plots (double reciprocal plots) and Dixon plots.
Lineweaver-Burk Plot
This plot shows the reciprocal of the reaction velocity (1/v) against the reciprocal of the substrate concentration (1/[S]). In the absence of an inhibitor, the plot yields a straight line with a y-intercept of 1/Vmax and an x-intercept of -1/Km.
With a competitive inhibitor present, several lines are generated, each representing a different inhibitor concentration. These lines will have:
- The same y-intercept (1/Vmax): Confirming that Vmax remains unchanged.
- Different x-intercepts (-1/Km(app)): Reflecting the increase in the apparent Km value with increasing inhibitor concentration. The lines will converge at the same y-intercept.
Dixon Plot
The Dixon plot is a useful tool to determine the inhibition constant (Ki), which is a measure of the inhibitor's affinity for the enzyme. It plots 1/v against the inhibitor concentration [I] at different substrate concentrations. In competitive inhibition, the lines intersect to the left of the y-axis, and the x-intercept represents -Ki.
Mechanisms Behind the Km Increase
The increase in Km in competitive inhibition stems from the direct competition between the substrate and the inhibitor for binding to the active site. The inhibitor's presence reduces the effective concentration of the free enzyme available to bind the substrate. Therefore, a higher substrate concentration is needed to saturate the available enzyme and reach half-maximal velocity. This directly translates to a higher apparent Km.
The inhibitor does not directly influence the catalytic rate (kcat) or the binding affinity of the enzyme-substrate complex. The observed effect is purely a matter of reduced enzyme availability due to inhibitor occupancy.
Distinguishing Competitive Inhibition from Other Types of Inhibition
It's crucial to distinguish competitive inhibition from other types of enzyme inhibition, such as uncompetitive and non-competitive inhibition. These other types of inhibition exhibit different effects on Km and Vmax.
- Uncompetitive Inhibition: In uncompetitive inhibition, the inhibitor binds only to the enzyme-substrate complex. This results in a decrease in both Km and Vmax.
- Non-competitive Inhibition: In non-competitive inhibition, the inhibitor can bind to both the free enzyme and the enzyme-substrate complex. This results in a decrease in Vmax but no change in Km.
Practical Applications and Significance
Understanding competitive inhibition is critical in various areas:
-
Drug Design: Many drugs act as competitive inhibitors, targeting specific enzymes involved in disease processes. By designing drugs that effectively compete with substrates for enzyme binding, we can modulate enzyme activity and alleviate disease symptoms. This is particularly relevant in areas such as antibiotic development and cancer therapy.
-
Metabolic Engineering: Competitive inhibitors can be used to manipulate metabolic pathways in microorganisms for biotechnological applications. By inhibiting specific enzymes, one can redirect metabolic fluxes to produce desired products.
-
Biochemical Assays: Competitive inhibition is frequently employed in enzyme assays to determine enzyme kinetics parameters and inhibitor potency.
Conclusion: A Critical Parameter in Enzyme Regulation
The increase in Km with competitive inhibition is not merely a theoretical concept; it’s a fundamental aspect of enzyme regulation with significant practical implications. This phenomenon highlights the dynamic interplay between enzymes, substrates, and inhibitors, shaping the overall efficiency of biochemical pathways. Understanding the mechanisms and consequences of competitive inhibition is vital for researchers and practitioners in various fields relying on enzyme kinetics to understand and manipulate biological systems. The accurate determination of Km and its changes in the presence of inhibitors is crucial for designing effective drugs, engineering metabolic pathways, and developing sensitive enzyme assays. The unchanging Vmax is a defining feature of this type of inhibition, readily distinguishable through graphical analysis like the Lineweaver-Burk and Dixon plots. Therefore, the knowledge of how Km is affected by competitive inhibition remains a cornerstone of biochemistry and related disciplines.
Latest Posts
Latest Posts
-
Which Is A Postulate Of The Kinetic Molecular Theory
Apr 04, 2025
-
What Are The Units For A Spring Constant
Apr 04, 2025
-
How To Make A Contact Sheet On Bridge
Apr 04, 2025
-
Pretest And Posttest Control Group Design
Apr 04, 2025
-
Max Rate Of Change Directional Derivative
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Does Km Increase With Competitive Inhibition . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
