How Many Unpaired Electrons Does Sulfur Have
Muz Play
Apr 05, 2025 · 5 min read
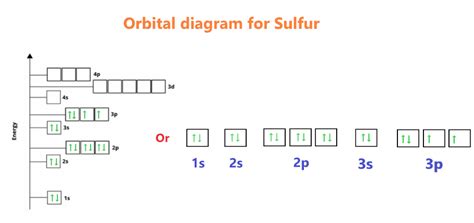
Table of Contents
How Many Unpaired Electrons Does Sulfur Have? A Deep Dive into Atomic Structure and Electron Configuration
Sulfur, a vibrant yellow nonmetal crucial to life and industry, holds a fascinating position in the periodic table. Understanding its electron configuration is key to unlocking its unique properties and reactivity. This article delves into the details of sulfur's atomic structure, explaining precisely how many unpaired electrons it possesses and why this matters.
Understanding Electron Configuration
Before we explore sulfur specifically, let's establish the fundamental principles of electron configuration. Atoms are made up of a nucleus containing protons and neutrons, surrounded by electrons orbiting in specific energy levels or shells. These electrons occupy orbitals, regions of space where there's a high probability of finding an electron. Each orbital can hold a maximum of two electrons, according to the Pauli Exclusion Principle, but these electrons must have opposite spins (one spin-up, one spin-down).
The filling of these orbitals follows specific rules, primarily the Aufbau principle (electrons fill lower energy levels first) and Hund's rule (electrons individually occupy orbitals within a subshell before pairing up). These rules dictate the electron configuration, which is a shorthand notation describing the arrangement of electrons in an atom's orbitals.
Sulfur's Position on the Periodic Table and its Implications
Sulfur (S) is located in Group 16 (also known as the chalcogens) and Period 3 of the periodic table. Its atomic number is 16, meaning it has 16 protons and, in a neutral atom, 16 electrons. This position dictates its electron configuration and, consequently, its number of unpaired electrons.
Determining Sulfur's Electron Configuration
Using the Aufbau principle and Hund's rule, we can determine the electron configuration of sulfur:
1s² 2s² 2p⁶ 3s² 3p⁴
Let's break this down:
- 1s²: The first energy level (n=1) contains one subshell, 's', which holds a maximum of two electrons. Both electrons are paired in the 1s orbital.
- 2s²: The second energy level (n=2) also contains an 's' subshell, holding another pair of electrons.
- 2p⁶: The second energy level also contains three 'p' subshells (px, py, pz), each capable of holding two electrons. All six electrons in the 2p subshells are paired.
- 3s²: The third energy level (n=3) has an 's' subshell with a paired electron duo.
- 3p⁴: This is where things get interesting. The third energy level also has three 'p' subshells. With four electrons to fill these three orbitals, Hund's rule comes into play. Each of the three 3p orbitals will receive one electron before any pairing occurs. This leaves one unpaired electron in the third 3p orbital.
The Number of Unpaired Electrons in Sulfur
From the electron configuration, we can clearly see that sulfur has two unpaired electrons. These are located in the 3p subshells.
Significance of Unpaired Electrons
The presence of unpaired electrons significantly influences sulfur's chemical behavior and properties. These unpaired electrons are readily available to participate in chemical bonds, making sulfur a highly reactive element. This reactivity is responsible for its diverse range of compounds and its importance in various biological and industrial processes.
Reactivity and Bonding
The two unpaired electrons contribute to sulfur's ability to form covalent bonds. It can form single, double, or even multiple bonds depending on the bonding partner. For example, sulfur forms single bonds in hydrogen sulfide (H₂S) and double bonds in sulfur dioxide (SO₂). This versatility is a direct consequence of its unpaired electrons.
Magnetic Properties
Unpaired electrons also give rise to paramagnetism. Paramagnetic substances are weakly attracted to magnetic fields due to the presence of unpaired electrons. Sulfur, with its two unpaired electrons, exhibits paramagnetic properties. This is a measurable property that further confirms the presence of unpaired electrons.
Oxidation States
Sulfur displays a wide range of oxidation states, largely due to its ability to gain or lose electrons to achieve a stable electron configuration. This flexibility in oxidation states further demonstrates the importance of its unpaired electrons. It can exist in oxidation states ranging from -2 (as in H₂S) to +6 (as in H₂SO₄).
Sulfur's Diverse Applications
The presence of these unpaired electrons is central to the vast array of applications sulfur enjoys:
-
Vulcanization of Rubber: Sulfur is crucial in vulcanizing rubber, a process that improves its strength and elasticity. The sulfur atoms form cross-links between the polymer chains, creating a more robust material. This crucial process relies heavily on the chemical reactivity derived from sulfur's unpaired electrons.
-
Production of Sulfuric Acid: Sulfuric acid (H₂SO₄), one of the most important industrial chemicals, is produced through a process involving sulfur. Its many applications include fertilizer production, metal processing, and the manufacture of various chemicals and materials. The unpaired electrons facilitate the various chemical reactions involved in its production.
-
Fertilizers: Sulfur is a vital component of many fertilizers, essential for plant growth. It's a critical element in the formation of amino acids and proteins, processes directly influenced by sulfur's reactivity.
-
Matches and Fireworks: The ability of sulfur to readily combust, another result of its unpaired electrons, makes it a key ingredient in matches and fireworks.
-
Pharmaceuticals: Sulfur finds applications in various pharmaceuticals, contributing to the efficacy of certain medications.
-
Biological Roles: Sulfur plays essential roles in biological systems. It's found in certain amino acids like cysteine and methionine, and is crucial for the formation of proteins and enzymes.
Conclusion
In conclusion, sulfur possesses two unpaired electrons, a fact directly determined by its electron configuration (1s² 2s² 2p⁶ 3s² 3p⁴). The presence of these unpaired electrons is fundamentally important to its chemical reactivity, magnetic properties, and the diverse range of oxidation states it exhibits. This reactivity is, in turn, the basis of its widespread and vital applications in various industrial processes, manufacturing, and biological systems. Understanding the significance of these unpaired electrons provides crucial insight into the behavior and importance of this essential element. Further exploration into sulfur's chemistry will continue to reveal its versatile applications and significance in our world.
Latest Posts
Latest Posts
-
Which Elements Have The Highest Ionization Energy
Apr 06, 2025
-
Calculate Total Magnification Of A Microscope
Apr 06, 2025
-
What Are The Resources Of A Business
Apr 06, 2025
-
Label The Bones Of The Appendicular Skeleton
Apr 06, 2025
-
The Carbohydrates Glucose Galactose And Fructose
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Unpaired Electrons Does Sulfur Have . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
