Electrons In The Outermost Energy Level Of An Atom
Muz Play
Apr 05, 2025 · 7 min read
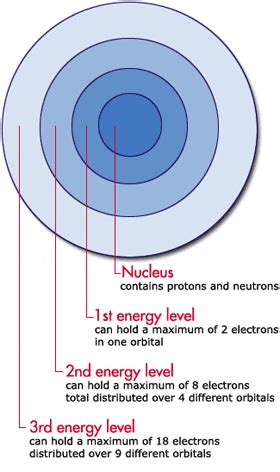
Table of Contents
Electrons in the Outermost Energy Level: Valence Electrons and Their Chemical Prowess
Electrons, the fundamental negatively charged particles within an atom, don't all behave the same. Their energy levels, dictated by their distance from the nucleus and the quantum mechanical rules governing their behavior, drastically influence their properties and, consequently, the atom's overall reactivity. Of particular importance are the valence electrons, those residing in the outermost energy level, also known as the valence shell. These electrons are the key players in chemical bonding, dictating how atoms interact with one another to form molecules and compounds. Understanding valence electrons is crucial to comprehending the nature of chemical reactions, material properties, and the very building blocks of the universe.
Understanding Electron Shells and Energy Levels
Before diving into the intricacies of valence electrons, it's crucial to establish a basic understanding of electron shells and energy levels. An atom's electrons are arranged in distinct energy levels or shells, each capable of holding a specific number of electrons. The energy levels are denoted by principal quantum numbers (n), where n=1 represents the shell closest to the nucleus (the lowest energy level), n=2 represents the next shell, and so on. The higher the value of n, the further the electron is from the nucleus and the higher its energy.
Each energy level is further subdivided into sublevels (s, p, d, f), each with its own unique shape and capacity for electrons. The s sublevel can hold a maximum of 2 electrons, the p sublevel can hold 6 electrons, the d sublevel can hold 10 electrons, and the f sublevel can hold 14 electrons.
The filling of these sublevels follows specific rules, most notably the Aufbau principle (electrons fill lower energy levels first) and Hund's rule (electrons fill orbitals individually before pairing up). These rules dictate the electron configuration of an atom, which is a representation of how its electrons are distributed among the various energy levels and sublevels.
Valence Electrons: The Chemical Architects
The outermost energy level, the valence shell, contains the valence electrons. These electrons are the furthest from the nucleus and experience the weakest attraction to the positively charged protons. This makes them the most easily influenced by external forces, such as the presence of other atoms. Their behavior dictates the atom's chemical properties, determining its reactivity and bonding capabilities.
The number of valence electrons an atom possesses is crucial in determining its chemical behavior. Atoms tend to react in ways that achieve a stable electron configuration, often resembling the electron configuration of a noble gas (Group 18 elements). This stability is often achieved through gaining, losing, or sharing electrons to attain a full valence shell (usually eight electrons, following the octet rule, though exceptions exist, particularly for elements with low atomic numbers).
The Octet Rule and its Exceptions
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer electron shell of eight electrons. This configuration provides exceptional stability, resembling the electronic structure of noble gases. While a powerful guiding principle, it's not without exceptions. Elements like hydrogen and lithium, with only one and three electrons respectively, strive for a duet (two electrons) in their valence shell, achieving stability resembling helium. Similarly, some heavier elements can expand their valence shell beyond eight electrons, accommodating more than the octet rule would predict.
Valence Electrons and Chemical Bonding
Valence electrons are directly responsible for chemical bonding, the forces that hold atoms together to form molecules and compounds. Three major types of chemical bonds involve valence electrons:
1. Ionic Bonds: The Transfer of Electrons
Ionic bonds form when one atom transfers one or more valence electrons to another atom. This transfer creates ions: positively charged cations (formed by losing electrons) and negatively charged anions (formed by gaining electrons). The electrostatic attraction between these oppositely charged ions constitutes the ionic bond. This type of bonding commonly occurs between metals (which tend to lose electrons easily) and nonmetals (which tend to gain electrons readily). Examples include NaCl (sodium chloride, table salt) where sodium (Na) loses one electron to chlorine (Cl).
2. Covalent Bonds: The Sharing of Electrons
Covalent bonds form when atoms share valence electrons to achieve a stable electron configuration. This sharing is often represented by lines connecting atoms in Lewis structures. These bonds are typically formed between nonmetals, which have similar electronegativities (a measure of an atom's ability to attract electrons). Examples include H₂ (hydrogen gas), where two hydrogen atoms share their single valence electron, and CH₄ (methane), where carbon shares its four valence electrons with four hydrogen atoms.
3. Metallic Bonds: A Sea of Electrons
Metallic bonds occur in metals, where valence electrons are delocalized, forming a "sea" of electrons that are not associated with any particular atom. This mobile electron sea explains the characteristic properties of metals, such as electrical and thermal conductivity, malleability, and ductility.
Valence Electrons and Periodicity
The periodic table is organized based on the electronic configurations of elements, with valence electrons playing a central role in defining an element's properties and its position on the table. Elements within the same group (vertical column) have the same number of valence electrons, resulting in similar chemical properties. For example, all alkali metals (Group 1) have one valence electron, making them highly reactive and prone to forming +1 ions.
The number of valence electrons increases across a period (horizontal row), reflecting the filling of the valence shell with electrons. This trend correlates with the gradual change in properties across a period. For example, electronegativity generally increases across a period as the number of valence electrons increases.
Determining the Number of Valence Electrons
Several methods can be used to determine the number of valence electrons in an atom:
- Using the Group Number (for main group elements): For main group elements (Groups 1-18), the group number often corresponds to the number of valence electrons. For example, elements in Group 14 have four valence electrons. However, this method doesn't apply to transition metals.
- Using the Electron Configuration: The electron configuration explicitly shows the distribution of electrons among energy levels and sublevels. The valence electrons are the electrons in the highest principal quantum number (n).
- Using Lewis Dot Structures: Lewis dot structures use dots to represent valence electrons, providing a visual representation of an atom's valence electrons.
Beyond the Basics: More Complex Scenarios
The concept of valence electrons, while seemingly straightforward, encompasses nuances and complexities. Transition metals, for instance, display variable valencies, meaning they can have different numbers of valence electrons depending on the chemical environment. Furthermore, the participation of d- and f-block electrons in bonding can introduce further complexity into the discussion of chemical bonding and reactivity.
The study of valence electrons extends beyond basic chemistry, playing a pivotal role in fields such as materials science, where manipulating the electronic structure of materials is essential for designing advanced materials with specific properties. Similarly, understanding the behavior of valence electrons is crucial in understanding semiconductor physics and the operation of electronic devices.
Conclusion: The Importance of Valence Electrons
Valence electrons are not merely abstract concepts; they are the driving force behind the chemical world. Their number, arrangement, and behavior dictate the properties of elements, their reactivity, and the types of bonds they form. A thorough understanding of valence electrons is foundational to comprehending chemical reactions, molecular structures, and the properties of materials, offering insights into the fascinating world of chemistry and beyond. From the simple ionic bond in salt crystals to the complex interactions within semiconductors, valence electrons are at the heart of it all. Their continued study will undoubtedly lead to further breakthroughs in our understanding of the physical world and enable the development of innovative technologies.
Latest Posts
Latest Posts
-
What Is The Difference Between An Equation And Inequality
Apr 05, 2025
-
Does Electric Field Go From Positive To Negative
Apr 05, 2025
-
What Tools Are Used To Measure Humidity
Apr 05, 2025
-
How To Solve Inequalities In Interval Notation
Apr 05, 2025
-
How To Make A Wet Mount
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Electrons In The Outermost Energy Level Of An Atom . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
