Are Proteins Polymers Of Amino Acids
Muz Play
Apr 06, 2025 · 7 min read
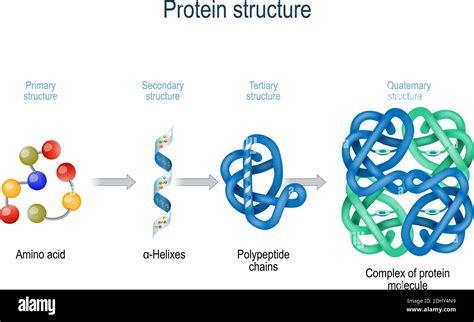
Table of Contents
Are Proteins Polymers of Amino Acids? A Deep Dive into the Structure and Function of Proteins
Proteins are the workhorses of life, involved in virtually every biological process imaginable. From catalyzing reactions as enzymes to providing structural support as in collagen, proteins perform a vast array of functions essential for the survival and thriving of all living organisms. But what are these remarkable molecules actually made of? The answer lies in their fundamental building blocks: amino acids. The question "Are proteins polymers of amino acids?" is a resounding yes, and this article will delve deep into the fascinating relationship between these monomers and the complex macromolecules they create.
Understanding Amino Acids: The Monomers of Proteins
Before exploring the polymeric nature of proteins, it's crucial to understand the individual components: amino acids. Amino acids are organic molecules containing a central carbon atom (the alpha carbon) bonded to four distinct groups:
- An amino group (-NH₂): This group is basic and imparts the characteristic "amino" property.
- A carboxyl group (-COOH): This group is acidic and contributes to the overall acidic nature of amino acids.
- A hydrogen atom (-H): This is a simple hydrogen atom.
- A side chain (R group): This is the variable group that distinguishes one amino acid from another. The R group can be as simple as a hydrogen atom (as in glycine) or complex and bulky (as in tryptophan). The properties of the R group (hydrophobic, hydrophilic, charged, etc.) significantly influence the protein's overall structure and function.
There are 20 standard amino acids that are genetically encoded and used by ribosomes to synthesize proteins. These 20 amino acids possess different R groups, which provide a diverse range of chemical properties and functionalities. This diversity is critical to the vast array of functions proteins perform.
The Importance of the R Group
The R group is the key to understanding the diversity of amino acids. Some R groups are nonpolar and hydrophobic (water-fearing), while others are polar and hydrophilic (water-loving). Some R groups are even charged, either positively or negatively. These different properties determine how amino acids interact with each other and their environment within a protein. For instance, hydrophobic R groups tend to cluster together in the interior of a protein, away from the aqueous environment of the cell, while hydrophilic R groups often reside on the protein's surface.
Peptide Bonds: Linking Amino Acids Together
The formation of proteins involves the joining of amino acids through a specific type of covalent bond called a peptide bond. This bond forms between the carboxyl group (-COOH) of one amino acid and the amino group (-NH₂) of another. This reaction releases a water molecule (H₂O), a process known as dehydration synthesis or condensation. The resulting bond is an amide bond, specifically called a peptide bond in the context of protein synthesis.
The Peptide Backbone
The repeating sequence of N-C-C-N-C-C… formed by the peptide bonds creates the peptide backbone of the protein. The side chains (R groups) project outwards from the backbone, and their interactions determine the protein's three-dimensional structure. A chain of amino acids linked by peptide bonds is called a polypeptide. Proteins can consist of one or more polypeptide chains.
Levels of Protein Structure: From Primary to Quaternary
The intricate three-dimensional structures of proteins are crucial to their function. These structures are hierarchical and can be categorized into four levels:
1. Primary Structure: The Amino Acid Sequence
The primary structure of a protein is simply the linear sequence of amino acids in a polypeptide chain. This sequence is dictated by the genetic code encoded in DNA. Even a single amino acid substitution can drastically alter a protein's structure and function, as famously illustrated by sickle cell anemia, a disease caused by a single amino acid change in the hemoglobin protein.
2. Secondary Structure: Local Folding Patterns
The primary structure folds into local, regular patterns called secondary structures. These patterns are stabilized by hydrogen bonds between the atoms of the peptide backbone. The two most common secondary structures are:
- Alpha-helices: These are right-handed coiled structures stabilized by hydrogen bonds between the carbonyl oxygen of one amino acid and the amide hydrogen of an amino acid four residues down the chain.
- Beta-sheets: These are flat, extended structures formed by hydrogen bonds between adjacent polypeptide chains (or segments of the same chain) arranged side-by-side. Beta-sheets can be parallel (chains running in the same direction) or antiparallel (chains running in opposite directions).
3. Tertiary Structure: The Overall 3D Shape
The tertiary structure represents the overall three-dimensional arrangement of a polypeptide chain. It's stabilized by a variety of interactions between the R groups of the amino acids, including:
- Hydrophobic interactions: Hydrophobic R groups cluster together in the protein's interior, away from water.
- Hydrogen bonds: Hydrogen bonds form between polar R groups.
- Ionic bonds (salt bridges): Ionic bonds form between oppositely charged R groups.
- Disulfide bonds: Covalent bonds between cysteine residues (amino acids containing a sulfhydryl group) create strong cross-links within the protein.
The tertiary structure is what determines the protein's unique shape and function. A protein's active site, the region where it interacts with other molecules, is a crucial aspect of its tertiary structure.
4. Quaternary Structure: Multiple Polypeptide Chains
Some proteins consist of multiple polypeptide chains (subunits) interacting together. The arrangement of these subunits forms the quaternary structure. The subunits are held together by the same types of interactions that stabilize tertiary structure: hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bonds. Hemoglobin, for example, is a tetramer (four subunits) with a quaternary structure.
The Significance of Protein Polymerization
The polymerization of amino acids into proteins is a fundamental process in biology. The specific sequence of amino acids, dictated by the genetic code, determines the protein's primary structure, which in turn influences the higher-order structures and ultimately, the protein's function. This remarkable precision is a testament to the sophistication of biological systems. Errors in this process can lead to misfolded proteins, which can have severe consequences, contributing to diseases such as Alzheimer's and Parkinson's.
Examples of Proteins and Their Functions
The diverse functions of proteins are a direct consequence of the vast array of possible amino acid sequences and the resulting three-dimensional structures. Here are a few examples:
- Enzymes: These biological catalysts accelerate biochemical reactions by lowering the activation energy. Examples include digestive enzymes like amylase and lipase.
- Structural proteins: These proteins provide support and structure to cells and tissues. Collagen, a major component of connective tissue, and keratin, a key protein in hair and nails, are examples.
- Transport proteins: These proteins facilitate the movement of molecules across cell membranes. Hemoglobin, which transports oxygen in the blood, is a classic example.
- Hormones: These signaling molecules regulate various physiological processes. Insulin, which regulates blood sugar levels, is an example.
- Antibodies: These proteins, part of the immune system, recognize and neutralize foreign invaders like bacteria and viruses.
- Motor proteins: These proteins generate movement within cells and tissues. Myosin, involved in muscle contraction, is a key example.
Conclusion: Proteins – Masterpieces of Polymerization
The statement "Proteins are polymers of amino acids" is not merely a statement of fact; it's a foundational concept in biochemistry. The intricate process of amino acid polymerization, guided by the genetic code, results in the creation of diverse and highly functional macromolecules essential for life. Understanding the relationship between amino acid sequence, protein structure, and function is crucial for comprehending the complexity and beauty of biological systems. From the simplest single-celled organism to the most complex multicellular life forms, proteins, these remarkable polymers of amino acids, are central players in the drama of life. Further research continues to unravel the mysteries of protein folding, function, and malfunction, leading to potential advancements in medicine, biotechnology, and materials science.
Latest Posts
Latest Posts
-
An Activity Series Of Metals Orders Metals By Their
Apr 06, 2025
-
The Study Of Matter And Its Changes
Apr 06, 2025
-
Find The Limit Of Trigonometric Functions
Apr 06, 2025
-
How Did Lincolns Assassination Affect Reconstruction
Apr 06, 2025
-
What Elements Cycle Between Living And Nonliving
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Are Proteins Polymers Of Amino Acids . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
