How To Make A Titration Curve In Excel
Muz Play
Apr 05, 2025 · 6 min read
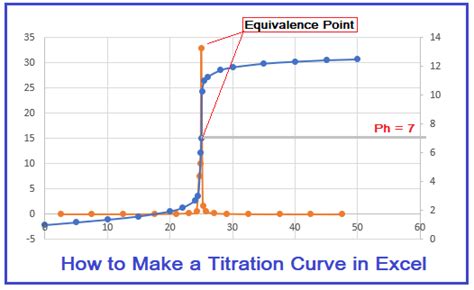
Table of Contents
How to Make a Titration Curve in Excel: A Comprehensive Guide
Titration is a fundamental analytical technique used in chemistry to determine the concentration of an unknown solution by reacting it with a solution of known concentration. Plotting the data obtained during a titration generates a titration curve, a graphical representation that provides valuable insights into the reaction's progress and the equivalence point. While manual plotting is possible, using Microsoft Excel significantly simplifies this process, allowing for quick analysis and accurate interpretation. This comprehensive guide will walk you through the steps of creating a precise and informative titration curve in Excel.
Understanding Titration Data
Before diving into Excel, let's review the data you'll need. A typical titration experiment involves recording the volume of titrant (the solution of known concentration) added and the corresponding pH (or other relevant measurement like conductivity or potential) of the solution. This data is usually presented in a table:
| Volume of Titrant (mL) | pH |
|---|---|
| 0 | 2.0 |
| 5 | 2.2 |
| 10 | 2.5 |
| 15 | 3.0 |
| 20 | 3.8 |
| 25 | 4.5 |
| 30 | 7.0 |
| 35 | 10.0 |
| 40 | 11.0 |
| 45 | 11.5 |
This table shows a strong acid-strong base titration. Notice the dramatic pH change near the equivalence point (around 30 mL). This is the characteristic 'S' shape of a titration curve. Understanding this data is crucial before you start plotting. Inaccurate data will lead to an inaccurate curve.
Importing Data into Excel
The first step is to import your titration data into Excel. You can do this by:
-
Manually entering the data: Open a new Excel spreadsheet and type your data into two columns. Label the columns clearly, for example, "Volume (mL)" and "pH."
-
Copying and pasting: If your data is in another program (like a lab notebook or a text file), copy the data and paste it into your Excel spreadsheet. Ensure that the data is pasted correctly, with each value in its own cell.
-
Importing from a file: If your data is in a CSV (Comma Separated Values) or other compatible file format, you can import it directly into Excel using the "Data" tab and selecting "From Text/CSV."
Creating the Titration Curve
With your data in Excel, creating the curve is straightforward:
-
Select the data: Highlight both columns of data (Volume and pH).
-
Insert a chart: Go to the "Insert" tab and choose a "Scatter" chart. Specifically, choose the "Scatter with Smooth Lines and Markers" option. This will create a visually appealing and informative curve. Other scatter chart options are also acceptable depending on your preferences.
-
Customize the chart: Now, let's customize the chart to improve its clarity and professionalism:
- Add Chart Title: Click on the chart title and rename it to something descriptive, such as "Titration Curve of Strong Acid with Strong Base".
- Axis Labels: Double-click on the axis labels to rename them to "Volume of Titrant (mL)" and "pH".
- Legend (if needed): If you have multiple datasets on the same chart, make sure the legend is clear and informative.
- Data Labels (optional): Consider adding data labels to highlight key points on the curve, particularly the equivalence point. This can be done by right-clicking on the data points and selecting "Add Data Labels".
- Gridlines: Adding major and minor gridlines can enhance readability, especially when determining the equivalence point visually.
Determining the Equivalence Point
The equivalence point is a crucial aspect of the titration curve. It represents the point at which the moles of titrant added equal the moles of analyte (the substance being titrated). There are several ways to determine this in Excel:
-
Visual Inspection: This is the simplest method. Look for the steepest part of the curve – the point of maximum slope. This point approximately represents the equivalence point. However, this method can be subjective and less accurate for less pronounced curves.
-
First Derivative: A more accurate method involves calculating the first derivative of the pH with respect to the volume. The point of maximum slope corresponds to the equivalence point, which will have the maximum value of the first derivative. This can be done directly in Excel using the following steps:
-
Calculate the change in pH: Add a new column next to the pH data, and calculate the change in pH between consecutive data points using the formula
=(B2-B1)(assuming your pH data starts in cell B1). -
Calculate the change in volume: Add another column next to the change in pH, and calculate the change in volume between consecutive data points using the formula
=(A2-A1)(assuming your volume data starts in cell A1). -
Calculate the first derivative: Add a final column and calculate the first derivative by dividing the change in pH by the change in volume using the formula
=C2/D2(assuming your change in pH is in column C and change in volume is in column D). -
Find the maximum value: Use the
MAXfunction in Excel to find the maximum value of the first derivative. The corresponding volume will be your approximate equivalence point.
-
-
Second Derivative: An even more precise method involves calculating the second derivative. The equivalence point is indicated by the point where the second derivative crosses zero. This method requires an additional calculation step similar to the first derivative but involves finding the difference in the first derivatives. This method is useful for complex curves with less clear inflection points.
Enhancing Your Titration Curve
Beyond the basic plot, you can further enhance your titration curve for better presentation and analysis:
-
Adding a Trendline: Excel's trendline feature can help to visually represent the overall shape of your data, particularly useful for approximating the equivalence point, although it’s not as precise as the derivative methods. You can choose different trendline types (linear, polynomial, exponential, etc.) to best fit your data.
-
Adding Error Bars: If you have error associated with your measurements (like uncertainty in pH readings), you can represent it visually using error bars. This improves the reliability and trustworthiness of your plot.
-
Multiple Titrations: If you have data from multiple titrations, you can overlay the curves on the same graph for comparison. This allows for evaluating the reproducibility of your experiment. To do this, simply select all your data (all volume and pH columns) and follow the chart creation steps as before. Excel will automatically create a separate line for each data set.
-
Exporting your Chart: Once satisfied, you can export your chart as a high-resolution image (PNG, JPG) or embed it directly into a document or presentation.
Troubleshooting Common Issues
-
Incorrect Data Entry: Double-check your data for any typos or inconsistencies. A small error in your data can drastically alter the appearance of the curve.
-
Chart Type: Make sure you are using the appropriate scatter chart type for a smooth line.
-
Axis Scaling: Adjust the axis scales as needed to ensure that all data points are clearly visible and the curve is properly displayed.
Conclusion
Creating a titration curve in Excel is a powerful tool for chemists and anyone working with titration data. By following the steps outlined above, you can generate accurate, professional-looking graphs that aid in interpreting your results, finding the equivalence point, and presenting your findings effectively. Remember, the accuracy of your curve is directly dependent on the accuracy of your experimental data. Using Excel's analytical features enhances the precision and ease of determining the crucial equivalence point. Mastering these techniques will streamline your analysis and contribute to more robust scientific findings.
Latest Posts
Latest Posts
-
What Are The Boiling And Freezing Points Of Water
Apr 06, 2025
-
Similarities Of Plant And Animal Cell
Apr 06, 2025
-
How Do You Determine The Species Of An Unknown Bacterium
Apr 06, 2025
-
If An Atom Gains An Electron It Becomes
Apr 06, 2025
-
Bartolome De Las Casas Describes The Exploitation Of Indigenous Peoples
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How To Make A Titration Curve In Excel . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
