Do Cations Lose Or Gain Electrons
Muz Play
Apr 04, 2025 · 5 min read
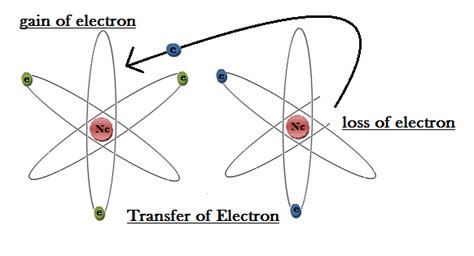
Table of Contents
Do Cations Lose or Gain Electrons? Understanding Ionic Bonding
The question of whether cations lose or gain electrons is fundamental to understanding chemistry, particularly the concept of ionic bonding. The short answer is that cations lose electrons. This seemingly simple statement, however, opens the door to a deeper exploration of atomic structure, electron configuration, and the forces that drive chemical reactions. This article will delve into this topic, examining the process of cation formation, its implications for chemical properties, and its broader context within the world of chemistry.
What are Cations?
Before we discuss electron transfer, let's define the term "cation." A cation is a positively charged ion. This positive charge arises from the loss of one or more electrons from a neutral atom. The loss of negatively charged electrons leaves behind a net positive charge on the atom, transforming it into a cation. This process is crucial in the formation of ionic compounds, which are held together by the electrostatic attraction between positively charged cations and negatively charged anions (negatively charged ions).
The Role of Electron Configuration
The tendency of an atom to lose or gain electrons is largely dictated by its electron configuration. Atoms are most stable when their outermost electron shell, also known as the valence shell, is full. For many elements, this means having eight electrons in their valence shell (the octet rule). Atoms that have relatively few electrons in their valence shell tend to lose these electrons to achieve a stable electron configuration, becoming cations in the process.
For example, consider sodium (Na), an alkali metal with an atomic number of 11. Its electron configuration is 1s²2s²2p⁶3s¹. Sodium has one electron in its outermost shell (3s¹). It's energetically favorable for sodium to lose this single electron, resulting in a stable configuration matching that of neon (1s²2s²2p⁶), a noble gas with a full valence shell. This loss of an electron transforms the neutral sodium atom into a sodium cation, Na⁺.
The Process of Cation Formation: Ionization Energy
The energy required to remove an electron from a neutral atom is called the ionization energy. This energy is a measure of how strongly an atom holds onto its electrons. Atoms with low ionization energies readily lose electrons to form cations. This is typically observed in metals, particularly those on the left side of the periodic table (alkali metals and alkaline earth metals).
The first ionization energy is the energy required to remove the first electron, the second ionization energy to remove the second electron, and so on. Each successive ionization energy is generally higher than the previous one, as removing an electron from a positively charged ion requires overcoming the stronger electrostatic attraction between the positive ion and the remaining electrons.
Examples of Cation Formation
Numerous elements readily form cations. Here are some prominent examples:
- Group 1 (Alkali Metals): These elements, such as lithium (Li), sodium (Na), potassium (K), readily lose one electron to form +1 cations (Li⁺, Na⁺, K⁺).
- Group 2 (Alkaline Earth Metals): These elements, such as beryllium (Be), magnesium (Mg), calcium (Ca), typically lose two electrons to form +2 cations (Be²⁺, Mg²⁺, Ca²⁺).
- Transition Metals: Transition metals exhibit a greater variety in the number of electrons they can lose. For example, iron (Fe) can form both Fe²⁺ and Fe³⁺ cations, depending on the chemical environment.
- Post-transition metals: Elements like aluminum (Al) typically lose three electrons to form Al³⁺ cations.
Cations in Ionic Compounds
The formation of cations is crucial in the formation of ionic compounds. These compounds are formed through the electrostatic attraction between cations and anions. The opposite charges of the ions create a strong ionic bond, resulting in a stable crystalline structure. For example, the common table salt, sodium chloride (NaCl), is formed from the electrostatic attraction between Na⁺ cations and Cl⁻ anions.
The properties of ionic compounds are significantly influenced by the presence of cations. For instance, the melting and boiling points of ionic compounds are typically high due to the strong electrostatic forces between the ions. They are also often soluble in water, as water molecules can interact with the ions and break the ionic bonds.
Contrasting Cation Formation with Anion Formation
It's important to contrast the formation of cations with that of anions. While cations lose electrons to achieve a stable electron configuration, anions gain electrons. Nonmetals, often located on the right side of the periodic table, tend to gain electrons to complete their valence shells. This gain of electrons results in a negative charge, forming an anion.
For example, chlorine (Cl), a halogen with an electron configuration of 1s²2s²2p⁶3s²3p⁵, readily gains one electron to achieve a stable octet, forming the chloride anion (Cl⁻).
Beyond the Octet Rule: Exceptions and Complications
While the octet rule provides a useful framework for understanding cation formation, there are exceptions. Some elements can form stable ions with less than or more than eight electrons in their valence shells. Transition metals, in particular, often form cations with incomplete valence shells. The complex interactions of d-orbitals and the influence of ligands (molecules or ions bound to the central metal ion) can lead to variations in the oxidation states and stability of transition metal cations.
Furthermore, the concept of effective nuclear charge plays a significant role in determining the ease with which an electron is lost. The effective nuclear charge is the net positive charge experienced by an electron in an atom, considering the shielding effect of other electrons. A higher effective nuclear charge means a stronger attraction to the nucleus, making it more difficult to remove an electron.
Conclusion: The Crucial Role of Cations in Chemistry
In conclusion, cations are positively charged ions formed by the loss of electrons from neutral atoms. This process is driven by the desire to achieve a stable electron configuration, often adhering to the octet rule. Cations play a vital role in the formation of ionic compounds, influencing their physical and chemical properties. Understanding cation formation requires considering the electron configuration, ionization energy, and the sometimes complex interactions that govern atomic behavior. This fundamental concept is essential for comprehending a wide range of chemical phenomena and is a cornerstone of chemical bonding and reactivity. The intricacies of cation formation, including exceptions to the octet rule and the influence of effective nuclear charge, highlight the richness and complexity of the world of chemistry.
Latest Posts
Latest Posts
-
When Does Dna Replication Occur In A Eukaryotic Cell
Apr 04, 2025
-
What Was Shown By Both Redis And Pasteurs Experiments
Apr 04, 2025
-
The Elements In Group 1 Are Called The
Apr 04, 2025
-
Proof Of The Inverse Function Theorem
Apr 04, 2025
-
Nonmetal Halogen Family Atomic Mass 35
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Do Cations Lose Or Gain Electrons . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
