Are Hydrogen Bonds Weak Or Strong
Muz Play
Apr 04, 2025 · 6 min read
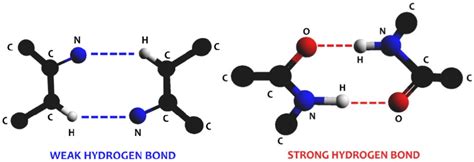
Table of Contents
Are Hydrogen Bonds Weak or Strong? A Deep Dive into Intermolecular Forces
The question of whether hydrogen bonds are weak or strong is a deceptively simple one. The answer, as with many things in science, is: it depends. While often categorized as "weak" compared to covalent or ionic bonds, hydrogen bonds are significantly stronger than other intermolecular forces like van der Waals interactions, and their strength plays a crucial role in a vast array of biological and chemical processes. This article delves into the nature of hydrogen bonds, exploring their strength relative to other bond types and examining the factors that influence their strength.
Understanding Hydrogen Bonds: The Basics
A hydrogen bond is a special type of dipole-dipole attraction between molecules, not a true chemical bond. It occurs when a hydrogen atom bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine) is attracted to another electronegative atom in a different molecule. This electronegativity difference creates a significant polarity, resulting in a partially positive hydrogen (δ+) and a partially negative electronegative atom (δ−). The electrostatic attraction between the δ+ hydrogen and the δ− atom constitutes the hydrogen bond.
Think of it like this: the electronegative atom "hogs" the shared electrons in the covalent bond with hydrogen, leaving the hydrogen with a partial positive charge. This partially positive hydrogen is then attracted to the partially negative charge on another electronegative atom in a neighboring molecule. This attraction is the hydrogen bond.
Key Characteristics of Hydrogen Bonds:
- Strength: Generally weaker than covalent and ionic bonds, but stronger than other intermolecular forces.
- Directionality: Hydrogen bonds are directional, meaning they are strongest when the hydrogen atom is aligned directly with the electronegative atom it is attracted to.
- Length: The length of a hydrogen bond is typically longer than a covalent bond.
- Number: Multiple hydrogen bonds can form between molecules, leading to a cumulative effect on overall strength and stability.
Comparing Hydrogen Bond Strength to Other Bond Types
To accurately assess the strength of hydrogen bonds, we need to compare them to other types of bonds:
1. Covalent Bonds: The Strongest Link
Covalent bonds involve the sharing of electrons between atoms. These bonds are significantly stronger than hydrogen bonds, requiring a considerable amount of energy to break. The strength of a covalent bond depends on factors like the atoms involved and the bond order (single, double, or triple bond).
Example: The O-H bond in water is a strong covalent bond, requiring substantial energy to break. The energy needed to break a covalent bond is typically in the range of 150-800 kJ/mol.
2. Ionic Bonds: Electrostatic Attraction
Ionic bonds result from the electrostatic attraction between oppositely charged ions. These bonds are also significantly stronger than hydrogen bonds, forming strong crystalline lattices in many ionic compounds. The strength of an ionic bond depends on the charge and size of the ions involved.
Example: The bond in sodium chloride (NaCl) is an ionic bond, requiring considerable energy to break. The energy required is typically in the range of 100-1000 kJ/mol.
3. Van der Waals Forces: Weak Intermolecular Interactions
Van der Waals forces are a collection of weak intermolecular attractions, including London dispersion forces, dipole-dipole interactions, and ion-dipole interactions. These forces are significantly weaker than hydrogen bonds. They arise from temporary or permanent fluctuations in electron distribution around molecules.
Example: The attraction between nonpolar molecules like methane (CH₄) is due to weak London dispersion forces. The energy associated with these forces is typically less than 40 kJ/mol.
Hydrogen Bonds: Bridging the Gap
Hydrogen bonds fall between the strong covalent/ionic bonds and the weak van der Waals forces. Their strength typically ranges from 4 to 40 kJ/mol. While this is significantly less than covalent and ionic bonds, it is considerably stronger than other intermolecular forces. This intermediate strength is what makes them so crucial in many biological systems.
Factors Affecting Hydrogen Bond Strength
Several factors influence the strength of a hydrogen bond:
-
Electronegativity of the donor and acceptor atoms: The greater the electronegativity difference between the hydrogen donor and acceptor atoms, the stronger the hydrogen bond. Bonds involving fluorine (F) are generally stronger than those involving oxygen (O) or nitrogen (N).
-
Geometry and orientation: The strength of a hydrogen bond is maximized when the hydrogen atom, the donor atom, and the acceptor atom are collinear. Deviations from linearity weaken the bond.
-
Solvent effects: The presence of other molecules (solvents) can affect hydrogen bond strength by competing for hydrogen bonds or influencing the polarity of the molecules involved. For instance, water, a highly polar solvent, can weaken hydrogen bonds between solute molecules by competing for the hydrogen atoms.
-
Temperature: Higher temperatures generally weaken hydrogen bonds as the increased kinetic energy disrupts the electrostatic attraction.
-
Pressure: Pressure can influence the strength and formation of hydrogen bonds by affecting the intermolecular distances and orientations.
The Importance of Hydrogen Bonds in Biological Systems
The unique strength and properties of hydrogen bonds are essential for the structure and function of many biological molecules and systems:
-
Water's Properties: Hydrogen bonds are responsible for many of water's unique properties, including its high boiling point, high specific heat capacity, and its ability to act as a universal solvent. These properties are vital for life as we know it.
-
Protein Structure: Hydrogen bonds play a crucial role in stabilizing the secondary, tertiary, and quaternary structures of proteins. Alpha-helices and beta-sheets, fundamental structural motifs in proteins, are stabilized by extensive hydrogen bonding networks.
-
DNA Structure: The double helix structure of DNA is maintained by hydrogen bonds between complementary base pairs (adenine-thymine and guanine-cytosine). These bonds are strong enough to maintain the stability of the DNA double helix but weak enough to allow for strand separation during DNA replication and transcription.
-
Enzyme-Substrate Interactions: Hydrogen bonds are often involved in the specific binding of substrates to enzymes, contributing to the catalytic activity of enzymes. The precise positioning of the substrate within the enzyme's active site is often facilitated by a network of hydrogen bonds.
-
Carbohydrate Structure: The three-dimensional structures of carbohydrates, crucial in energy storage and cell recognition, are significantly influenced by hydrogen bonding.
-
Lipid Bilayers: While primarily hydrophobic, the interactions at the periphery of lipid bilayers, and even in some specialized lipid structures, are influenced by hydrogen bonding.
Conclusion: Context Matters
The question of whether hydrogen bonds are weak or strong lacks a straightforward yes or no answer. They are undeniably weaker than covalent and ionic bonds. However, they are considerably stronger than other intermolecular forces and their cumulative effects can be substantial. Their intermediate strength, combined with their directionality and ability to form multiple bonds, makes them crucial in a wide variety of biological and chemical contexts. Understanding the nuances of hydrogen bond strength, along with the factors influencing it, is vital to comprehending many fundamental processes in nature. The impact of hydrogen bonds far surpasses their seemingly modest energy compared to stronger bond types; they are the unsung heroes driving countless essential biological processes. Their delicate balance between strength and reversibility is a testament to the elegance and efficiency of nature’s design.
Latest Posts
Latest Posts
-
Identify The Oxidation Half Reaction Of Select One
Apr 05, 2025
-
Matter Is Conserved Physical Or Chemica Change
Apr 05, 2025
-
Difference Between Nuclear Reaction And Chemical Reaction
Apr 05, 2025
-
Matter That Has A Uniform And Definite Composition
Apr 05, 2025
-
Why Does 4s Fill Before 3d
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Are Hydrogen Bonds Weak Or Strong . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
