Can A Stereocenter Have A Double Bond
Muz Play
Apr 05, 2025 · 6 min read
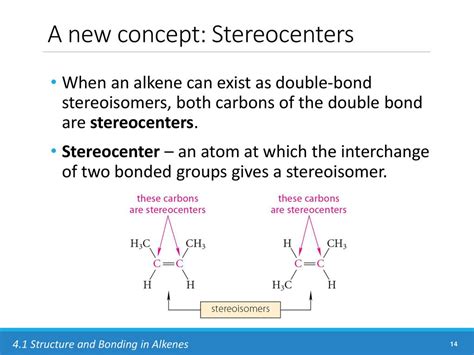
Table of Contents
Can a Stereocenter Have a Double Bond? Exploring the Relationship Between Stereochemistry and Unsaturation
Stereochemistry, the study of the three-dimensional arrangement of atoms and molecules, plays a crucial role in determining the properties and reactivity of chemical compounds. A key concept in stereochemistry is the stereocenter, also known as a chiral center, which is an atom bearing four different groups. This asymmetry leads to the existence of stereoisomers, molecules with the same connectivity but different spatial arrangements. However, the presence of a double bond introduces a different layer of complexity. This article will delve into the question: can a stereocenter have a double bond? The answer, as we'll see, is nuanced and depends on the specific definition of "stereocenter" and the molecular structure involved.
Understanding Stereocenters and Chirality
Before addressing the central question, let's solidify our understanding of stereocenters. A stereocenter is typically defined as an atom (usually carbon, but can also be other atoms like silicon or phosphorus) that has four different groups attached. This asymmetry leads to chirality, meaning the molecule is non-superimposable on its mirror image. These non-superimposable mirror images are called enantiomers. A classic example is bromochlorofluoromethane (CHBrClF), where the central carbon atom is a stereocenter.
The Tetrahedral Carbon and Stereocenters
The most common type of stereocenter is a tetrahedral carbon atom. The tetrahedral geometry arises from the sp³ hybridization of the carbon atom, resulting in four distinct bonding orbitals arranged in a tetrahedron. This specific geometry is essential for the existence of enantiomers and, consequently, chirality. If any two groups attached to the carbon atom are identical, the molecule becomes achiral.
Double Bonds and their Impact on Stereochemistry
Double bonds, formed by the overlap of two p orbitals, introduce a significant constraint to molecular geometry. The atoms involved in a double bond exhibit sp² hybridization, leading to a planar arrangement of the atoms. This planarity restricts rotation around the double bond, which has profound implications for stereochemistry.
Geometric Isomerism (Cis-Trans Isomerism)
The restricted rotation around the double bond leads to the existence of geometric isomers, also known as cis-trans isomers or E/Z isomers. These isomers differ in the spatial arrangement of substituents around the double bond. In cis isomers, identical or similar groups are on the same side of the double bond, while in trans isomers, they are on opposite sides. This type of isomerism is distinct from enantiomerism.
Can a sp² hybridized carbon be a stereocenter?
The presence of a double bond, with its associated sp² hybridization, immediately raises questions about the possibility of a stereocenter at that carbon atom. A simple answer would be no. A carbon atom with a double bond has only three groups directly bonded to it, not four. Therefore, it can't meet the standard definition of a stereocenter as having four different groups attached. However, the subtlety comes into play with certain specific molecular structures.
Exceptions and Subtleties
While a simple double-bonded carbon cannot be a stereocenter, there are exceptions and more nuanced scenarios to consider:
-
Molecules with multiple stereocenters: A molecule can possess multiple stereocenters, some of which might be sp³ hybridized and thus true stereocenters, while others might be sp² hybridized carbons participating in double bonds. The overall stereochemistry of the molecule will be determined by the configuration at each individual stereocenter.
-
Restricted Rotation: Although the carbon atom within the double bond is not itself a stereocenter, the restricted rotation around the double bond can impact the molecule's overall stereochemistry. It is the arrangement of groups across the double bond that influence the properties.
-
Cyclic structures: In cyclic compounds containing double bonds, the restricted rotation around the double bond and the ring structure can influence the stereochemistry in ways that don't fit the simple definition. Certain ring systems can exhibit axial and equatorial arrangements of groups impacting the overall properties.
-
Stereogenic axis: While not a stereocenter in the traditional sense, an allene (a molecule with two adjacent double bonds) can possess a stereogenic axis. In an allene, the central carbon atom is sp hybridized, and the molecule exhibits chirality due to the perpendicular orientation of the two double bonds. This is a special case where chirality is linked with the presence of double bonds but not due to a stereocenter in the classical definition.
-
Stereogenic plane: Molecules possessing a stereogenic plane, meaning a plane of symmetry that cannot be superimposed on its mirror image, also exhibits chirality. These molecules may have double bonds, demonstrating that chirality does not always necessitate a single stereocenter.
Analyzing Specific Examples
To illustrate the complexities, let's consider a few examples:
Example 1: A simple alkene
Consider propene (CH3CH=CH2). The sp² hybridized carbon atom in the double bond does not qualify as a stereocenter; it only has three groups attached: a methyl group, a hydrogen atom, and another CH2 group.
Example 2: A molecule with multiple stereocenters
Consider a molecule with two stereocenters, one being sp³ hybridized and the other sp² hybridized. The molecule would exhibit stereoisomerism due to the different configurations at the sp³ hybridized carbon, but the sp² hybridized carbon wouldn't itself be a stereocenter. The overall stereochemistry of the molecule would be significantly affected by the presence of the double bond, due to the resulting geometric isomerism.
Example 3: A cyclic molecule with a double bond
In a cyclic molecule containing a double bond, the double bond's position and the ring structure influence the overall molecular configuration, which often leads to isomerism. However, a single carbon atom within the double bond usually won't be a stereocenter by the common definition.
Conclusion: Redefining the Stereocenter Concept
The question of whether a stereocenter can have a double bond highlights the limitations of strictly adhering to the traditional definition of a stereocenter. While a carbon atom involved in a double bond (sp² hybridized) cannot be a stereocenter in the classic sense (having four different groups attached), the presence of double bonds invariably impacts the molecule's overall stereochemistry through geometric isomerism and other effects like restricted rotation and the presence of stereogenic axes or planes.
Therefore, it's more accurate to state that while a double-bonded carbon atom itself isn't a stereocenter, its presence significantly influences the stereochemistry of the molecule as a whole. The impact of double bonds on overall molecular configuration is paramount in understanding and predicting the properties and reactivity of molecules. The sophisticated interplay of double bonds and other stereochemical features necessitates a deeper, more nuanced understanding than simply relying on the basic definition of a stereocenter. We must consider the overall molecular architecture and employ the concepts of geometric isomerism, stereogenic axes and planes to fully grasp the stereochemical complexity of these molecules.
Latest Posts
Latest Posts
-
How To Make A Titration Curve In Excel
Apr 05, 2025
-
Which Describes The Complex Carbohydrate Cellulose
Apr 05, 2025
-
After Glycolysis The Pyruvate Molecules Go To The
Apr 05, 2025
-
Converting Polar Equations To Cartesian Equations
Apr 05, 2025
-
Classify Environmental Hazards As Physical Chemical Or Biological
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Can A Stereocenter Have A Double Bond . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
