An Oxidizing Agent Is A Substance That
Muz Play
Apr 05, 2025 · 6 min read
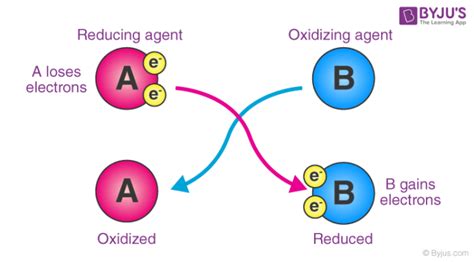
Table of Contents
An Oxidizing Agent is a Substance That… Accepts Electrons! A Deep Dive into Oxidation and Reduction
An oxidizing agent, also known as an oxidant, is a substance that accepts electrons from another substance, causing it to be oxidized. This seemingly simple definition underpins a vast array of chemical processes crucial to life, industry, and the environment. Understanding oxidizing agents requires a firm grasp of redox reactions, the fundamental principles governing electron transfer. This comprehensive guide will delve into the intricacies of oxidizing agents, exploring their properties, common examples, applications, and safety considerations.
Understanding Redox Reactions: The Foundation of Oxidizing Agents
Redox reactions, short for reduction-oxidation reactions, are chemical reactions involving the transfer of electrons between two species. One species loses electrons (oxidation), while another gains electrons (reduction). These two processes are always coupled; you cannot have oxidation without reduction, and vice versa. This is why they're called redox reactions.
-
Oxidation: The loss of electrons by a substance. The oxidation state of the substance increases. Think of it as losing negatively charged electrons, making the species more positive.
-
Reduction: The gain of electrons by a substance. The oxidation state of the substance decreases. Think of it as gaining negatively charged electrons, making the species more negative.
Oxidizing agents are the substances responsible for causing oxidation in other species. Because they accept electrons, they themselves undergo reduction. Therefore, oxidizing agents are also called reducing agents. This seemingly contradictory terminology highlights the coupled nature of redox reactions. One substance’s oxidation is always accompanied by another’s reduction.
Identifying Oxidizing Agents: Key Characteristics
Several characteristics can help you identify a potential oxidizing agent:
-
High Electronegativity: Oxidizing agents tend to be highly electronegative, meaning they strongly attract electrons. Elements like oxygen, fluorine, chlorine, and bromine are prime examples.
-
High Oxidation State: A substance in a high oxidation state is more likely to accept electrons and be reduced, thus acting as an oxidizing agent. For instance, permanganate (MnO₄⁻) with manganese in a +7 oxidation state is a strong oxidizing agent.
-
Presence of Easily Reduced Atoms: The presence of atoms that can readily gain electrons contributes to the oxidizing power of a substance. For example, the presence of oxygen atoms in many oxidizing agents facilitates electron acceptance.
-
Ability to Accept Electrons: This is the defining characteristic. An oxidizing agent's inherent ability to readily accept electrons determines its strength as an oxidizing agent.
Common Examples of Oxidizing Agents
The world is brimming with oxidizing agents, each with its unique properties and applications. Here are some prominent examples:
1. Oxygen (O₂): The Ubiquitous Oxidant
Oxygen is perhaps the most ubiquitous and vital oxidizing agent. It's essential for respiration in living organisms, where it acts as the terminal electron acceptor in the electron transport chain. Combustion, the rapid oxidation of a fuel, also heavily relies on oxygen. Rusting of iron, a slow oxidation process, is another familiar example of oxygen's oxidizing power.
2. Ozone (O₃): A Potent Oxidizer
Ozone is a highly reactive allotrope of oxygen, a much stronger oxidizing agent than molecular oxygen (O₂). Its potent oxidizing abilities make it useful in water purification and sterilization. However, its reactivity also makes it a significant pollutant in the lower atmosphere.
3. Hydrogen Peroxide (H₂O₂): Versatile Oxidizing Agent
Hydrogen peroxide is a common household antiseptic and bleaching agent. Its oxidizing power stems from the peroxide bond (O-O), which readily releases oxygen. This released oxygen then oxidizes various substances.
4. Potassium Permanganate (KMnO₄): Strong Oxidizing Agent
Potassium permanganate is a powerful oxidizing agent widely used in titrations, water treatment, and as a disinfectant. The manganese in permanganate (MnO₄⁻) has a +7 oxidation state, making it highly susceptible to reduction.
5. Potassium Dichromate (K₂Cr₂O₇): Another Powerful Oxidant
Potassium dichromate is another strong oxidizing agent often used in organic chemistry for oxidation reactions. The chromium in dichromate has a +6 oxidation state. Its orange color changes to green upon reduction.
6. Chlorine (Cl₂): Disinfectant and Bleaching Agent
Chlorine is a potent oxidizing agent used extensively as a disinfectant in water treatment and as a bleaching agent in the textile industry. Its oxidizing power stems from its high electronegativity.
7. Nitric Acid (HNO₃): Powerful Oxidizing and Acidic Agent
Nitric acid is a strong oxidizing agent and acid. It oxidizes many metals and is used in various industrial processes and laboratory settings.
8. Chromic Acid (H₂CrO₄): Strong Oxidizing Agent in Organic Chemistry
Chromic acid is a strong oxidizing agent commonly used in organic chemistry to oxidize alcohols to ketones or aldehydes.
Applications of Oxidizing Agents: A Wide Spectrum
The applications of oxidizing agents are incredibly diverse, spanning various fields:
1. Industry: Bleaching, Metallurgy, and Chemical Synthesis
Oxidizing agents are crucial in numerous industrial processes. Bleaching agents, like hydrogen peroxide and chlorine, are used to whiten fabrics and paper. Metallurgy uses oxidizing agents for processes like ore extraction and metal refining. Chemical synthesis relies heavily on oxidizing agents to create various compounds.
2. Medicine and Healthcare: Disinfectants and Antiseptics
Oxidizing agents play a vital role in medicine and healthcare. Hydrogen peroxide and iodine are common antiseptics used to disinfect wounds. Other oxidizing agents are used in the development of pharmaceuticals and medical treatments.
3. Environmental Science: Water Treatment and Pollution Control
Oxidizing agents are essential in water treatment, removing pollutants and disinfecting water supplies. Ozone and chlorine are commonly used for this purpose. They are also employed in various environmental remediation processes.
Safety Considerations When Handling Oxidizing Agents
Many oxidizing agents are highly reactive and can pose significant safety risks. Appropriate safety precautions are essential when handling them:
-
Protective Equipment: Always use appropriate personal protective equipment (PPE), including gloves, safety goggles, and lab coats.
-
Ventilation: Ensure adequate ventilation to prevent inhalation of hazardous fumes.
-
Storage: Store oxidizing agents separately from reducing agents to prevent accidental reactions.
-
Disposal: Follow proper procedures for disposing of oxidizing agents to avoid environmental contamination.
-
Fire Safety: Be aware of the fire hazards associated with certain oxidizing agents and have appropriate fire suppression equipment available.
Conclusion: The Essential Role of Oxidizing Agents
Oxidizing agents are fundamental to a vast array of chemical reactions and processes. Their ability to accept electrons drives numerous reactions crucial to life, industry, and environmental processes. Understanding their properties, applications, and safety considerations is vital for anyone working with chemicals or interested in chemistry. From the simple act of rusting to complex industrial processes and medical applications, oxidizing agents play an irreplaceable role in our world. Continued research and development in this field will undoubtedly lead to new and innovative applications in the future. Remembering that they are always paired with a reducing agent during a redox reaction helps to fully appreciate their importance in the chemical world. Their impact is extensive and deeply integrated into the fabric of our existence.
Latest Posts
Latest Posts
-
Delta S Delta H Delta G Chart
Apr 05, 2025
-
How Many Unpaired Electrons Does Sulfur Have
Apr 05, 2025
-
The Shape Of A Water Molecule Is
Apr 05, 2025
-
Study Of The Evolutionary Relationships Among Organisms
Apr 05, 2025
-
What Is The Disadvantage Of A Corporation
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about An Oxidizing Agent Is A Substance That . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
