Boiling Point Is A Chemical Property
Muz Play
Apr 03, 2025 · 7 min read
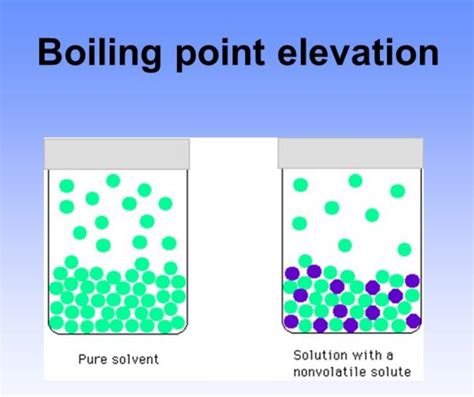
Table of Contents
Boiling Point: A Fundamental Chemical Property
Boiling point, a seemingly simple concept, is actually a rich and crucial aspect of chemistry. It's not merely a number on a datasheet; it's a fundamental chemical property reflecting the intricate interplay of intermolecular forces and molecular structure. Understanding boiling point offers insight into a substance's identity, purity, and behavior under various conditions. This article delves deep into the nature of boiling point, exploring its definition, influencing factors, applications, and importance in diverse scientific fields.
Defining Boiling Point: More Than Just a Temperature
The boiling point of a liquid is defined as the temperature at which its vapor pressure equals the surrounding atmospheric pressure. This means that at the boiling point, the liquid's molecules possess enough kinetic energy to overcome the intermolecular forces holding them together, transitioning into the gaseous phase. It's important to emphasize the dependence on atmospheric pressure: a liquid boils at a lower temperature at higher altitudes (lower atmospheric pressure) and vice versa. Standard boiling point refers to the temperature at which a liquid boils at exactly 1 atmosphere (atm) of pressure.
Vapor Pressure: The Driving Force Behind Boiling
Understanding vapor pressure is key to grasping the concept of boiling point. Vapor pressure is the pressure exerted by the vapor of a liquid in a closed system at equilibrium. This equilibrium is established when the rate of evaporation (liquid turning into gas) equals the rate of condensation (gas turning back into liquid). As temperature increases, more molecules have enough energy to escape the liquid phase, leading to a higher vapor pressure. When the vapor pressure equals the external pressure, boiling occurs.
Factors Influencing Boiling Point: A Molecular Perspective
The boiling point of a substance is intrinsically linked to its molecular structure and the strength of intermolecular forces. Several factors play significant roles in determining a substance's boiling point:
1. Intermolecular Forces: The Glue Holding Molecules Together
The strength of intermolecular forces is the dominant factor influencing boiling point. Stronger intermolecular forces require more energy to overcome, resulting in a higher boiling point. These forces include:
-
London Dispersion Forces (LDFs): Present in all molecules, these weak forces arise from temporary fluctuations in electron distribution. Larger molecules with more electrons generally have stronger LDFs and higher boiling points.
-
Dipole-Dipole Interactions: Occur in polar molecules where there's a permanent separation of charge. The stronger the dipole moment, the stronger the dipole-dipole interactions and the higher the boiling point.
-
Hydrogen Bonding: A special type of dipole-dipole interaction involving hydrogen bonded to a highly electronegative atom (like oxygen, nitrogen, or fluorine). Hydrogen bonding is exceptionally strong and leads to significantly higher boiling points compared to similar molecules lacking hydrogen bonds. For example, water (H₂O) has a remarkably high boiling point due to its extensive hydrogen bonding network.
2. Molecular Weight: Size Matters
Generally, larger molecules with higher molecular weights exhibit higher boiling points. This is primarily due to the increased strength of London Dispersion Forces. Larger molecules have a larger surface area and more electrons, leading to stronger temporary dipoles and stronger LDFs.
3. Molecular Shape: Geometry Plays a Role
Molecular shape can influence the extent of intermolecular interactions. Linear molecules tend to pack more closely together, leading to stronger interactions and higher boiling points compared to branched molecules of the same molecular weight. This is because branched molecules have less surface area in contact with neighboring molecules.
4. Polarity: The Impact of Charge Distribution
Polar molecules, with uneven charge distribution, experience stronger intermolecular forces (dipole-dipole interactions) than nonpolar molecules. This leads to higher boiling points for polar substances. The polarity of a molecule can be predicted based on its molecular geometry and the electronegativity of its constituent atoms.
Applications of Boiling Point: A Versatile Chemical Property
Boiling point is a crucial property with widespread applications across various fields:
1. Purification and Separation: Distillation
Distillation, a widely used separation technique, relies on the difference in boiling points of components in a mixture. By carefully controlling the temperature, one can selectively vaporize and condense components with different boiling points, leading to their separation. This is crucial in many industrial processes, such as the refining of petroleum and the production of alcoholic beverages.
2. Identification of Substances: A Fingerprinting Tool
Boiling point is a characteristic physical property that helps in identifying unknown substances. By determining the boiling point of an unknown compound and comparing it to known values, chemists can often identify the substance. This is often used in conjunction with other analytical techniques for a definitive identification.
3. Monitoring Purity: Assessing Substance Integrity
The boiling point of a pure substance is usually sharp and well-defined. Impurities often broaden the boiling point range, indicating the presence of contaminants. This property allows chemists to assess the purity of a substance and monitor the effectiveness of purification processes.
4. Reaction Monitoring: Observing Chemical Changes
In certain chemical reactions, the boiling point of the reactants or products can be monitored to track the progress of the reaction or identify the formation of specific compounds. This allows for real-time monitoring and optimization of reaction conditions.
5. Safety and Handling: Predicting Substance Behavior
Knowledge of a substance's boiling point is crucial for safety and handling procedures. Understanding its boiling point helps determine appropriate storage conditions, avoiding potential hazards associated with volatile substances.
Boiling Point and Other Chemical Properties: Interdependence and Context
Boiling point doesn't exist in isolation; it's intricately connected to other chemical properties. For instance, its relationship with melting point provides insights into the overall strength of intermolecular forces. Substances with strong intermolecular forces usually exhibit both high melting and boiling points. Similarly, boiling point is closely related to vapor pressure, as explained earlier – a high vapor pressure indicates a low boiling point and vice versa. Solubility can also be related, although not directly proportional. Polar substances with high boiling points often dissolve well in polar solvents, while nonpolar substances with low boiling points are more soluble in nonpolar solvents.
Beyond the Basics: Advanced Concepts and Applications
While the fundamental concepts of boiling point are relatively straightforward, advanced applications and concepts offer further insights:
1. Clausius-Clapeyron Equation: Predicting Boiling Point at Different Pressures
The Clausius-Clapeyron equation is a powerful tool used to predict how the boiling point changes with alterations in atmospheric pressure. It establishes a mathematical relationship between vapor pressure, temperature, and the enthalpy of vaporization (the energy required to transform a liquid into a gas). This equation is invaluable in applications such as high-altitude cooking, where the lower atmospheric pressure leads to a lower boiling point of water.
2. Azeotropes: Mixtures with Constant Boiling Points
Azeotropes are mixtures of liquids that have a constant boiling point and composition throughout the distillation process. They cannot be separated by simple distillation because the vapor phase has the same composition as the liquid phase. Understanding azeotropes is critical in chemical engineering and industrial separations.
3. Superheating and Nucleation: Beyond Normal Boiling
Under specific conditions, a liquid can be heated above its boiling point without actually boiling. This phenomenon, known as superheating, occurs when there are no nucleation sites (tiny imperfections or impurities) for the formation of vapor bubbles. Once a nucleation site is introduced, rapid and often violent boiling occurs.
4. Boiling Point Elevation and Freezing Point Depression: Colligative Properties
Boiling point elevation and freezing point depression are colligative properties, meaning they depend on the concentration of solute particles in a solution, not on the identity of the solute. Adding a non-volatile solute to a solvent raises its boiling point and lowers its freezing point. This principle has numerous applications, including antifreeze formulations and the determination of molar mass.
Conclusion: Boiling Point – A Window into Molecular Interactions
Boiling point, while seemingly a simple numerical value, serves as a powerful window into the molecular world. It reveals crucial information about intermolecular forces, molecular structure, and the behavior of substances under various conditions. Its applications are vast and span diverse scientific and industrial domains. A thorough understanding of boiling point and its underlying principles is fundamental to numerous fields, from chemical analysis and synthesis to industrial processing and environmental science. By appreciating its complexity and significance, we unlock a deeper understanding of the intricate nature of matter and its transformations.
Latest Posts
Latest Posts
-
Identify The Features Associated With Waxes
Apr 04, 2025
-
Rna Differs From Dna In That Rna
Apr 04, 2025
-
What Is The Net Change In Math
Apr 04, 2025
-
Why Cant The Subscripts Be Changed In A Chemical Equation
Apr 04, 2025
-
How Many Bonds Does Boron Form
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Boiling Point Is A Chemical Property . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
