Matter Is Conserved Physical Or Chemica Change
Muz Play
Apr 05, 2025 · 6 min read
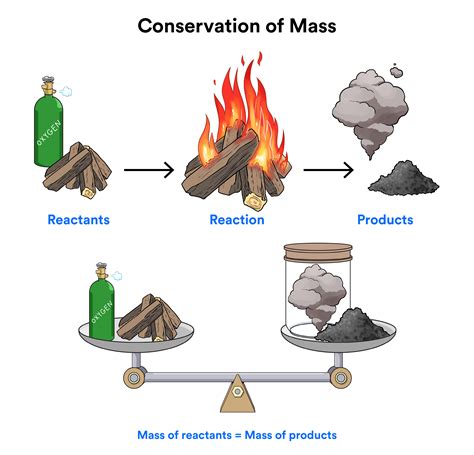
Table of Contents
Matter is Conserved: A Deep Dive into Physical and Chemical Changes
The principle of the conservation of mass is a cornerstone of both physics and chemistry. It dictates that matter can neither be created nor destroyed, only transformed. This seemingly simple statement has profound implications for understanding physical and chemical changes, guiding our interpretations of reactions and interactions within the universe. Let's explore this fundamental principle, delving into the specifics of how it applies to physical and chemical transformations.
Understanding the Conservation of Mass
At its core, the conservation of mass principle states that in a closed system (a system where neither matter nor energy can enter or leave), the total mass of the system remains constant, regardless of any changes occurring within it. This doesn't mean that the form of the matter remains constant – far from it. Instead, it means that the total quantity of matter remains the same.
This principle has been refined over time, particularly with the advent of Einstein's theory of relativity. Relativity showed that mass and energy are interconvertible, related through the famous equation E=mc². In high-energy processes, such as nuclear reactions, a small amount of mass can be converted into a significant amount of energy, or vice versa. However, even in these scenarios, the total mass-energy of the system remains conserved. For most everyday chemical and physical changes, however, the relativistic effects are negligible, and the classic conservation of mass principle provides an excellent approximation.
Physical Changes: A Transformation Without Alteration
Physical changes are transformations that alter the form or appearance of matter without changing its chemical composition. No new substances are formed during a physical change. The same atoms are present before and after the change, simply arranged differently. Crucially, the conservation of mass principle holds true during all physical changes.
Examples of Physical Changes:
- Changes of state: Melting ice (solid to liquid), boiling water (liquid to gas), freezing water (liquid to solid), and deposition (gas to solid) are classic examples. The water molecules remain H₂O throughout, only the arrangement and energy of the molecules change. The total mass of the water remains unchanged throughout these transformations.
- Dissolution: Dissolving salt in water is another prime example. The salt crystals break down into individual ions (Na⁺ and Cl⁻) surrounded by water molecules, but the chemical identity of the salt and water remains unchanged. The total mass of the solution (salt + water) is equal to the sum of the masses of the salt and water before mixing.
- Crushing, cutting, and bending: These actions alter the physical shape of a substance but do not alter its chemical makeup. Crushing a rock into smaller pieces changes its form but not its elemental composition. The total mass of the rock remains the same.
Conservation of Mass in Physical Changes:
In each of these examples, the conservation of mass principle is evident. While the physical properties of the matter may change, its chemical composition and therefore its overall mass remains constant. This is demonstrably true through careful experimental measurements of the mass before and after the physical change. Any apparent loss or gain in mass is usually attributed to experimental error or unaccounted-for factors, such as evaporation or absorption of moisture.
Chemical Changes: New Substances Emerge, but Mass Remains Constant
Chemical changes, also known as chemical reactions, involve the rearrangement of atoms to form new substances with different chemical properties. The bonds between atoms break, and new bonds form, resulting in the creation of products that differ significantly from the original reactants. Despite this dramatic transformation, the conservation of mass principle still holds.
Examples of Chemical Changes:
- Combustion: Burning wood is a chemical change. The wood (primarily cellulose) reacts with oxygen in the air to produce carbon dioxide, water vapor, and ash. The atoms of carbon, hydrogen, and oxygen are rearranged, forming new molecules. However, the total mass of the reactants (wood + oxygen) equals the total mass of the products (carbon dioxide + water vapor + ash), ignoring the minor mass loss due to the escape of gases.
- Rusting: The rusting of iron is another example. Iron reacts with oxygen in the presence of water to form iron oxide (rust). The iron atoms combine with oxygen atoms to form a new compound. The total mass of the iron and oxygen consumed equals the total mass of the rust formed.
- Photosynthesis: Plants convert carbon dioxide and water into glucose (sugar) and oxygen. The atoms of carbon, hydrogen, and oxygen are rearranged to form new molecules. The total mass of the reactants (carbon dioxide + water) equals the total mass of the products (glucose + oxygen).
Conservation of Mass in Chemical Changes:
While it might seem intuitive that burning wood results in a mass decrease (ashes weigh less than the original wood), a more accurate analysis accounts for the mass of the gases (carbon dioxide and water vapor) produced. Similarly, rusting adds mass to the iron object because it combines with oxygen from the air. Precise measurements demonstrate that the total mass is conserved in chemical reactions, even when the phase or form of the matter changes drastically.
Practical Applications and Importance of Mass Conservation
The principle of conservation of mass is not just a theoretical concept; it has wide-ranging practical applications:
- Stoichiometry: In chemistry, stoichiometry uses the conservation of mass principle to perform calculations relating the amounts of reactants and products in chemical reactions. It's essential for determining the yield of a reaction, balancing chemical equations, and designing industrial chemical processes.
- Environmental science: Understanding mass conservation is crucial for analyzing environmental processes, such as pollution control and waste management. Tracking the movement and transformation of pollutants allows for effective remediation strategies.
- Engineering: In engineering applications, the conservation of mass is critical in designing and analyzing various systems, such as fluid flow in pipelines, the combustion process in engines, and nuclear reactors.
- Forensic Science: In forensic science, mass conservation plays a key role in several analyses. For example, examining the mass of materials in a crime scene can be used in recreating events and developing crucial evidence.
Challenges and Refinements to the Principle
While the law of conservation of mass is exceptionally useful in most scenarios, it's important to acknowledge its limitations:
- Relativistic Effects: As previously mentioned, at speeds approaching the speed of light, or in nuclear reactions involving high energy transformations, mass and energy are interconvertible. The total mass-energy of the system remains conserved, but the classical concept of mass conservation needs modification.
- Open Systems: The law applies strictly to closed systems. If matter can enter or leave a system, then the total mass within the system will not necessarily remain constant. This necessitates careful consideration of boundaries when applying the law.
Conclusion: A Fundamental Principle with Lasting Significance
The principle of the conservation of mass is a fundamental concept in science that underpins our understanding of physical and chemical changes. It emphasizes that matter is neither created nor destroyed, only transformed, even in reactions that dramatically alter the form and properties of substances. While relativistic effects modify the principle at extreme energy levels, the law remains a powerfully useful approximation in the vast majority of physical and chemical phenomena, forming a crucial basis for numerous scientific and engineering applications. Its continued relevance highlights its enduring importance within the scientific community. A thorough grasp of this principle is essential for anyone seeking a deeper understanding of the natural world and its transformative processes.
Latest Posts
Latest Posts
-
Can A Stereocenter Have A Double Bond
Apr 05, 2025
-
Is The Trp Operon Inducible Or Repressible
Apr 05, 2025
-
How Do You Calculate Current In A Series Circuit
Apr 05, 2025
-
How Do You Solve Systems Of Equations Algebraically
Apr 05, 2025
-
What Is The Relationship Between Anatomy And Physiology
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Matter Is Conserved Physical Or Chemica Change . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
