Arrhenius Acid Vs Bronsted Lowry Acid
Muz Play
Apr 02, 2025 · 6 min read
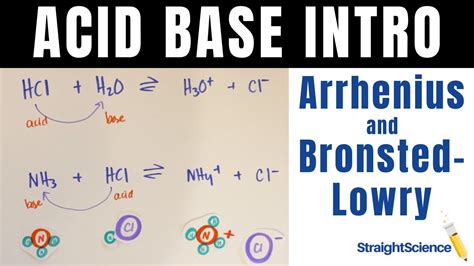
Table of Contents
Arrhenius Acid vs. Brønsted-Lowry Acid: A Comprehensive Comparison
Understanding the nature of acids is fundamental to chemistry. While seemingly straightforward, the definition of an acid has evolved over time, leading to different theoretical frameworks. This article delves into the nuances of two prominent acid definitions: the Arrhenius definition and the Brønsted-Lowry definition. We'll explore their similarities, key differences, limitations, and the broader implications of each approach in understanding acid-base chemistry.
The Arrhenius Definition: A Historical Perspective
The Arrhenius definition, proposed by Svante Arrhenius in 1884, was a groundbreaking contribution to acid-base chemistry. It defined acids and bases based on their behavior in aqueous solutions:
-
Arrhenius Acid: An Arrhenius acid is a substance that increases the concentration of hydrogen ions (H⁺) when dissolved in water. The hydrogen ion, often represented as H⁺, is more accurately described as a proton (H⁺) because it lacks an electron. Strong Arrhenius acids, like hydrochloric acid (HCl) and sulfuric acid (H₂SO₄), completely dissociate in water, releasing all their hydrogen ions. Weak Arrhenius acids, such as acetic acid (CH₃COOH), only partially dissociate.
-
Arrhenius Base: An Arrhenius base is a substance that increases the concentration of hydroxide ions (OH⁻) when dissolved in water. Sodium hydroxide (NaOH) and potassium hydroxide (KOH) are examples of strong Arrhenius bases, while ammonia (NH₃) is a weak Arrhenius base.
Example: When hydrochloric acid (HCl) dissolves in water, it dissociates completely into hydrogen ions (H⁺) and chloride ions (Cl⁻):
HCl(aq) → H⁺(aq) + Cl⁻(aq)
The Brønsted-Lowry Definition: A Broader Scope
The Brønsted-Lowry definition, independently proposed by Johannes Nicolaus Brønsted and Thomas Martin Lowry in 1923, offered a more comprehensive and versatile approach. This definition broadened the understanding of acids and bases beyond the constraints of aqueous solutions:
-
Brønsted-Lowry Acid: A Brønsted-Lowry acid is a substance that donates a proton (H⁺) to another substance. This definition doesn't require the presence of water; the proton transfer can occur in any solvent or even in the gas phase.
-
Brønsted-Lowry Base: A Brønsted-Lowry base is a substance that accepts a proton (H⁺) from another substance.
Example: Consider the reaction between hydrochloric acid (HCl) and ammonia (NH₃):
HCl(g) + NH₃(g) → NH₄⁺(g) + Cl⁻(g)
In this gas-phase reaction, HCl acts as a Brønsted-Lowry acid by donating a proton to NH₃, which acts as a Brønsted-Lowry base by accepting the proton. This reaction doesn't involve water, highlighting the broader applicability of the Brønsted-Lowry definition.
Conjugate Acid-Base Pairs: A Key Concept in Brønsted-Lowry Theory
A crucial aspect of the Brønsted-Lowry definition is the concept of conjugate acid-base pairs. When an acid donates a proton, it forms its conjugate base, which is the species remaining after the proton is donated. Similarly, when a base accepts a proton, it forms its conjugate acid. In the HCl/NH₃ example above:
- HCl is the acid, and Cl⁻ is its conjugate base.
- NH₃ is the base, and NH₄⁺ is its conjugate acid.
Comparing Arrhenius and Brønsted-Lowry Definitions: Key Differences
While both definitions aim to describe acids and bases, there are fundamental differences:
| Feature | Arrhenius Definition | Brønsted-Lowry Definition |
|---|---|---|
| Solvent | Requires aqueous solutions | No solvent restriction; applies to various media |
| Acid Definition | Increases H⁺ concentration in water | Donates a proton (H⁺) |
| Base Definition | Increases OH⁻ concentration in water | Accepts a proton (H⁺) |
| Scope | Limited to aqueous solutions and specific ions | Broader scope; encompasses a wider range of reactions |
| Examples | HCl, H₂SO₄ (acids); NaOH, KOH (bases) | HCl, CH₃COOH (acids); NH₃, H₂O (bases) |
Limitations of the Arrhenius Definition
The Arrhenius definition, despite its historical significance, suffers from several limitations:
- Solvent Dependence: It is strictly limited to aqueous solutions, excluding acid-base reactions in other solvents or in the gas phase.
- Limited Scope: It fails to explain acid-base reactions that do not involve H⁺ and OH⁻ ions. For instance, it cannot explain the reaction between ammonia (NH₃) and boron trifluoride (BF₃), which involves a proton transfer but no hydroxide ions.
- Inadequate Explanation: It doesn't adequately explain the amphoteric nature of certain substances, like water, which can act as both an acid and a base.
The Advantages of the Brønsted-Lowry Definition
The Brønsted-Lowry definition overcomes many of the limitations of the Arrhenius definition:
- Universality: Its applicability extends beyond aqueous solutions, encompassing various solvents and reaction media.
- Broader Scope: It encompasses a wider range of acid-base reactions, including those that don't involve hydroxide ions.
- Amphoteric Behavior: It effectively explains the amphoteric behavior of substances like water, which can donate or accept protons.
- Conjugate Acid-Base Pairs: The introduction of conjugate acid-base pairs provides a more comprehensive understanding of acid-base equilibrium.
Beyond Brønsted-Lowry: The Lewis Definition
Even the Brønsted-Lowry definition has limitations. It doesn't account for acid-base reactions that don't involve proton transfer. This led to the development of the Lewis definition of acids and bases, proposed by Gilbert N. Lewis.
- Lewis Acid: A Lewis acid is a substance that accepts an electron pair.
- Lewis Base: A Lewis base is a substance that donates an electron pair.
The Lewis definition is the most general and encompasses the Brønsted-Lowry definition as a subset. Many reactions considered acid-base reactions according to the Lewis definition are not considered such according to the Arrhenius or Brønsted-Lowry definitions.
Practical Applications and Significance
The understanding of acids and bases, particularly through the lens of the Brønsted-Lowry definition, has profound implications across various fields:
- Analytical Chemistry: Titrations, a fundamental analytical technique, rely heavily on the principles of acid-base chemistry. Understanding acid-base equilibria is crucial for accurate quantitative analysis.
- Biochemistry: Many biological processes depend on acid-base reactions. Enzymes, for instance, often require specific pH conditions to function optimally. Buffers, solutions that resist changes in pH, are crucial for maintaining physiological pH levels in biological systems.
- Environmental Science: Acid rain, a significant environmental problem, involves the deposition of acidic substances (like sulfuric and nitric acids) from the atmosphere, impacting ecosystems and infrastructure.
- Industrial Chemistry: Many industrial processes, such as the production of fertilizers and pharmaceuticals, utilize acid-base reactions. Understanding acid-base catalysis is crucial for efficient and controlled chemical synthesis.
Conclusion
The journey from the Arrhenius definition to the Brønsted-Lowry and subsequently the Lewis definition reflects the progressive refinement of our understanding of acid-base chemistry. While the Arrhenius definition provided a crucial starting point, the Brønsted-Lowry definition offered a significantly expanded and more versatile framework for comprehending acid-base reactions. The concepts of proton transfer, conjugate acid-base pairs, and the broader applicability of the Brønsted-Lowry theory are essential for a comprehensive grasp of acid-base chemistry and its far-reaching applications in diverse scientific and technological fields. The limitations of each definition ultimately highlight the iterative nature of scientific progress and the continuous refinement of our understanding of fundamental chemical concepts. Understanding these different definitions allows for a more complete and nuanced understanding of the complex world of acid-base chemistry.
Latest Posts
Latest Posts
-
Difference Between Systemic And Pulmonary Circulation
Apr 03, 2025
-
Amino Acids Can Be Distinguished From One Another By
Apr 03, 2025
-
Balancing Equations Practice Worksheet With Answers
Apr 03, 2025
-
How Does The Predator Prey Relationship Affect A Population
Apr 03, 2025
-
The Axons Of Parasympathetic Postganglionic Neurons Are
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Arrhenius Acid Vs Bronsted Lowry Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
