A Chemical Reaction Has Reached Equilibrium When
Muz Play
Apr 05, 2025 · 6 min read
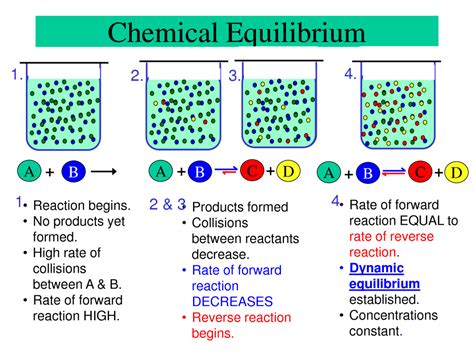
Table of Contents
A Chemical Reaction Has Reached Equilibrium When… Understanding Dynamic Equilibrium
Chemical equilibrium is a fundamental concept in chemistry, crucial for understanding how reactions proceed and the final state they achieve. It's not a static state where all reactions cease, but rather a dynamic equilibrium, where the rates of the forward and reverse reactions become equal. This means that while individual molecules continue to react, the overall concentrations of reactants and products remain constant. Understanding this dynamic balance is key to predicting reaction outcomes and controlling chemical processes.
What is Chemical Equilibrium?
At its core, chemical equilibrium describes a state where the rates of the forward and reverse reactions are equal. This doesn't imply that the concentrations of reactants and products are equal; rather, it means that the net change in their concentrations is zero. Consider a simple reversible reaction:
A + B ⇌ C + D
In this reaction, A and B react to form C and D (forward reaction), while C and D can also react to reform A and B (reverse reaction). At equilibrium, the rate of the forward reaction (A + B → C + D) is precisely balanced by the rate of the reverse reaction (C + D → A + B).
The Dynamic Nature of Equilibrium
It's crucial to understand that equilibrium is a dynamic process. Individual molecules of A and B are still colliding and reacting to form C and D, and similarly, molecules of C and D are still reacting to reform A and B. However, these forward and reverse reactions occur at the same rate, leading to no net change in the concentrations of reactants or products. This constant molecular activity is what makes equilibrium a dynamic, rather than static, state.
Think of it like a crowded marketplace. People are constantly entering and leaving, buying and selling. At a certain point, the number of people entering equals the number of people leaving, and the overall number of people in the marketplace remains relatively constant. This is analogous to chemical equilibrium: the "flow" of reactants and products is constant, but the overall amounts remain stable.
Factors Affecting Equilibrium: Le Chatelier's Principle
The position of equilibrium—the relative amounts of reactants and products—can be influenced by several factors. Le Chatelier's principle provides a useful framework for understanding these influences: If a change of condition is applied to a system in equilibrium, the system will shift in a direction that relieves the stress.
This principle applies to several key factors:
1. Concentration Changes
Changing the concentration of reactants or products will shift the equilibrium to counteract the change. Increasing the concentration of a reactant will shift the equilibrium towards the products (favoring the forward reaction) to consume the added reactant. Conversely, increasing the concentration of a product will shift the equilibrium towards the reactants (favoring the reverse reaction) to consume the added product.
2. Temperature Changes
Temperature changes affect the equilibrium constant (K), which quantifies the relative amounts of reactants and products at equilibrium. The effect of temperature depends on whether the reaction is exothermic (releases heat) or endothermic (absorbs heat).
-
Exothermic reactions: Increasing the temperature shifts the equilibrium towards the reactants (favoring the reverse reaction), as the system attempts to absorb the added heat. Decreasing the temperature shifts the equilibrium towards the products (favoring the forward reaction).
-
Endothermic reactions: Increasing the temperature shifts the equilibrium towards the products (favoring the forward reaction), as the system absorbs the added heat. Decreasing the temperature shifts the equilibrium towards the reactants (favoring the reverse reaction).
3. Pressure Changes
Pressure changes primarily affect equilibrium in reactions involving gases. Increasing the pressure shifts the equilibrium towards the side with fewer moles of gas, reducing the total pressure. Decreasing the pressure shifts the equilibrium towards the side with more moles of gas. If the number of moles of gas is the same on both sides of the equation, pressure changes have no effect on the equilibrium position.
4. Addition of a Catalyst
A catalyst speeds up both the forward and reverse reactions equally. Therefore, a catalyst does not affect the position of equilibrium. It only speeds up the rate at which equilibrium is reached.
The Equilibrium Constant (K)
The equilibrium constant (K) is a quantitative measure of the position of equilibrium. It's the ratio of the concentrations of products to the concentrations of reactants, each raised to the power of its stoichiometric coefficient in the balanced chemical equation. For the general reversible reaction:
aA + bB ⇌ cC + dD
The equilibrium constant expression is:
K = ([C]<sup>c</sup>[D]<sup>d</sup>) / ([A]<sup>a</sup>[B]<sup>b</sup>)
where [A], [B], [C], and [D] represent the equilibrium concentrations of the respective species.
-
A large value of K (K >> 1) indicates that the equilibrium lies far to the right, meaning that the concentration of products is significantly higher than the concentration of reactants at equilibrium. The reaction is product-favored.
-
A small value of K (K << 1) indicates that the equilibrium lies far to the left, meaning that the concentration of reactants is significantly higher than the concentration of products at equilibrium. The reaction is reactant-favored.
-
A value of K ≈ 1 indicates that the equilibrium lies in the middle, meaning that the concentrations of reactants and products are comparable at equilibrium.
Determining if Equilibrium is Achieved
Experimentally determining if a reaction has reached equilibrium often involves monitoring the concentrations of reactants and products over time. If the concentrations remain constant over a significant period, it suggests that equilibrium has been established. Spectroscopic techniques, such as UV-Vis or NMR spectroscopy, can be used to quantitatively measure these concentrations.
The time required to reach equilibrium varies depending on the reaction's kinetics. Some reactions reach equilibrium rapidly, while others may take hours, days, or even longer. Factors like temperature, concentration, and the presence of a catalyst influence the rate at which equilibrium is attained.
Applications of Equilibrium
The principles of chemical equilibrium have broad applications across various fields:
-
Industrial Processes: Many industrial chemical processes, such as the Haber-Bosch process for ammonia synthesis, are designed to maximize product yield by manipulating equilibrium conditions.
-
Environmental Chemistry: Equilibrium concepts are crucial in understanding environmental processes, such as acid-base reactions in water bodies and the distribution of pollutants in the atmosphere.
-
Biochemistry: Metabolic pathways in living organisms involve numerous reversible reactions operating near equilibrium. Understanding equilibrium helps us understand how these pathways regulate cellular processes.
-
Analytical Chemistry: Equilibrium principles underlie many analytical techniques, such as titrations, which are used to determine the concentration of substances.
Beyond Simple Equilibria: Complex Systems
While we've primarily discussed simple reversible reactions, many real-world systems involve more complex equilibria. These can include:
-
Simultaneous Equilibria: Multiple equilibria may coexist in a single system, influencing each other.
-
Heterogeneous Equilibria: Reactions involving different phases (e.g., solid, liquid, gas) present additional considerations, such as the surface area of solids.
-
Ionic Equilibria: Equilibria involving ions in solution, such as acid-base reactions and solubility equilibria, require specialized treatment due to the effects of ionic strength and activity coefficients.
Understanding chemical equilibrium is crucial for a comprehensive grasp of chemistry and its applications. It's a dynamic process that governs the relative amounts of reactants and products in reversible reactions, influencing a wide array of phenomena in the natural world and chemical industries. By mastering the principles of equilibrium and Le Chatelier's principle, we gain the ability to predict reaction outcomes and manipulate chemical systems to achieve desired results.
Latest Posts
Latest Posts
-
Excluded Values Of A Rational Expression
Apr 05, 2025
-
Iron Iii Chloride Test For Aspirin
Apr 05, 2025
-
When To Use Brackets Or Parentheses In Domain And Range
Apr 05, 2025
-
Which Step In Cellular Respiration Produces The Most Atp
Apr 05, 2025
-
The Highness Or Lowness Of A Sound Is Called
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about A Chemical Reaction Has Reached Equilibrium When . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
