Classify The Sugars As Aldoses Or Ketoses
Muz Play
Apr 03, 2025 · 6 min read
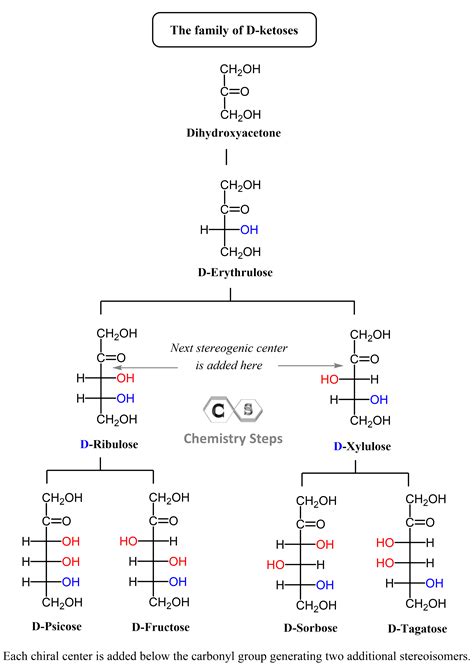
Table of Contents
Classifying Sugars: Aldoses vs. Ketoses – A Comprehensive Guide
Sugars, the simplest forms of carbohydrates, are fundamental to life, providing energy and forming structural components within organisms. Understanding their classification is crucial to comprehending their diverse roles in biological processes. This comprehensive guide delves into the classification of sugars as either aldoses or ketoses, exploring their structural differences, chemical properties, and biological significance.
The Foundation: Carbonyl Groups and Sugar Classification
The primary determinant in classifying a sugar as an aldose or ketose lies in the location of its carbonyl group—a functional group containing a carbon atom double-bonded to an oxygen atom. This seemingly small detail dictates the sugar's reactivity and overall chemical behavior.
Aldoses: The Aldehyde Sugars
Aldoses are monosaccharides (simple sugars) that possess an aldehyde group (–CHO) at the end of their carbon chain. The aldehyde group is characterized by the carbonyl carbon being bonded to a hydrogen atom and another carbon atom. This structural feature renders aldoses highly reactive, participating in various chemical reactions essential for metabolic pathways.
Examples of Aldoses:
- Glyceraldehyde: The simplest aldose, with three carbon atoms. It's a crucial intermediate in glycolysis.
- Erythrose: A four-carbon aldose involved in the pentose phosphate pathway.
- Ribose: A five-carbon aldose, a structural component of RNA and ATP.
- Glucose: A six-carbon aldose, the primary energy source for many organisms.
- Galactose: A six-carbon aldose, often found in lactose (milk sugar).
- Mannose: A six-carbon aldose, found in certain glycoproteins.
Ketoses: The Ketone Sugars
Ketoses, conversely, contain a ketone group (–C=O) within their carbon chain. The ketone group’s carbonyl carbon is bonded to two other carbon atoms. While sharing some similarities with aldoses, ketoses exhibit distinct chemical properties due to the internal location of their carbonyl group. This subtle difference leads to variations in reactivity and metabolic roles.
Examples of Ketoses:
- Dihydroxyacetone: The simplest ketose, with three carbon atoms. An intermediate in glycolysis.
- Fructose: A six-carbon ketose, found in fruits and honey, commonly known as fruit sugar. It's quickly metabolized.
- Xylulose: A five-carbon ketose involved in the pentose phosphate pathway.
- Sorbose: A six-carbon ketose, used in the industrial production of vitamin C (ascorbic acid).
Delving Deeper: Structural Isomerism and Sugar Diversity
The classification of sugars extends beyond simply identifying the carbonyl group's position. Isomerism plays a significant role in diversifying the types of aldoses and ketoses. Isomers are molecules with the same chemical formula but different structural arrangements. This leads to numerous variations within each aldose and ketose category.
Stereoisomerism: Chiral Centers and Enantiomers
Many sugars contain chiral centers – carbon atoms bonded to four different groups. This chirality results in stereoisomers, molecules with the same connectivity but different spatial arrangements. Enantiomers are a type of stereoisomer that are mirror images of each other, non-superimposable. The presence of multiple chiral centers significantly increases the number of possible stereoisomers. For example, glucose has four chiral centers, leading to a total of 16 possible stereoisomers.
Epimers: Subtle Differences, Significant Impacts
Epimers are a specific type of diastereomer (stereoismers that are not mirror images) that differ in the configuration around only one chiral center. For instance, glucose and galactose are epimers, differing only at the C4 carbon. This seemingly small difference impacts their biological roles and metabolic pathways. Glucose is a primary energy source, while galactose must be converted to glucose before it can be utilized for energy.
Chemical Properties: Reactions Distinguishing Aldoses and Ketoses
The differing positions of the carbonyl group in aldoses and ketoses lead to distinct chemical reactivity. These differences are exploited in various analytical techniques to identify and distinguish between these sugar types.
Oxidation and Reduction Reactions: A Key Differentiator
Aldoses, with their terminal aldehyde group, are easily oxidized, meaning they readily lose electrons. This property forms the basis of many chemical tests, such as Benedict's test and Fehling's test, which are used to detect the presence of reducing sugars (sugars capable of reducing other substances). Ketoses, while capable of reduction, are generally less readily oxidized than aldoses. However, under specific conditions (e.g., in alkaline solutions), ketoses can undergo isomerization to form aldoses, subsequently allowing them to participate in oxidation reactions.
Formation of Glycosides: Linking Sugars Together
Both aldoses and ketoses can participate in glycoside formation. This reaction involves the reaction of the carbonyl group with an alcohol, resulting in a glycosidic bond and the formation of a glycoside. Glycosidic bonds are crucial for the formation of disaccharides (such as sucrose and lactose) and polysaccharides (such as starch and cellulose). The nature of the glycosidic bond (α or β) influences the properties and biological function of the resulting carbohydrate.
Biological Significance: The Diverse Roles of Aldoses and Ketoses
Aldoses and ketoses play diverse and crucial roles in biological systems, underpinning many vital processes.
Aldoses: Energy and Structure
- Glucose: The quintessential energy source for most organisms. Its metabolism through glycolysis and the citric acid cycle generates ATP, the primary cellular energy currency.
- Ribose: A crucial component of RNA, the molecule responsible for protein synthesis. Also, a key structural element in ATP and other nucleotides.
- Galactose: A component of lactose (milk sugar), crucial for infant nutrition. It’s also found in glycolipids and glycoproteins.
Ketoses: Metabolic Intermediates and Sweetness
- Fructose: A significant energy source, commonly found in fruits and honey. It's rapidly metabolized in the liver. Its high sweetness makes it widely used in the food industry.
- Dihydroxyacetone: A crucial intermediate in glycolysis, the central metabolic pathway for glucose breakdown.
- Xylulose: Plays a role in the pentose phosphate pathway, a metabolic route that generates NADPH (a reducing agent) and precursors for nucleotide biosynthesis.
Analytical Techniques: Identifying Aldoses and Ketoses
Several analytical methods are used to identify and quantify aldoses and ketoses in various samples.
- Chromatography: Techniques like thin-layer chromatography (TLC) and high-performance liquid chromatography (HPLC) separate sugars based on their polarity and other physical properties, allowing for identification and quantification.
- Spectroscopy: Techniques like nuclear magnetic resonance (NMR) spectroscopy provide detailed structural information about sugars, confirming their identity as aldoses or ketoses.
- Colorimetric assays: Tests like Benedict's test and Fehling's test exploit the reducing properties of aldoses (and indirectly ketoses) to provide a qualitative indication of their presence.
Conclusion: A Fundamental Classification with Far-Reaching Implications
The classification of sugars into aldoses and ketoses is a fundamental concept in biochemistry. The seemingly small difference in the position of the carbonyl group profoundly impacts their chemical properties and biological functions. Understanding this classification is key to comprehending the diverse roles of sugars in energy metabolism, structural components, and numerous other biological processes. From the simple sugars like glucose and fructose to complex carbohydrates like starch and cellulose, the distinction between aldoses and ketoses remains a cornerstone of carbohydrate chemistry and biology. Further research into the complexities of sugar chemistry continues to uncover new facets of their vital contributions to life itself.
Latest Posts
Latest Posts
-
Are Ion Channels Active Or Passive
Apr 04, 2025
-
Prokaryotes That Obtain Their Energy From Chemical Compounds Are Called
Apr 04, 2025
-
What Does High Absorbance Mean In Spectrophotometry
Apr 04, 2025
-
Electric Field Due To A Disk Of Charge
Apr 04, 2025
-
Compare And Contrast Skeletal Smooth And Cardiac Muscle
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Classify The Sugars As Aldoses Or Ketoses . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
